Abstract
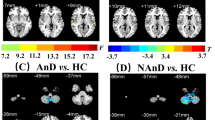
Antidepressant medications represent the most common treatment option for major depressive disorder (MDD), but the neuro-psychological mechanisms by which antidepressants act to improve depressive symptoms remain under-specified. We designed this study to assess the effects of escitalopram treatment on spontaneous brain activity of MDD patients using functional magnetic resonance imaging (fMRI). Twenty first-episode drug-naive MDD patients received resting-state fMRI scans before and after 8 weeks of treatment with a selective serotonin reuptake inhibitor - escitalopram. Twenty age- and gender-matched healthy controls were also scanned twice with an 8-week interval. The fractional amplitude of low-frequency fluctuation (fALFF) was used to characterize the spontaneous brain activity. The analysis of covariance (ANCOVA) was performed to determine treatment-related changes in fALFF. The symptoms were significantly improved in MDD patients after treatment. We observed significant group-by-time interaction on fALFF in the left dorsomedial prefrontal cortex, the right middle frontal gyrus, and the left putamen. Post-hoc analyses showed that the fALFF values in these regions were significantly higher in the MDD patients compared to healthy controls at baseline and were reduced after treatment. The findings suggest that abnormalities in the brain areas involved in emotional processing and regulation could be normalized by effective antidepressant treatment with escitalopram in the MDD patients and free of a task situation.


Similar content being viewed by others
References
Anand A, Li Y, Wang Y, et al. (2005) Antidepressant effect on connectivity of the mood-regulating circuit: an FMRI study. Neuropsychopharmacology 30:1334–1344
Arnone D, McKie S, Elliott R, Thomas EJ, et al. (2012) Increased amygdale responses to sad but not fearful faces in major depression: relation to mood state and pharmacological treatment. Am J Psychiatry 169:841–850
Beauregard M, Paquette V, Lévesque J (2006) Dysfunction in the neural circuitry of emotional selfregulation in major depressive disorder. NeuroReport 17:843–846
Bigos KL, Pollock BG, Aizenstein HJ, et al. (2008) Acute 5-HT reuptake blockade potentiates human amygdala reactivity. Neuropsychopharmacology 33:3221–3225
Biswal B, Yetkin FZ, Haughton VM, et al. (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34:537–541
Buckner RL, Sepulcre J, Talukdar T, et al. (2009) Cortical hubs revealed by intrinsic functional connectivity: mapping, assessment of stability, and relation to Alzheimer's disease. J Neurosci 29:1860–1873
Chao-Gan Y, Yu-Feng Z (2010) DPARSF: a MATLAB toolbox for "pipe0line" data analysis of resting-state fMRI. Front Syst Neurosci 4:13
Del-Ben CM, Deakin JF, Mckie S, et al. (2005) The effect of citalopram pretreatment on neuronal responses to neuropsychological tasks in normal volunteers: an fMRI study. Neuropsychopharmacology 30:1724–1734
Delaveau P, Jabourian M, Lemogne C, et al. (2011) Brain effects of antidepressants in major depression: a meta-analysis of emotionalprocessing studies. J Affect Disord 130:66–74
Epstein J, Pan H, Kocsis JH, et al. (2006) Lack of ventral striatal response to positive stimuli in depressed versus normal subjects. Am J Psychiatry 163:1784–1790
Fox MD, Raichle ME (2007) Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat Rev Neurosci 8:700–711
Fu CH, Williams SC, Cleare AJ, et al. (2004) Attenuation of the neural response to sad faces in major depression by antidepressant treatment: a prospective, event-related functional magnetic resonance imaging study. Arch Gen Psychiatry 61:877–889
Gyurak A, Patenaude B, Korgaonkar MS, et al (2016) Frontoparietal activation during response inhibition predicts remission to antidepressants in patients with major depression. Biol Psychiatry 79:274–281
Hamilton M (1967) Development of a rating scale for primary depressive illness. Br J Soc Clin Psychol 6:278–296
Hasler G, Drevets WC, Husseini KM, et al. (2004) Discovering endophenotypes for major depression. Neuropsychopharmacol 29:1765–1781
He Y, Wang J, Wang L, et al. (2009) Uncovering intrinsic modular organization of spontaneous brain activity in humans. PLoS One 4:e5226
Heller AS, Johnstone T, Peterson MJ, et al. (2013) Increased prefrontal cortex activity during negative emotion regulation as a predictor of depression symptom severity trajectory over 6 months. JAMA Psychiatry 70:1181–1119
Keedwell PA, Andrew C, Williams SC, et al. (2005) A double dissociation of ventromedial prefrontal cortical responses to sad and happy stimuli in depressed and healthy individuals. Biol Psychiatry 58:495–503
Kennedy SH, Evans KR, Krüger S, et al. (2001) Changes in regional brain glucose metabolism measured with positron emission tomography after paroxetine treatment of major depression. Am J Psychiatry 158:899–905
Koenigs M, Grafman J (2009) The functional neuroanatomy of depression: distinct roles for ventromedial and dorsolateral prefrontalcortex. Behav Brain Res 201:239–243
Kupfer DJ, Frank E, Phillips ML (2012) Major depressive disorder: new clinical, neurobiological, and treatment perspectives. Lancet 379:1045–1055
Lanzenberger R, Kranz GS, Haeusler D, et al. (2012) Prediction of SSRI treatment response in major depression based on serotonin transporter interplay between median raphe nucleus and projection areas. NeuroImage 63:874–881
Ledberg A, Akerman S, Roland PE (1998) Estimation of the probabilities of 3D clusters in functional brain images. NeuroImage 8:113–128
Lepine JP, Briley M (2011) The increasing burden of depression. Neuropsychiatr Dis Treat 7:3–7
Liu Y, Du L, Li Y, et al. (2015) Antidepressant effects of electroconvulsive therapy correlate with subgenual anterior cingulate activity and connectivity in depression. Medicine (Baltimore) 94:e2033
Ma Y (2015) Neuropsychological mechanism underlying antidepressant effect: a systematic meta-analysis. Mol Psychiatry 20:311–319
Maier W, Buller R, Philipp M, et al. (1988) The Hamilton anxiety scale: reliability, validity and sensitivity to change in anxiety and depressive disorders. J Affect Disord 14:61–68
Mayberg HS, Liotti M, Brannan SK, et al. (1999) Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am J Psychiatry 156:675–682
McCabe C, Mishor Z (2011) Antidepressant medications reduce subcortical-cortical resting-state functional connectivity in healthy volunteers. NeuroImage 57:1317–1323
McCabe C, Mishor Z, Filippini N, et al. (2011) SSRI administration reduces resting state functional connectivity in dorso-medial prefrontal cortex. Mol Psychiatry 16:592–594
O’Doherty J, Dayan P, Schultz J, et al. (2004) Dissociable roles of ventral and dorsal striatum in instrumental conditioning. Science 304:452–454
Perrin JS, Merz S, Bennett DM, et al. (2012) Electroconvulsive therapy reduces frontal cortical connectivity in severe depressive disorder. Proc Natl Acad Sci U S A 109:5464–5468
Pizzagalli DA, Holmes AJ, Dillon DG, et al. (2009) Reduced caudate and nucleus accumbens response to rewards in unmedicated individuals with major depressive disorder. Am J Psychiatry 166:702–710
Posner J, Hellerstein DJ, Gat I, et al. (2013) Antidepressants normalize the default mode network in patients with dysthymia. JAMA Psychiatry 70:373–382
Power JD, Barnes KA, Snyder AZ, et al. (2012) Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. NeuroImage 59:2142–2154
Rosenblau G, Sterzer P, Stoy M, et al. (2012) Functional neuroanatomy of emotion processing in major depressive disorder is altered after successful antidepressant therapy. J Psychopharmacol 26:1424–1433
Schaefer HS, Putnam KM, Benca RM, et al. (2006) Event-related functional magnetic resonance imaging measures of neural activity to positive social stimuli in pre- and post-treatment depression. Biol Psychiatry 60:974–986
Sheehan DV, Lecrubier Y, Sheehan KH, et al. (1998) The mini-international neuropsychiatric interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 59:22–33
Sheline YI, Price JL, Yan Z, et al. (2010) Resting-state functional MRI in depression unmasks increased connectivity between networks via the dorsal nexus. Proc Natl Acad Sci U S A 107:11020–11025
Song XW, Dong ZY, Long XY, et al. (2011) REST: a toolkit for resting-state functional magnetic resonance imaging data processing. PLoS One 6:e25031
Stuhrmann A, Suslow T, Dannlowski U (2011) Facial emotion processing in major depression: a systematic review of neuroimaging findings. Biol Mood Anxiety Disord 1:10
Surguladze S, Brammer MJ, Keedwell P, et al. (2005) A differential pattern of neural response toward sad versus happy facial expressions in major depressive disorder. Biol Psychiatry 57:201–209
Tahmasian M, Knight DC, Manoliu A, et al. (2013) Aberrant intrinsic connectivity of hippocampus and amygdala overlap in the fronto-insular and dorsomedial-prefrontal cortex in major depressive disorder. Front Hum Neurosci 7:639
Tremblay LK, Naranjo CA, Graham SJ (2005) Functional neuroanatomical substrates of altered reward processing in major depressive disorder revealed by a dopaminergic probe. Arch Gen Psychiatry 62:1228–1236
Wang Z, Yan C, Zhao C, et al. (2011) Spatial patterns of intrinsic brain activity in mild cognitive impairment and Alzheimer’s disease: a resting-state functional MRI study. Hum Brain Mapp 32:1720–1740
Wang L, Dai Z, Peng H, et al. (2014) Overlapping and segregated resting-state functional connectivity in patients with major depressive disorder with and without childhood neglect. Hum Brain Mapp 35:1154–1166
Woodward LJ, Clark CA, Bora S, et al. (2012) Neonatal white matter abnormalities an important predictor of neurocognitive outcome for very preterm children. PLoS One 7:e51879
Zang YF, He Y, Zhu CZ, et al. (2007) Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain Dev 29:83–91
Zou QH, Zhu CZ, Yang Y, et al. (2008) An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF. J Neurosci Methods 172:137–141
Acknowledgments
We thank the funding from National Key Technology R&D Program (2015BAI13B01), National Key Basic Research Program of China (973 Program) (2013CB531305; 2012CB720704) and research grants from the “12th Five-year-plan” of National Key Technologies R&D Program of China (2011ZX09302-004).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors report no conflict of interest in conducting this study and preparing the manuscript.
Additional information
Li Wang, Xueni Li contributed equally to the paper.
Electronic Supplementary Material
ESM 1
(DOC 5.25 mb)
Rights and permissions
About this article
Cite this article
Wang, L., Li, X., Li, K. et al. Mapping the effect of escitalopram treatment on amplitude of low-frequency fluctuations in patients with depression: a resting-state fMRI study. Metab Brain Dis 32, 147–154 (2017). https://doi.org/10.1007/s11011-016-9871-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9871-5