Abstract
Purpose
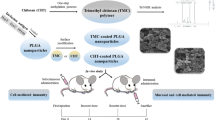
The present report embarks on rational designing of stable and functionalized chitosan nanoparticles for oral mucosal immunization.
Methods
Stable glucomannosylated sCh-GM-NPs were prepared by tandem cross linking method followed by lyophilization. The in vitro stability of antigen and formulation, cellular uptake and immunostimulatory response were assessed by suitable experimental protocol.
Results
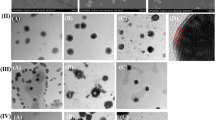
Stability testing ensured the chemical and conformation permanency of encapsulated TT as well as robustness of sCh-GM-NPs in simulated biological media. The antigen release from sCh-GM-NPs followed initial burst followed by controlled Weibull’s type of release profile. The higher intracellular uptake of sCh-GM-NPs in Raw 264.7 and Caco-2 was concentration and time dependent which mainly attributed to Clathrin and receptor mediated endocytosis via mannose and glucose receptor. The in vivo evaluation in animals revealed that sCh-GM-NPs posed significantly (p < 0.001) higher humoral, mucosal and cellular immune response than other counterparts. More importantly, commercial TT vaccine administered through oral or intramuscular route was unable to elicit all type of immune response.
Conclusion
The sCh-GM-NPs could be considered as promising vaccine adjuvant for oral tetanus immunization. Additionally, this technology expected to benefit the design and development of stable peroral formulation for administration of protein, peptides and variety of other antigens.







Similar content being viewed by others
References
World Health Organization. Global vaccine action plan. 2011–2020. 2013.
Arora NK, Lal AA, Hombach JM, Santos JI, Bhutta ZA, Sow SO, et al. The need for targeted implementation research to improve coverage of basic vaccines and introduction of new vaccines. Vaccine. 2013;31(2):B129–36.
Jain S, Harde H, Indulkar A, Agrawal AK. Improved stability and immunological potential of tetanus toxoid containing surface engineered bilosomes following oral administration. Nanomedicine. 2014;10(2):431–40.
Centers for Disease Control Prevention. Global routine vaccination coverage-2012. MMWR Morb Mortal Wkly Rep. 2013;62(43):858–61.
Harde H, Das M, Jain S. Solid lipid nanoparticles: an oral bioavailability enhancer vehicle. Expert Opin Drug Deliv. 2011;8(11):1407–24.
Thanki K, Gangwal R, Sangamwar AT, Jain S. Oral delivery of anticancer drugs: challenges and opportunities. J Control Release. 2013;170(1):15–40.
Lopez Leon T, Carvalho ELS, Seijo B, Ortega Vinuesa JL, Bastos Gonzalez D. Physicochemical characterization of chitosan nanoparticles: electrokinetic and stability behavior. J Colloid Interface Sci. 2005;283(2):344–51.
Sonaje K, Chen Y-J, Chen H-L, Wey S-P, Juang J-H, Nguyen H-N, et al. Enteric-coated capsules filled with freeze-dried chitosan/poly (γ-glutamic acid) nanoparticles for oral insulin delivery. Biomaterials. 2010;31(12):3384–94.
Makhlof A, Tozuka Y, Takeuchi H. Design and evaluation of novel pH-sensitive chitosan nanoparticles for oral insulin delivery. Eur J Pharm Sci. 2011;42(5):445–51.
Sarmento B, Ribeiro A, Veiga F, Sampaio P, Neufeld R, Ferreira D. Alginate/chitosan nanoparticles are effective for oral insulin delivery. Pharm Res. 2007;24(12):2198–206.
Jain S, Indulkar A, Harde H, Agrawal AK. Oral mucosal immunization using glucomannosylated bilosomes. J Biomed Nanotechnol. 2014;10(6):932–47.
Pan Y, Li Y, Zhao H, Zheng J, Xu H, Wei G, et al. Bioadhesive polysaccharide in protein delivery system: chitosan nanoparticles improve the intestinal absorption of insulin in vivo. Int J Pharm. 2002;249(1–2):139–47.
Fernandez-Urrusuno R, Calvo P, Remunan-Lopez C, Vila-Jato JL, Alonso MJ. Enhancement of nasal absorption of insulin using chitosan nanoparticles. Pharm Res. 1999;16(10):1576–81.
Ma Z, Yeoh HH, Lim LY. Formulation pH modulates the interaction of insulin with chitosan nanoparticles. J Pharm Sci. 2002;91(6):1396–404.
Jain S, Chauhan DS, Jain AK, Swarnakar NK, Harde H, Mahajan RR, et al. Inventors A universal step-wise freeze drying process for lyophilization of pharmaceutical products. Indian Patent Application No. 2559/DEL/2011. Filed on 2011.
Harde H, Agrawal AK, Jain S. Development of stabilized glucomannosylated Ch-NPs using tandem crosslinking method for oral vaccine delivery. Nanomedicine. 2014. doi:10.2217/NNM.13.225.
Jain S, Vyas S. Mannosylated niosomes as adjuvant-carrier system for oral mucosal immunization. J Lipos Res. 2006;16(4):331–45.
Lyng J, Bentzon MW. The quantitative estimation of diphtheria and tetanus toxoids. 1. The flocculation test and the Lf-unit. J Biol Stand. 1987;15(1):27–37.
Jain S, Rathi VV, Jain AK, Das M, Godugu C. Folate-decorated PLGA nanoparticles as a rationally designed vehicle for the oral delivery of insulin. Nanomedicine. 2012;7(9):1311–37.
Shan X, Liu C, Yuan Y, Xu F, Tao X, Sheng Y, et al. In vitro macrophage uptake and in vivo biodistribution of long-circulation nanoparticles with poly(ethylene-glycol)-modified PLA (BAB type) triblock copolymer. Colloids Surf B. 2009;72(2):303–11.
Jain AK, Swarnakar NK, Godugu C, Singh RP, Jain S. The effect of the oral administration of polymeric nanoparticles on the efficacy and toxicity of tamoxifen. Biomaterials. 2011;32(2):503–15.
Harde H, Agrawal AK, Jain S. Trilateral ‘3P’ mechanics of stabilized layersomes technology for efficient oral immunization. J Biomed Nanotechnol. 2014;10:1–19. doi:10.1166/jbn.2014.1913.
Agrawal AK, Harde HP, Thanki K, Jain S. Improved stability and antidiabetic potential of insulin containing folic acid functionalized polymer stabilized multilayered liposomes following oral administration. Biomacromolecules. 2014;15(1):350–60.
Sarti F, Perera G, Hintzen F, Kotti K, Karageorgiou V, Kammona O, et al. In vivo evidence of oral vaccination with PLGA nanoparticles containing the immunostimulant monophosphoryl lipid A. Biomaterials. 2011;32(16):4052–7.
Singh M, Li X-M, Wang H, McGee J, Zamb T, Koff W, et al. Immunogenicity and protection in small-animal models with controlled-release tetanus toxoid microparticles as a single-dose vaccine. Infect Immun. 1997;65(5):1716–21.
Men Y, Thomasin C, Merkle HP, Gander B, Corradin G. A single administration of tetanus toxoid in biodegradable microspheres elicits T cell and antibody responses similar or superior to those obtained with aluminum hydroxide. Vaccine. 1995;13(7):683–9.
Gan Q, Wang T, Cochrane C, McCarron P. Modulation of surface charge, particle size and morphological properties of chitosan-TPP nanoparticles intended for gene delivery. Colloids Surf B. 2005;44(2–3):65–73.
Takigawa T, Endo Y. Effects of glutaraldehyde exposure on human health. J Occup Health. 2006;48(2):75–87.
Silin DS, Lyubomska OV, Jirathitikal V, Bourinbaiar AS. Oral vaccination: where we are? Expert Opin Drug Deliv. 2007;4(4):323–40.
Thomasin C, Corradin G, Men Y, Merkle HP, Gander B. Tetanus toxoid and synthetic malaria antigen containing poly (lactide)/poly (lactide-co-glycolide) microspheres: importance of polymer degradation and antigen release for immune response. J Control Release. 1996;41(1):131–45.
Keler T, Ramakrishna V, Fanger MW. Mannose receptor-targeted vaccines. Expert Opin Biol Ther. 2004;4(12):1953–62.
Al-Hasani H, Hinck CS, Cushman SW. Endocytosis of the glucose transporter GLUT4 is mediated by the GTPase dynamin. J Biol Chem. 1998;273(28):17504–10.
Desai MP, Labhasetwar V, Walter E, Levy RJ, Amidon GL. The mechanism of uptake of biodegradable microparticles in Caco-2 cells is size dependent. Pharm Res. 1997;14(11):1568–73.
Jain AK, Thanki K, Jain S. Solidified self-nanoemulsifying formulation for oral delivery of combinatorial therapeutic regimen: part I. Formulation development, statistical optimization, and in vitro characterization. Pharm Res. 2014;31(4):923–45.
Mathiowitz E, Jacob JS, Jong YS, Carino GP, Chickering DE, Chaturvedi P, et al. Biologically erodable microspheres as potential oral drug delivery systems. Nature. 1997;386(6623):410–4.
Des Rieux A, Fievez V, Garinot M, Schneider YJ, Préat V. Nanoparticles as potential oral delivery systems of proteins and vaccines: a mechanistic approach. J Control Release. 2006;116(1):1–27.
O’Hagan DT, Rappuoli R. Novel approaches to vaccine delivery. Pharm Res. 2004;21(9):1519–30.
Borrow R, Balmer P, Roper M. The immunological basis for immunization series. Module 3: tetanus. Geneva: Department of Immunization, Vaccines and Biologicals. World Health Organization; 2006. p. 8–10.
Plotkin SA. Correlates of protection induced by vaccination. Clin Vaccine Immunol. 2010;17(7):1055–65.
Acknowledgments and Disclosures
Authors are grateful to Dr. M. L Mago and Lavit Jambu, Panacea Biotech, Lalru, Punjab, India for gift samples of TT, tetanus antitoxin, and necessary training. Authors are also thankful to Department of Biotechnology (DBT), Govt. of India, India for financial assistance, Council of Scientific and Industrial Research (CSIR) Govt. of India, India for providing fellowship to Mr. AKA, and Director, NIPER, SAS Nagar for providing necessary infrastructure facilities. Technical assistance provided by Mr. Rahul Mahajan in SEM analysis and Mr. Vinod Kumar in TEM analysis is also duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 99 kb)
Rights and permissions
About this article
Cite this article
Harde, H., Agrawal, A.K. & Jain, S. Tetanus Toxoids Loaded Glucomannosylated Chitosan Based Nanohoming Vaccine Adjuvant with Improved Oral Stability and Immunostimulatory Response. Pharm Res 32, 122–134 (2015). https://doi.org/10.1007/s11095-014-1449-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-014-1449-5