ABSTRACT
Purpose
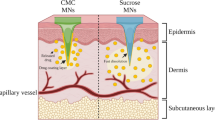
Feasibility study of two-layered dissolving microneedles for percutaneous delivery of peptide/proteins using recombinant human growth hormone (rhGH) and desmopressin (DDAVP).
Methods
Two-layered dissolving microneedles were administered percutaneously to the rat skin. Plasma rhGH and DDAVP concentrations were measured by EIA and LC/MS/MS. In vivo dissolution and diffusion rates of drugs in the skin were studied using tracer dyes, lissamine green B (LG) for rhGH and evans blue (EB) for DDAVP. Diffusion of drugs vertically into the skin was studied using FITC-dextran (MW = 20 kDa)-loaded dissolving microneedles. Stability experiments were performed at −80°C and 4°C.
Results
The absorption half-lives, t 1/2a, of rhGH and DDAVP from dissolving microneedles were 23.7 ± 4.3–28.9 ± 5.2 and 14.4 ± 2.9–14.1 ± 1.1 min; the extents of bioavailability were 72.8 ± 4.2–89.9 ± 10.0% and 90.0 ± 15.4–93.1 ± 10.3%, respectively. LG and EB disappeared from the administered site within 2 h and 3 h after administration. Five green fluorescein spots were detected at 15 s and enlarged transversally at 30 s. FITC-dextran was delivered into the microcapillaries at 5 min and 10 min. The rhGH and DDAVP were stable in dissolving microneedles for one month at −80°C and 4°C.
Conclusions
Results suggest that the two-layered dissolving microneedles are useful as an immediate-release transdermal DDS for peptide/protein drugs.







Similar content being viewed by others
REFERENCES
Walsh G. Biopharmaceuticals. West Sussex: Wiley; 2003.
Shibata N, Ito Y, Takada K. Pharmacokinetics. In: Gad SC, editor. Handbook of pharmaceutical biotechnology. MA: Wiley-Interscience; 2007. p. 757–814.
Morishita I, Morishita M, Takayama K, Machida Y, Nagai T. Hypoglycemic effect of novel oral microspheres of insulin with protease inhibitor in normal and diabetic rats. Int J Pharm. 1992;78:9–16.
Yamamoto A, Taniguchi T, Rikyuu K, Tsuji T, Murakami M, Muranishi S. Effects of various protease inhibitors on the intestinal absorption and degradation of insulin in rats. Pharm Res. 1994;11:1496–500.
Amino Y, Kawada K, Toi K, Kumashiro I, Fukushima K. Phenylalanine derivatives enhancing intestinal absorption of insulin in mice. Chem Pharm Bull. 1988;36:4426–34.
Morishita M, Morishita I, Takayama K, Machida Y, Nagai T. Site dependent effect of aprotinin, sodium caprate Na2EDTA and sodium glycocholate on intestinal absorption of insulin. Biol Pharm Bull. 1993;16:68–72.
Utoguchi N, Watanabe Y, Shida T, Matsumoto M. Nitric oxide donors enhance rectal absorption of macromolecules in rabbits. Pharm Res. 1998;15:870–6.
Takeuchi H, Yamamoto H, Niwa T, Hino T, Kawashima Y. Enteral absorption of insulin in rats from mucoadhesive chitosan coated liposomes. Pharm Res. 1996;13:896–901.
Tozaki H, Komoike J, Tada C, Maruyama T, Terabe A, Suzuki J. Chitosan capsules for colon-specific drug delivery: improvement of insulin absorption from the rat colon. J Pharm Sci. 1997;86:1016–21.
Matsuzawa A, Morishita M, Takayama K, Nagai T. Absorption of insulin using water-in-oil-in-water emulsion from an enteral loop in rats. Biol Pharm Bull. 1995;18:1718–23.
Cunha SA, Grossiord LJ, Puisieux F, Seiller M. Insulin in W/O/W multiple emulsions: biological activity after oral administration in normal and diabetic rats. J Microencapsul. 1997;14:321–33.
Morishita M, Matsuzawa A, Takayama K, Isowa K, Nagai T. Improving insulin enteral absorption using water-in-oil-in-water emulsion. Int J Pharm. 1998;172:189–98.
Suzuki A, Morishita M, Kajita M, Takayama K, Isowa K, Chiba Y, et al. Enhanced colonic and rectal absorption of insulin using a multiple emulsion containing eicosapentaenoic acid and docosahexaenoic acid. J Pharm Sci. 1998;87:1196–202.
Takada K. Oral delivery of haematopoietic factors: Progress with gastrointestinal mucoadhesive patches, microdevices and other microfabrication technologies. Am J Drug Deliv. 2006;4:65–77.
Khafagy El-S, Morishita M, Isowa K, Imai J, Takayama K. Effect of cell-penetrating peptides on the nasal absorption of insulin. J Control Release 2009;133:103–8.
Kamei N, Morishita M, Takayama K. Importance of intermolecular interaction on the improvement of intestinal therapeutic peptide/protein absorption using cell-penetrating peptides. J Control Release 2009;136:179–86.
Barry B, Williams A. Penetration enhancers. Adv Drug Deliv Rev. 2004;56:603–18.
Cevc G. Lipid vesicles and other colloids as drug carriers on the skin. Adv Drug Deliv Rev. 2004;56:675–711.
Preat V, Vanbever R. Skin electroporation for transdermal and topical delivery. Adv Drug Deliv Rev. 2004;56:659–74.
Doukas A. Transdermal delivery with a pressure wave. Adv Drug Deliv Rev. 2004;56:559–79.
Mitragotri S, Kost J. Low-frequency sonophoresis: a review. Adv Drug Deliv Rev. 2004;56:589–601.
Cormier M, Johnson B, Ameri M, Nyam K, Libiran L, Zhang DD, et al. Transdermal delivery of desmopressin using a coated microneedle array patch system. J Control Release 2004;97:503–11.
Prausnitz RM. Microneedles for transdermal drug delivery. Adv Drug Deliv Rev. 2004;56:581–7.
Levin G, Gershonowitz A, Sacks H, Stern M, Sherman A, Rudaev S, et al. Transdermal delivery of human growth hormone through RF-microchannels. Pharm Res. 2005;22:550–5.
Qiu Y, Gao Y, Hu K, Li F. Enhancement of skin permeation of docetaxel: A novel approach combining microneedle and elastic liposomes. J Control Release 2008;129:144–50.
Wermeling DP, Banks SL, Hudson DA, Gill HS, Gupta J, Prausnitz MR, et al. Microneedles permit transdermal delivery of a skin-impermeant medication to humans. Proc Natl Acad Sci USA. 2008;105:2058–63.
Donnelly RF, Morrow DIJ, McCarron PA, Woolfson AD, Morrissey A, Juzenas P, et al. Microneedle mediated intradermal delivery of 5-aminolevulinic acid: Potential for enhanced topical photodynamic therapy. J Control Release 2008;129:154–62.
Ito Y, Hagiwara E, Saeki A, Sugioka N, Takada K. Feasibility of microneedles for percutaneous absorption of insulin. Eur J Pharm Sci. 2006;29:82–8.
Ito Y, Murakami A, Maeda T, Sugioka N, Takada K. Evaluation of self-dissolving needles containing low molecular weight heparin (LMWH) in rats. Int J Pharm. 2008;349:124–9.
Ito Y, Ohashi Y, Shiroyama K, Sugioka N, Takada K. Self-dissolving micropiles for the percutaneous absorption of human growth hormone in rats. Biol Pharm Bull. 2008;31:1631–3.
Ito Y, Yoshimitsu J, Shiroyama K, Sugioka N, Takada K. Self-dissolving microneedles fir the percutaneous absorption of EPO in mice. J Drug Target 2006;14:255–62.
Ito Y, Saeki A, Shiroyama K, Sugioka N, Takada K. Percutaneous absorption of interferon-α by self-dissolving micropiles. J Drug Target 2008;16:243–9.
Ito Y, Ohashi Y, Saeki A, Sugioka N, Takada K. Antihyperglycemic effect of insulin from self-dissolving micropiles in dogs. Chem Pharm Bull. 2008;56:243–6.
Takada K. Microfabrication derived DDS: From batch to individual production. Drug Discov Ther. 2008;2:140–55.
Schmitz T, Huck CW, Bernkop-Schnurch A. Characterization of the thiol-disulphide chemistry of desmopressin by LC, μg-LC, LC-ESI-MS and Maldi-Tof. Amino Acids 2006;30:35–42.
Getie M, Neubert RH. LC-MS determination of desmopressin acetate in human skin samples. J Pharm Biomed Anal. 2004;35:921–7.
Kluge M, Riedl S, Erhart-Hofmann B, Hartmann J, Waldhauser F. Improved extraction procedure and RIA for determination of arginine8-vasopressin in plasma: role of premeasurement sample treatment and reference values in children. Clin Chem. 1999;45:98–103.
Agerso H, Seiding Larsen L, Riis A, Lovgren U, Karlsson MO, Senderovitz T. Pharmacokinetics and renal excretion of desmopressin after intravenous administration to healthy subjects and renally impaired patients. Br J Clin Pharmacol. 2004;58:352–8.
Gibaldi M, Perrier D. Pharmacokinetics. New York: Marcel Dekker; 2006.
Ito Y, Ise A, Sugioka N, Takada K. Molecular weight dependence on bioavailabity of FITC-dextran after administration of self-dissolving micropile to rat skin. Drug Dev Ind Pharm. in press (2010).
Monteiro-Riviere AN, Bristol GD, Manning OT, Rogers AR, Riviere EJ. Interspecies and interregional analysis of the comparative histologic thickness and laser Doppler blood flow measurements at five cutaneous sites in nine species. J Invest Dermatol. 1990;95:582–6.
Bauer J, Bahmer AF, Worl J, Neuhuber W, Schuler G, Fartasch M. A strikingly constant ration exists between Langerhans cells and other epidermal cells in human skin. A stereologic study using the optical dissector method and the confocal laser scanning microscope. J Invest Dermatol. 2001;116:313–8.
Wermeling PD, Banks LS, Hudson AD, Gill SH, Gupta J, Prausnitz RM, et al. Microneedles permit transdermal delivery of a skin-impermeant medication to human. Proc Natl Acad Sci USA. 2008;105:2058–2063.
Caspers JP, Lucassen WG, Bruining AH, Puppels JG. Automated depth-scanning confocal Raman microspectrometer for rapid in vivo determination of water concentration profiles in human skin. J Raman Spectrosc. 2000;31:813–8.
Ito Y, Hasegawa R, Fukushima K, Sugioka N, Takada K. Self-dissolving micropile array chip as percutaneous delivery system of protein drug. Biol Pharm Bull. in press (2010).
Kolli CS, Banga AK. Charcterization of solid maltose microneedles and their use for transdermal delivery. Pharm Res. 2007;25:104–13.
Wermeling PD, Banks LS, Hudson AD, Gill SH, Gupta J, Prausnitz RM, et al. Micropiles permit transdermal delivery of a skin-impermeant medication to humans. Proc Natl Acad Sci USA. 2008;105:2058–63.
Duan H-G, Wei Y-H, Li B-X, Qin H-Y, Wu X-A. Improving the dissolution and oral bioavailability of the poorly water-soluble drug aloe-emodin by solid dispersion with polyethylene glycol 6000. Drug Dev Res. 2009;70:363–9.
Park YJ, Kwon R, Quan ZQ, Oh HD, Kim OJ, Hwang RM, et al. Development of novel ibuprofen-loaded solid dispersion with improved bioavailability using aqueous solution. Arch Pharm Res. 2009;32:767–72.
Kennedy M, Hu J, Gao P, Li L, Ali-Reynolds A, Chal B, et al. Enhanced bioavailability of a poorly soluble VR1 antagonist using an amorphous solid dispersion approach: a case study. Mol Pharm. 2008;5:981–93.
Fukushima K, Haraya K, Terasaka S, Ito Y, Sugioka N, Takada K. Long-term pharmacokinetic efficacy and safety of low-dose ritonavir as a booster and atazanavir pharmaceutical formulation based on solid dispersion system in rats. Biol Pharm Bull. 2008;31:1209–14.
Vasconcelos T, Sarmento B, Costa P. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov Today 2007;12:1068–75.
Cheng YH, Dyer AM, Jabbal-Gill I, Hinchcliffe M, Nankervis R, Smith A, et al. Intranasal delivery of recombinant human growth hormone (somatropin) in sheep using chitosan-based powder formulations. Eur J Pharm Sci. 2005;26:9–15.
Moore JA, Pletcher SA, Ross MJ. Absorption enhancement of growth hormone from the gastrointestinal tract of rats. Int J Pharm. 1986;34:35–43.
Li H, Song JW, Park JS, Han K. Polyethylene glycol-coated liposomes for oral delivery of recombinant human epidermal growth factor. Int J Pharm. 2003;258:11–9.
Lam XM, Duenas ET, Cleland JL. Encapsulation and stabilization of nerve growth factor into poly (lactic-co-glycolic) acid microspheres. J Pharm Sci. 2001;90:1356–65.
Kim HK, Park TG. Microencapsulation of dissociable human growth hormone aggregates within poly (D, L-lactic-co-glycolic) acid microparticles for sustained release. Int J Pharm. 2001;229:107–16.
Joukhadar C, Schenk B, Kaehler ST, Kollenz CJ, Bauer P, Muller M, et al. A replicate study design for testing bioequivalence: a case study on two desmopressin nasal spray preparations. Eur J Clin Pharmacol. 2003;59:631–6.
Steiner IM, Kaehler ST, Sauermann R, Rinosl H, Muller M, Joukhadar C. Plasma pharmacokinetics of desmopressin following sublingual administration: an exploratory dose-escalation study in healthy male volunteers. Int J Clin Pharm Ther. 2006;44:172–9.
Cormier M, Johnson B, Ameri M, Nyam K, Libiran L, Zhang D, et al. Transdermal delivery of desmopressin using a coated microneedle array patch system. J Control Release 2004;97:503–11.
de Jager MW, Ponec M, Bouwstra JA. The lipid organization in stratum corneum and model systems based on ceramides. In: Touitou E, Barry BW, editors. Enhancement in drug delivery. London: CRC; 2007. p. 217–32.
Chabri F, Bouris K, Jones T, Barrow D, Hann A, Allender C, et al. Microfabricated silicon microneedles for nonviral cutancous gene delivery. Br J Dermatol. 2004;150:869–77.
Gardeniers HJGE, Luttge R, Berenschot EJW, de Boer MJ, Yeshurun SY, Hefetz M, van’t Oever R, van den Berg A. Silicon micromachined hollow microneedles for transdermal liquid transport. J Microelectromech Syst. 2003;12:855–62.
Ito Y, Hagiwara E, Saeki A, Sugioka N, Takada K. Feasibility of microneedles for percutaneous absorption of insulin. Eur J Pharm Sci. 2006;29:82–8.
Davis PS, Martanto W, Allen GM, Prausnitz RM. Hollow metal micropiles for insulin delivery to diabetic rats. IEEE Trams Biomed Eng. 2005;52:909–15.
Arora A, Prausnitz MR, Mitragotri S. Micro-scale devices for transdermal drug delivery. Int J Pharm. 2008;364:227–36.
Morawski M, Alpár A, Brückner G, Fiedler A, Jäger C, Gati G, et al. Chondroitin sulfate proteoglycan-based extracellular matrix in chicken (Gallus domesticus) brain. Brain Res. 2009;1275:10–23.
Faissner A, Clement A, Lochter A, Streit A, Mandl C, Schachner M. Isolation of a neural chondmitin sulfate proteoglycan with neurite outgrowth promoting properties. J Cell Biol. 1994;126:783–99.
Umehara Y, Yamada S, Nishimura S, Shioi J, Robakis NK, Sugahara K. Chondroitin sulfate of appican, the proteoglycan form of amyloid precursor protein, produced by C6 glioma cells interacts with heparin-binding neuroregulatory factors. FEBS Lett. 2004;557:233–8.
Wilson TM, Snow DM. Chondroitin sulfate proteoglycan expression pattern in hippocampal development: potential regulation of axon tract formation. J Comp Neurol. 2000;424:532–46.
Sintov A, Di-Capua N, Rubinstein A. Cross-linked chondroitin sulfate: characterization for drug delivery purposes. Biomaterials 1995;16:473–8.
Tsai FM, Chiang LY, Wang FL, Huang WG, Wu CP. Oral sustained delivery of diclofenac sodium using calcium chondroitin sulfate matrix. J Biomater Sci Polym Ed. 2005;16:1319–l331.
Lee JW, Park J-H, Prausnitz MR. Dissolving microneedles for transdermal drug delivery. Biomaterials 2008;29:2113–24.
ACKNOWLEDGMENTS
This study was supported by a strategic fund of MEXT (Ministry of Education, Culture, Sports, Science and Technology, MEXT) from 2008 to 2013 for establishing research foundation in private universities of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukushima, K., Ise, A., Morita, H. et al. Two-Layered Dissolving Microneedles for Percutaneous Delivery of Peptide/Protein Drugs in Rats. Pharm Res 28, 7–21 (2011). https://doi.org/10.1007/s11095-010-0097-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-010-0097-7