Abstract
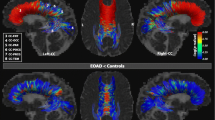
The aim of this study was to evaluate the structural integrity of the thalamic connectivity of specific fiber tracts in different stages of Alzheimer’s disease (AD) using diffusion tensor imaging (DTI). Thirty-five patients with AD and 22 normal control (NC) subjects were recruited. Based on Mini Mental State Examination score, the AD patients were divided into three subgroups for comparison with the NC group: mild (mi-AD, n = 14), moderate (mo-AD, n = 12), and severe (se-AD, n = 9) AD. The fornix (FX), anterior thalamic radiation (ATR), and posterior thalamic radiation (PTR) were selected to represent the thalamic connectivity with other brain regions. The fornix was divided into the column and body of the fornix (FX-1) and the bilateral fornix (crus)/stria terminalis (FX-2/ST) based on the atlas. Through the atlas-based analysis and fiber tracking method, we measured fractional anisotropy (FA), mean diffusivity (MD), and tract volume to reflect the microstructural and macrostructural changes of these fibers during AD progression. There were significant differences in the FA and MD of all fibers, except the right PTR, between the AD and NC subjects. Further subgroup analyses revealed that the mi-AD subgroup had decreased FA only in the FX-1 and increased MD in the FX-1 and bilateral ATR, the mo-AD subgroup showed declined FA and increased MD in the FX-1, bilateral FX-2/ST and ATR; the se-AD subgroup exhibited lower FA and higher MD values in all fibers except the right PTR. We also found reduced tract volume values in the FX and left ATR in the AD patients. Further subgroup analyses revealed that these differences only existed in the se-AD patients. Our DTI analyses indicate that the integrity of thalamic connectivity is progressively disrupted following cognitive decline in AD and that DTI parameters in the column and body of the fornix show promise as potential markers for the early diagnosis of AD and for monitoring disease progression.




Similar content being viewed by others
References
Acosta-Cabronero J, Williams GB, Pengas G, Nestor PJ (2010) Absolute diffusivities define the landscape of white matter degeneration in Alzheimer’s disease. Brain 133:529–539
Agosta F, Pievani M, Sala S, Geroldi C, Galluzzi S, Frisoni GB, Filippi M (2011) White matter damage in Alzheimer disease and its relationship to gray matter atrophy. Radiology 258:853–863
Alexander AL, Hurley SA, Samsonov AA, Adluru N, Hosseinbor AP, Mossahebi P, Tromp do PM, Zakszewski E, Field AS (2011) Characterization of cerebral white matter properties using quantitative magnetic resonance imaging stains. Brain Connectivity 1:423–446
Alzheimer’s Disease International, Alzheimer’s Australia (2015) Dementia warning for the asia-pacific region. The Lancet Neurol 14:1
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders, 4th edn, Washington, DC, p. 390
Bartzokis G (2011) Alzheimer’s disease as homeostatic responses to age-related myelin breakdown. Neurobiol Aging 32:1341–1371
Bozoki AC, Korolev IO, Davis NC, Hoisington LA, Berger KL (2012) Disruption of limbic white matter pathways in mild cognitive impairment and Alzheimer’s disease: a DTI/FDG-PET study. Hum Brain Mapp 33:1792–1802
Bozzali M, Giulietti G, Basile B, Serra L, Spano B, Perri R, Giubilei F, Marra C, Caltagirone C, Cercignani M (2012) Damage to the cingulum contributes to Alzheimer’s disease pathophysiology by deafferentation mechanism. Hum Brain Mapp 33:1295–1308
Braak H, Braak E (1991) Alzheimer’s disease affects limbic nuclei of the thalamus. Acta Neuropathol 81:261–268
Braak H, Alafuzoff I, Arzberger T, Kretzschmar H, Del Tredici K (2006) Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol 112:389–404
Carlson HL, Laliberte C, Brooks BL, Hodge J, Kirton A, Bello-Espinosa L, Hader W, Sherman EM (2014) Reliability and variability of diffusion tensor imaging (DTI) tractography in pediatric epilepsy. Epilepsy Behav 37:116–122
Chaddock-Heyman L, Erickson KI, Voss MW, Powers JP, Knecht AM, Pontifex MB, Drollette ES, Moore RD, Raine LB, Scudder MR, Hillman CH, Kramer AF (2013) White matter microstructure is associated with cognitive control in children. Biol Psychol 94:109–115
Cho H, Kim JH, Kim C, Ye BS, Kim HJ, Yoon CW, Noh Y, Kim GH, Kim YJ, Kim JH, Kim CH, Kang SJ, Chin J, Kim ST, Lee KH, Na DL, Seong JK, Seo SW (2014) Shape changes of the basal ganglia and thalamus in Alzheimer’s disease: a three-year longitudinal study. J Alzheimers Dis 40:285–295
Coleman M (2005) Axon degeneration mechanisms: commonality amid diversity. Nat Rev Neurosci 6:889–898
Concha L (2014) A macroscopic view of microstructure: using diffusion-weighted images to infer damage, repair, and plasticity of white matter. Neuroscience 276:14–28
Cui Z, Zhong S, Xu P, He Y, Gong G (2013) PANDA: a pipeline toolbox for analyzing brain diffusion images. Front Hum Neurosci 7:42
Danielian LE, Iwata NK, Thomasson DM, Floeter MK (2010) Reliability of fiber tracking measurements in diffusion tensor imaging for longitudinal study. NeuroImage 49:1572–1580
Di Paola M, Luders E, Di Iulio F, Cherubini A, Passafiume D, Thompson PM, Caltagirone C, Toga AW, Spalletta G (2010) Callosal atrophy in mild cognitive impairment and alzheimer’s disease: different effects in different stages. NeuroImage 49:141–149
Douaud G, Jbabdi S, Behrens TE, Menke RA, Gass A, Monsch AU, Rao A, Whitcher B, Kindlmann G, Matthews PM, Smith S (2011) DTI measures in crossing-fibre areas: increased diffusion anisotropy reveals early white matter alteration in MCI and mild Alzheimer’s disease. NeuroImage 55:880–890
Fitzsimmons J, Hamoda HM, Swisher T, Terry D, Rosenberger G, Seidman LJ, Goldstein J, Mesholam-Gately R, Petryshen T, Wojcik J, Kikinis R, Kubicki M (2014) Diffusion tensor imaging study of the fornix in first episode schizophrenia and in healthy controls. Schizophr Res 156:157–160
Floresco SB, Grace AA (2003) Gating of hippocampal-evoked activity in prefrontal cortical neurons by inputs from the mediodorsal thalamus and ventral tegmental area. J Neurosci 23:3930–3943
Fritzsche KH, Stieltjes B, Schlindwein S, van Bruggen T, Essig M, Meinzer HP (2010) Automated MR morphometry to predict alzheimer’s disease in mild cognitive impairment. Int J Comput Assist Radiol Surg 5:623–632
Gaffan D, Easton A, Parker A (2002) Interaction of inferior temporal cortex with frontal cortex and basal forebrain: double dissociation in strategy implementation and associative learning. J Neurosci 22:7288–7296
Gao J, Cheung RT, Lee TM, Chu LW, Chan YS, Mak HK, Zhang JX, Qiu D, Fung G, Cheung C (2014) Possible retrogenesis observed with fiber tracking: an anteroposterior pattern of white matter disintegrity in normal aging and alzheimer’s disease. J Alzheimers Dis 26:47–58
Hong YJ, Yoon B, Lim SC, Shim YS, Kim JY, Ahn KJ, Han IW, Yang DW (2013) Microstructural changes in the hippocampus and posterior cingulate in mild cognitive impairment and Alzheimer’s disease: a diffusion tensor imaging study. Neurol Sci 34:1215–1221
Jiang H, van Zijl PC, Kim J, Pearlson GD, Mori S (2006) DtiStudio: resource program for diffusion tensor computation and fiber bundle tracking. Comput Methods Prog Biomed 81:106–116
Kamali A, Kramer LA, Hasan KM (2010) Feasibility of prefronto-caudate pathway tractography using high resolution diffusion tensor tractography data at 3T. J Neurosci Methods 191:249–254
Knight WD, Okello AA, Ryan NS, Turkheimer FE, Martinez R, de Llano S, Edison P, Douglas J, Fox NC, Brooks DJ, Rossor MN (2011) Carbon-11-Pittsburgh compound B positron emission tomography imaging of amyloid deposition in presenilin 1 mutation carriers. Brain 134:293–300
Lee DY, Fletcher E, Carmichael OT, Singh B, Mungas D, Reed B, Martinez O, Buonocore MH, Persianinova M, Decarli C (2012) Sub-regional hippocampal injury is associated with fornix degeneration in alzheimer’s disease. Front Aging Neurosci 4:1
Lee GJ, Lu PH, Medina LD, Rodriguez-Agudelo Y, Melchor S, Coppola G, Braskie MN, Hua X, Apostolova LG, Leow AD, Thompson PM, Ringman JM (2013) Regional brain volume differences in symptomatic and presymptomatic carriers of familial alzheimer’s disease mutations. J Neurol Neurosurg Psychiatry 84:154–162
Leemans A, Jones DK (2009) The B-matrix must be rotated when correcting for subject motion in DTI data. Magn Reson Med 61:1336–1349
Liu Y, Spulber G, Lehtimaki KK, Kononen M, Hallikainen I, Grohn H, Kivipelto M, Hallikainen M, Vanninen R, Soininen H (2011) Diffusion tensor imaging and tract-based spatial statistics in Alzheimer’s disease and mild cognitive impairment. Neurobiol Aging 32:1558–1571
Liu J, Yin C, Xia S, Jia L, Guo Y, Zhao Z, Li X, Han Y, Jia J (2013) White matter changes in patients with amnestic mild cognitive impairment detected by diffusion tensor imaging. PLoS One 8:e59440
Mamah D, Conturo TE, Harms MP, Akbudak E, Wang L, McMichael AR, Gado MH, Barch DM, Csernansky JG (2010) Anterior thalamic radiation integrity in schizophrenia: a diffusion-tensor imaging study. Psychiatry Res 183:144–150
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA work group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939–944
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR, Jr., Kawas CH, Klunk WE, Koroshetz WJ, Manly JJ, Mayeux R, Mohs RC, Morris JC, Rossor MN, Scheltens P, Carrillo MC, Thies B, Weintraub S, Phelps CH (2011) The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7:263–269.
Mielke MM, Okonkwo OC, Oishi K, Mori S, Tighe S, Miller MI, Ceritoglu C, Brown T, Albert M, Lyketsos CG (2012) Fornix integrity and hippocampal volume predict memory decline and progression to Alzheimer’s disease. Alzheimers Dement 8:105–113
Mori S, Oishi K, Jiang H, Jiang L, Li X, Akhter K, Hua K, Faria AV, Mahmood A, Woods R, Toga AW, Pike GB, Neto PR, Evans A, Zhang J, Huang H, Miller MI, van Zijl P, Mazziotta J (2008) Stereotaxic white matter atlas based on diffusion tensor imaging in an ICBM template. NeuroImage 40:570–582
Mormina E, Arrigo A, Calamuneri A, Granata F, Quartarone A, Ghilardi MF, Inglese M, Di Rocco A, Milardi D, Anastasi GP, Gaeta M (2014) Diffusion tensor imaging parameters' changes of cerebellar hemispheres in Parkinson’s disease. Neuroradiology 57:327–334
Morris JC (1997) Clinical dementia rating: a reliable and valid diagnostic and staging measure for dementia of the Alzheimer type. Int Psychogeriatr/IPA 9(Suppl 1):173–176 discussion 177-178
Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699
Niida A, Niida R, Kuniyoshi K, Motomura M, Uechi A (2013) Usefulness of visual evaluation of the anterior thalamic radiation by diffusion tensor tractography for differentiating between Alzheimer’s disease and elderly major depressive disorder patients. Int J Gen Med 6:189–200
Nir TM, Jahanshad N, Villalon-Reina JE, Toga AW, Jack CR, Weiner MW, Thompson PM, Alzheimer’s Disease Neuroimaging I (2013) Effectiveness of regional DTI measures in distinguishing Alzheimer’s disease, MCI, and normal aging. NeuroImage Clin 3:180–195
Nowrangi MA, Lyketsos CG, Leoutsakos JM, Oishi K, Albert M, Mori S, Mielke MM (2013) Longitudinal, region-specific course of diffusion tensor imaging measures in mild cognitive impairment and Alzheimer’s disease. Alzheimers Dement 9:519–528
O’Dwyer L, Lamberton F, Bokde AL, Ewers M, Faluyi YO, Tanner C, Mazoyer B, O’Neill D, Bartley M, Collins DR, Coughlan T, Prvulovic D, Hampel H (2011) Multiple indices of diffusion identifies white matter damage in mild cognitive impairment and Alzheimer’s disease. PLoS One 6:e21745
Oishi K, Mielke MM, Albert M, Lyketsos CG, Mori S (2012) The fornix sign: a potential sign for Alzheimer’s disease based on diffusion tensor imaging. J Neuroimaging 22:365–374
Querfurth HW, LaFerla FM (2010) Alzheimer’s disease. N Engl J Med 362:329–344
Reich DS, Smith SA, Jones CK, Zackowski KM, van Zijl PC, Calabresi PA, Mori S (2006) Quantitative characterization of the corticospinal tract at 3 T. AJNR Am J Neuroradiol 27:2168–2178
Reitz C, Brayne C, Mayeux R (2011) Epidemiology of Alzheimer disease. Nat Rev Neurol 7:137–152
Rive B, Gauthier S, Costello S, Marre C, Francois C (2013) Synthesis and comparison of the meta-analyses evaluating the efficacy of memantine in moderate to severe stages of Alzheimer’s disease. CNS Drugs 27:573–582
Rub U, Del Tredici K, Del Turco D, Braak H (2002) The intralaminar nuclei assigned to the medial pain system and other components of this system are early and progressively affected by the Alzheimer’s disease-related cytoskeletal pathology. J Chem Neuroanat 23:279–290
Ryan NS, Keihaninejad S, Shakespeare TJ, Lehmann M, Crutch SJ, Malone IB, Thornton JS, Mancini L, Hyare H, Yousry T, Ridgway GR, Zhang H, Modat M, Alexander DC, Rossor MN, Ourselin S, Fox NC (2013) Magnetic resonance imaging evidence for presymptomatic change in thalamus and caudate in familial Alzheimer’s disease. Brain 136:1399–1414
Sexton CE, McDermott L, Kalu UG, Herrmann LL, Bradley KM, Allan CL, Le Masurier M, Mackay CE, Ebmeier KP (2012) Exploring the pattern and neural correlates of neuropsychological impairment in late-life depression. Psychol Med 42:1195–1202
Smith SM (2002) Fast robust automated brain extraction. Hum Brain Mapp 17:143–155
Smith SM, Johansen-Berg H, Jenkinson M, Rueckert D, Nichols TE, Miller KL, Robson MD, Jones DK, Klein JC, Bartsch AJ, Behrens TE (2007) Acquisition and voxelwise analysis of multi-subject diffusion data with tract-based spatial statistics. Nat Protoc 2:499–503
Song SK, Yoshino J, Le TQ, Lin SJ, Sun SW, Cross AH, Armstrong RC (2005) Demyelination increases radial diffusivity in corpus callosum of mouse brain. NeuroImage 26:132–140
Syc SB, Harrison DM, Saidha S, Seigo M, Calabresi PA, Reich DS (2013) Quantitative MRI demonstrates abnormality of the fornix and cingulum in multiple sclerosis. Mult Scler Int 2013:838719
Tayeb HO, Yang HD, Price BH, Tarazi FI (2012) Pharmacotherapies for Alzheimer’s disease: beyond cholinesterase inhibitors. Pharmacol Ther 134:8–25
Teipel SJ, Bokde AL, Meindl T, Amaro E, Jr., Soldner J, Reiser MF, Herpertz SC, Moller HJ, Hampel H (2010) White matter microstructure underlying default mode network connectivity in the human brain. NeuroImage 49:2021–2032.
Teipel SJ, Grothe MJ, Filippi M, Fellgiebel A, Dyrba M, Frisoni GB, Meindl T, Bokde AL, Hampel H, Kloppel S, Hauenstein K, group Es (2014) Fractional anisotropy changes in Alzheimer’s disease depend on the underlying fiber tract architecture: a multiparametric DTI study using joint independent component analysis. J Alzheimers Dis 41:69–83
Van der Werf YD, Jolles J, Witter MP, Uylings HB (2003) Contributions of thalamic nuclei to declarative memory functioning. Cortex 39:1047–1062
Villain N, Desgranges B, Viader F, de la Sayette V, Mezenge F, Landeau B, Baron JC, Eustache F, Chetelat G (2008) Relationships between hippocampal atrophy, white matter disruption, and gray matter hypometabolism in Alzheimer’s disease. J Neurosci 28:6174–6181
Wakana S, Caprihan A, Panzenboeck MM, Fallon JH, Perry M, Gollub RL, Hua K, Zhang J, Jiang H, Dubey P, Blitz A, van Zijl P, Mori S (2007) Reproducibility of quantitative tractography methods applied to cerebral white matter. NeuroImage 36:630–644
Waldorff FB, Buss DV, Eckermann A, Rasmussen ML, Keiding N, Rishoj S, Siersma V, Sorensen J, Sorensen LV, Vogel A, Waldemar G (2012) Efficacy of psychosocial intervention in patients with mild Alzheimer’s disease: the multicentre, rater blinded, randomised Danish Alzheimer Intervention Study (DAISY). BMJ 345:e4693
Wang JY, Abdi H, Bakhadirov K, Diaz-Arrastia R, Devous MD, Sr. (2012) A comprehensive reliability assessment of quantitative diffusion tensor tractography. NeuroImage 60:1127–1138.
Wegrzyn M, Teipel SJ, Oltmann I, Bauer A, Thome J, Grossmann A, Hauenstein K, Hoppner J (2013) Structural and functional cortical disconnection in Alzheimer’s disease: a combined study using diffusion tensor imaging and transcranial magnetic stimulation. Psychiatry Res 212:192–200
Woods RP, Grafton ST, Holmes CJ, Cherry SR, Mazziotta JC (1998) Automated image registration: I. General methods and intrasubject, intramodality validation. J Comput Assist Tomogr 22:139–152
Yoon B, Shim YS, Hong YJ, Koo BB, Kim YD, Lee KO, Yang DW (2011) Comparison of diffusion tensor imaging and voxel-based morphometry to detect white matter damage in Alzheimer’s disease. J Neurol Sci 302:89–95
Yoshida S, Hayakawa K, Yamamoto A, Okano S, Kanda T, Yamori Y, Yoshida N, Hirota H (2010) Quantitative diffusion tensor tractography of the motor and sensory tract in children with cerebral palsy. Dev Med Child Neurol 52:935–940
Zarei M, Patenaude B, Damoiseaux J, Morgese C, Smith S, Matthews PM, Barkhof F, Rombouts SA, Sanz-Arigita E, Jenkinson M (2010) Combining shape and connectivity analysis: an MRI study of thalamic degeneration in Alzheimer’s disease. NeuroImage 49:1–8
Zhou SY, Suzuki M, Hagino H, Takahashi T, Kawasaki Y, Nohara S, Yamashita I, Seto H, Kurachi M (2003) Decreased volume and increased asymmetry of the anterior limb of the internal capsule in patients with schizophrenia. Biol Psychiatry 54:427–436
Acknowledgments
We thank all the participants in this study. This work was supported by the National Natural Science Foundation of China (Grant no. 81100243, 81171131, 81272564, 81272795, 81100893, 81172197, and 81372484), the special fund for Scientific Research of Doctor-degree Subjects in Colleges and Universities (20102104110009), the Natural Science Foundation of Liaoning Province in China (No. L2013296), and the Liaoning Science and Technology Plan Projects (No. 2011225020).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, QY., Bi, SW., Yao, XT. et al. Disruption of thalamic connectivity in Alzheimer’s disease: a diffusion tensor imaging study. Metab Brain Dis 30, 1295–1308 (2015). https://doi.org/10.1007/s11011-015-9708-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-015-9708-7