Abstract
Background and aims
Plant nutrient uptake is affected by environmental stress, but how plants respond to cation-nutrient stress is poorly understood. We assessed the impact of varying degrees of cation-nutrient stress on cation uptake in an experimental plant-mineral system.
Methods
Column experiments, with red pine (Pinus resinosa Ait.) seedlings growing in sand/mineral mixtures, were conducted for up to 9 months. The Ca and K were supplied from both minerals and nutrient solutions with varying Ca and K concentrations.
Results
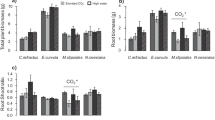
Cation nutrient stress had little impact on carbon allocation after 9 months of plant growth and K was the limiting nutrient for biomass production. Measurement of Ca/Sr and K/Rb ratios allowed independent estimation of dissolution incongruency and discrimination against Sr and Rb during cation uptake processes. The fraction of K in biomass from biotite increased with decreasing K supply from nutrient solutions. The mineral anorthite was consistently the major source of Ca, regardless of nutrient treatment.
Conclusions
Red pine seedlings exploited more mineral K in response to more severe K deficiency. This did not occur for Ca since Ca was not limiting plant growth. Plant discrimination factors must be carefully considered to accurately identify nutrient sources using cation tracers.







Similar content being viewed by others
References
Augusto L, Turpault MP, Ranger J (2000) Impact of forest tree species on feldspar weathering rates. Geoderma 96(3):215–237. doi:10.1016/s0016-7061(00)00021-5
Bakker MR, George E, Turpault MP, Zhang JL, Zeller B (2004) Impact of Douglas-fir and Scots pine seedlings on plagioclase weathering under acidic conditions. Plant Soil 266(1–2):247–259
Baligar VC (1985) Potassium uptake by plants, as characterized by root density, species and K/Rb ratio. Plant Soil 85(1):43–53. doi:10.1007/bf02197799
Balogh-Brunstad Z, Keller CK, Bormann BT, O’Brien R, Wang D, Hawley G (2008a) Chemical weathering and chemical denudation dynamics through ecosystem development and disturbance. Global Biogeochem Cy 22(1):Gb1007. doi:10.1029/2007gb002957
Balogh-Brunstad Z, Keller CK, Gill RA, Bormann BT, Li CY (2008b) The effect of bacteria and fungi on chemical weathering and chemical denudation fluxes in pine growth experiments. Biogeochem 88(2):153–167. doi:10.1007/s10533-008-9202-y
Beauregard F, Cote B (2008) Test of soil extractants for their suitability in predicting Ca/Sr ratios in leaves and stems of sugar maple seedlings. Biogeochem 88(2):195–203. doi:10.1007/s10533-008-9208-5
Blum A, Lasaga A (1988) Role of surface speciation in the low-temperature dissolution of minerals. Nature 331(6155):431–433
Blum JD, Klaue A, Nezat CA, Driscoll CT, Johnson CE, Siccama TG, Eagar C, Fahey TJ, Likens GE (2002) Mycorrhizal weathering of apatite as an important calcium source in base-poor forest ecosystems. Nature 417(6890):729–731. doi:10.1038/nature00793
Blum JD, Dasch AA, Hamburg SP, Yanai RD, Arthur MA (2008) Use of foliar Ca/Sr discrimination and (87)Sr/(86)Sr ratios to determine soil Ca sources to sugar maple foliage in a northern hardwood forest. Biogeochem 87(3):287–296. doi:10.1007/s10533-008-9184-9
Blum JD, Hamburg SP, Yanai RD, Arthur MA (2012) Determination of foliar Ca/Sr discrimination factors for six tree species and implications for Ca sources in northern hardwood forests. Plant Soil 356(1–2):303–314. doi:10.1007/s11104-011-1122-2
Bullen TD, Bailey SW (2005) Identifying calcium sources at an acid deposition-impacted spruce forest: a strontium isotope, alkaline earth element multi-tracer approach. Biogeochem 74(1):63–99. doi:10.1007/s10533-004-2619-z
Clarkson DT (1984) Calcium-transport between tissues and its distribution in the plant. Plant Cell Environ 7(6):449–456. doi:10.1111/j.1365-3040.1984.tb01435.x
Dasch AA, Blum JD, Eagar C, Fahey TJ, Driscoll CT, Siccama TG (2006) The relative uptake of Ca and Sr into tree foliage using a whole-watershed calcium addition. Biogeochem 80(1):21–41. doi:10.1007/s10533-005-6008-z
Deer WA, Howie RA, Zussman J (1992) An introduction to the rock-forming minerals, 2nd edn. John Wiley & Sons, Inc, New York
Drever JI (1994) The effect of land plants on weathering rates of silicate minerals. Geochim Cosmochim Ac 58(10):2325–2332. doi:10.1016/0016-7037(94)90013-2
Drever JI (1997) The geochemistry of natural waters: Surface and groundwater envrionments. 3rd edn. Prentice Hall
Drobner U, Tyler G (1998) Conditions controlling relative uptake of potassium and rubidium by plants from soils. Plant Soil 201(2):285–293. doi:10.1023/a:1004319803952
Drouet T, Herbauts J (2008) Evaluation of the mobility and discrimination of Ca, Sr and Ba in forest ecosystems: consequence on the use of alkaline-earth element ratios as tracers of Ca. Plant Soil 302(1–2):105–124. doi:10.1007/s11104-007-9459-2
Ericsson T (1995) Growth and shoot—root ratio of seedlings in relation to nutrient availability. Plant Soil 168:205–214. doi:10.1007/bf00029330
Federer CA, Hornbeck JW, Tritton LM, Martin CW, Pierce RS, Smith CT (1989) Long-term depletion of calcium and other nutrients in eastern-United-States forests. Environ Manage 13(5):593–601. doi:10.1007/bf01874965
Fink S (1991) Structural-changes in conifer needles due to Mg-deficiency and K-deficiency. Fert Res 27(1):23–27. doi:10.1007/bf01048605
Ingestad T (1979) Mineral Nutrient Requirements of Pinus sill’estris and Picea abies seedlings. Physio Plant 45:373–380
Kelly EF, Chadwick OA, Hilinski TE (1998) The effect of plants on mineral weathering. Biogeochem 42(1–2):21–53. doi:10.1023/a:1005919306687
Koide RT, Kabir Z (2001) Nutrient economy of red pine is affected by interactions between Pisolithus tinctorius and other forest-floor microbes. New Phytol 150(1):179–188. doi:10.1046/j.1469-8137.2001.00073.x
Likens GE, Driscoll CT, Buso DC, Siccama TG, Johnson CE, Lovett GM, Ryan DF, Fahey T, Reiners WA (1994) The biogeochemistry of potassium at Hubbard Brook. Biogeochem 25(2):61–125
Likens GE, Driscoll CT, Buso DC, Siccama TG, Johnson CE, Lovett GM, Fahey TJ, Reiners WA, Ryan DF, Martin CW, Bailey SW (1998) The biogeochemistry of calcium at Hubbard Brook. Biogeochem 41(2):89–173. doi:10.1023/a:1005984620681
Lucash MS, Yanai RD, Blum JD, Park BB (2012) Foliar nutrient concentrations related to soil sources across a range of sites in the Northeastern United States. Soil Sci Soc Am J 76(2):674–683. doi:10.2136/sssaj2011.0160
Marschner H (1995) Mineral nutrition of higher plants, 2nd edn. Academic Press, London
Marschner H, Kirkby EA, Cakmak I (1996) Effect of mineral nutritional status on shoot-root partitioning of photoassimilates and cycling of mineral nutrients. J Exp Bot 47:1255–1263. doi:10.1093/jxb/47.Special_Issue.1255
Perry DA, Oren R, Hart SC (2008) Forest Ecosystems, 2nd edn. The Johns Hopkins University Press, Baltimore
Plank (1992) Plant analysis reference procedures for the southern region of the United States http://www.cropsoil.uga.edu/~oplank/sera368.pdf. Accessed August 31, 2013
Poszwa A, Dambrine E, Pollier B, Atteia O (2000) A comparison between Ca and Sr cycling in forest ecosystems. Plant Soil 225(1–2):299–310. doi:10.1023/a:1026570812307
Price JR, Hardy CR, Tefend KS, Szymanski DW (2012) Solute geochemical mass-balances and mineral weathering rates in small watersheds II: Biomass nutrient uptake, more equations in more unknowns, and land use/land cover effects. Appl Geochem 27(6):1247–1265. doi:10.1016/j.apgeochem.2012.01.020
Rantanen L, Palomaki V, Harrison AF, Lucas PW, Mansfield TA (1994) Interactions between combined exposure to SO2 and NO2 and nutrient status of trees—effects on nutrient content and uptake, growth, needle ultrastructure and pigments. New Phytol 128(4):689–701. doi:10.1111/j.1469-8137.1994.tb04033.x
Reddy ASN (2001) Calcium: silver bullet in signaling. Plant Sci 160(3):381–404. doi:10.1016/s0168-9452(00)00386-1
Semhi K, Clauer N, Chaudhuri S (2012) Variable element transfers from an illite-rich substrate to growing plants during a three-month experiment. Appl Clay Sci 57:17–24. doi:10.1016/j.clay.2011.12.002
Tyler G (1983) Rubidium—availability and plant uptake in natural soils. Commun Soil Sci Plan 14(11):1075–1089. doi:10.1080/00103628309367433
Uroz S, Calvaruso C, Turpault MP, Frey-Klett P (2009) Mineral weathering by bacteria: ecology, actors and mechanisms. Trends Microbiol 17(8):378–387. doi:10.1016/j.tim.2009.05.004
Van Scholl L, Smits MM, Hoffland E (2006) Ectomycorrhizal weathering of the soil minerals muscovite and hornblende. New Phytol 171(4):805–814. doi:10.1111/j.1469-8137.2006.01790.x
White AF, Schulz MS, Vivit DV, Bullen TD, Fitzpatrick J (2012) The impact of biotic/abiotic interfaces in mineral nutrient cycling: A study of soils of the Santa Cruz chronosequence, California. Geochim Cosmochim Ac 77:62–85. doi:10.1016/j.gca.2011.10.029
Witter E, Johansson G (2001) Potassium uptake from the subsoil by green manure crops. Biol Agric Hortic 19(2):127–141
Acknowledgments
We thank Jeff Boyle, Scott Boroughs, and Charles Knaack at Washington State University for analytical assistance and helpful discussions. We thank our collaborators at Washington State University, particularly generous and ongoing accommodations within the School of Biological Sciences Plant Growth Service Center. This work is supported by National Science Foundation grant No. 0952399 to Kent Keller and collaborators. A portion of this research was performed at the Environmental Molecular Sciences laboratory (EMSL), a national scientific user facility sponsored by the U.S. DOE’s Office of Biological and Environmental Research (OBER), located at the Pacific Northwest National Laboratory (PNNL). PNNL is operated for the DOE by Battelle Memorial Institute under contract DE-AC06-76RLO 1830.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: N. Jim Barrow.
Rights and permissions
About this article
Cite this article
Shi, Z., Balogh-Brunstad, Z., Grant, M. et al. Cation uptake and allocation by red pine seedlings under cation-nutrient stress in a column growth experiment. Plant Soil 378, 83–98 (2014). https://doi.org/10.1007/s11104-013-2016-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-013-2016-2