Abstract
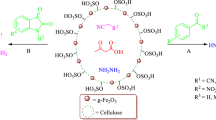
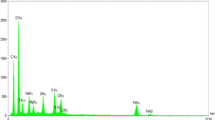
5-Methyl-7-aryl-4,7-dihydrotetrazolo[1,5-a]pyrimidine-6-carboxylic ester derivatives were efficiently synthesized by reaction of methyl or ethyl acetoacetate, 5-aminotetrazole (produced from 2-cyanoguanidine and sodium azide), and various aromatic aldehydes in presence of a new cellulose-based Ag-loaded magnetic bionanostructure. This protocol offers many advantages such as short reaction time, high yield, the remarkable magnetic property of the nanocomposite, and easy separation of the nanocatalyst from the reaction mixture without considerable loss of catalytic activity. Furthermore, X-ray diffraction analysis, field-emission scanning electron microscopy, and energy-dispersive X-ray (EDX) analysis were used to characterize the prepared nanocatalyst.






Similar content being viewed by others
References
F. Bauer, U. Decker, H. Ernst, M. Findeisen, H. Langguth, R. Mehnert, V. Sauerland, R. Hinterwaldner, Int. J. Adhes. 26, 567–570 (2006)
P. Innocenzi, B. Lebeau, J. Mater. Chem. 15, 3821–3831 (2005)
M. Darder, E. Ruiz-Hitzky, J. Mater. Chem. 15, 3913–3918 (2005)
C. Sanchez, B. Julián, P. Belleville, M. Popall, J. Mater. Chem. 15, 3559–3592 (2005)
Y. Feng, J. Zhang, J. He, J. Zhang, Carbohydr. Polym. 147, 171–177 (2016)
K. Kueseng, K.I. Jacob, Eur. Polym. J. 42, 220–227 (2006)
V.L. Finkenstadt, Appl. Microbiol. Biotechnol. 67, 735–745 (2005)
D. Klemm, B. Heublein, H. Fink, A. Bohn, Angew. Chem. Int. Ed. 44, 3358–3393 (2005)
W. Zhu, W. Li, Y. He, T. Duan, Appl. Surf. Sci. 338, 22–26 (2015)
H. Ullah, F. Wahid, H.A. Santos, T. Khan, Carbohydr. Polym. 150, 330–352 (2016)
S.-M. Li, N. Jia, M.-G. Ma, Z. Zhang, Q.-H. Liu, R.-C. Sun, Carbohydr. Polym. 86, 441–447 (2011)
R.T. Olsson, M.A.S.A. Samir, G. Salazar-Alvarez, L. Belova, V. Ström, L.A. Berglund, O. Ikkala, J. Nogues, U.W. Gedde, Nat. Nanotechnol. 5, 584–588 (2010)
S.J. Eichhorn, A. Dufresne, M. Aranguren, N.E. Marcovich, J.R. Capadona, S.J. Rowan, C. Weder, W. Thielemans, M. Roman, S. Renneckar, J. Mater. Sci. 45, 1–33 (2010)
S.-M. Li, N. Jia, J.-F. Zhu, M.-G. Ma, F. Xu, B. Wang, R.-C. Sun, Carbohydr. Polym. 83, 422–429 (2011)
Y.-Y. Dong, S.-M. Li, M.-G. Ma, K. Yao, R.C. Sun, Carbohydr. Polym. 106, 14–21 (2014)
F. Quignard, A. Choplin, Chem. Commun. 1, 21–22 (2001)
E. Coronado, C. Martí-Gastaldo, E. Navarro-Moratalla, A. Ribera, S.J. Blundell, P.J. Baker, Nat. Chem. 2, 1031–1036 (2010)
S. Lin, Y. Cheng, J. Liu, M.R. Wiesner, Langmuir 28, 4178–4186 (2012)
U. Chatterjee, S.K. Jewrajka, S. Guha, Polym. Compos. 30, 827–834 (2009)
A. Maleki, H. Movahed, R. Paydar, RSC Adv. 6, 13657–13665 (2016)
A. Maleki, A. Sarvary, RSC Adv. 5, 60938–60955 (2015)
A. Sarvary, A. Maleki, Mol. Divers. 19, 189–212 (2015)
O.V. Fedorova, M.S. Zhidovinova, G.L. Rusinov, I.G. Ovchinnikova, Russ. Chem. Bull. 52, 1768–1769 (2003)
J. Hashim, N. Arshad, I. Khan, S. Nisar, B. Ali, M.I. Choudhary, Tetrahedron 70, 8582–8587 (2014)
P.N. Gaponik, S.V. Voitekhovich, O.A. Ivashkevich, Russ. Chem. Rev. 75, 507–539 (2006)
V.A. Chebanov, Y.I. Sakhno, S.M. Desenko, S.V. Shishkina, V.I. Musatov, O.V. Shishkin, I.V. Knyazeva, Synthesis 2005, 2597–2601 (2005)
J. Zhu, H. Bienaymé, Multicomponent reactions (Wiley, Hoboken, 2006)
L. Weber, Curr. Med. Chem. 9, 2085–2093 (2002)
A. Maleki, H. Movahed, P. Ravaghi, Carbohydr. Polym. 156, 259–267 (2017)
A. Maleki, H. Movahed, P. Ravaghi, T. Kari, RSC Adv. 6, 98777–98787 (2016)
A. Maleki, M. Aghaei, H.R. Hafizi-Atabak, M. Ferdowsi, Ultrason. Sonochem. 37, 260–266 (2017)
A. Maleki, M. Aghaei, R. Paydar, J. Iran. Chem. Soc. 14, 485–490 (2017)
A. Maleki, T. Kari, M. Aghaei, J. Porous Mater. doi: 10.1007/s10934-017-0388-z
C. Yao, S. Lei, C. Wang, C. Yu, S. Tu, J. Heterocycl. Chem. 45, 1609–1613 (2008)
V.L. Gein, I.N. Vladimirov, O.V. Fedorova, A.A. Kurbatova, N.V. Nosova, I.V. Krylova, M.I. Vakhrin, Russ. J. Org. Chem. 46, 699–705 (2010)
A. Haleel, P. Arthi, N.D. Reddy, V. Veena, N. Sakthivel, Y. Arun, P.T. Perumal, A.K. Rahiman, RSC Adv. 4, 60816–60830 (2014)
A. Haleel, D. Mahendiran, V. Veena, N. Sakthivel, A.K. Rahiman, Mater. Sci. Eng. C 68, 366–382 (2016)
Acknowledgements
The authors gratefully acknowledge partial support from the Research Council of the Iran University of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maleki, A., Ravaghi, P., Aghaei, M. et al. A novel magnetically recyclable silver-loaded cellulose-based bionanocomposite catalyst for green synthesis of tetrazolo[1,5-a]pyrimidines. Res Chem Intermed 43, 5485–5494 (2017). https://doi.org/10.1007/s11164-017-2941-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-017-2941-4