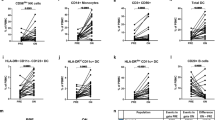
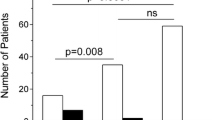
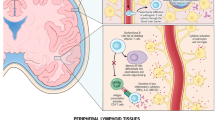
Abstract
Applying different technologies to monitor disease activity and treatment efficacy are essential in a complex disease such as multiple sclerosis. Combining current assays with flow cytometry could create a powerful tool for such analyses. The cell surface expression level of CD74, the MHC class II invariant chain, is a potential disease biomarker that could be monitored by FACS analysis in order to assess disease progression and the clinical efficacy of partial MHC class II constructs in treating MS. These constructs, which can bind to and down-regulate CD74 cell-surface expression on monocytes and inhibit macrophage migration inhibitory factor (MIF) effects, can reverse clinical and histological signs of EAE. These properties of partial class II constructs are highly compatible with a flow cytometry approach for monitoring CD74 expression as a possible biomarker for disease activity/progression and as a treatment response marker.





Similar content being viewed by others
References
Ajami B, Bennett JL, Krieger C, McNagny KM, Rossi FM (2011) Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat Neurosci 14:1142–1149. doi:10.1038/nn.2887
Ashman JB, Miller J (1999) A role for the transmembrane domain in the trimerization of the MHC class II-associated invariant chain. J Immunol 163:2704–2712
Bauer J, Huitinga I, Zhao W, Lassmann H, Hickey WF, Dijkstra CD (1995) The role of macrophages, perivascular cells, and microglial cells in the pathogenesis of experimental autoimmune encephalomyelitis. Glia 15:437–446. doi:10.1002/glia.440150407
Benedek G et al (2013) Partial MHC class II constructs inhibit MIF/CD74 binding and downstream effects. Eur J Immunol 43:1309–1321. doi:10.1002/eji.201243162
Benedek G et al (2014) A novel HLA-DRalpha1-MOG-35–55 construct treats experimental stroke. Metab Brain Dis 29:37–45. doi:10.1007/s11011-013-9440-0
Bernhagen J et al (1993) MIF is a pituitary-derived cytokine that potentiates lethal endotoxaemia. Nature 365:756–759. doi:10.1038/365756a0
Bernhagen J et al (2007) MIF is a noncognate ligand of CXC chemokine receptors in inflammatory and atherogenic cell recruitment. Nat Med 13:587–596. doi:10.1038/nm1567
Brosnan CF, Bornstein MB, Bloom BR (1981) The effects of macrophage depletion on the clinical and pathologic expression of experimental allergic encephalomyelitis. J Immunol 126:614–620
Burrows GG, Chang JW, Bachinger HP, Bourdette DN, Offner H, Vandenbark AA (1999) Design, engineering and production of functional single-chain T cell receptor ligands. Protein Eng 12:771–778
Burrows GG et al (2001) Rudimentary TCR signaling triggers default IL-10 secretion by human Th1 cells. J Immunol 167:4386–4395
Calandra T, Roger T (2003) Macrophage migration inhibitory factor: a regulator of innate immunity. Nat Rev Immunol 3:791–800. doi:10.1038/nri1200
Cepok S et al (2001) Patterns of cerebrospinal fluid pathology correlate with disease progression in multiple sclerosis. Brain 124:2169–2176
Cresswell P (1996) Invariant chain structure and MHC class II function. Cell 84:505–507
Dahan R et al (2011) TCR-like antibodies distinguish conformational and functional differences in two- versus four-domain auto reactive MHC class II-peptide complexes. Eur J Immunol 41:1465–1479. doi:10.1002/eji.201041241
Dixon AM, Stanley BJ, Matthews EE, Dawson JP, Engelman DM (2006) Invariant chain transmembrane domain trimerization: a step in MHC class II assembly. Biochemistry 45:5228–5234. doi:10.1021/bi052112e
Fan H et al (2011) Macrophage migration inhibitory factor and CD74 regulate macrophage chemotactic responses via MAPK and Rho GTPase. J Immunol 186:4915–4924. doi:10.4049/jimmunol.1003713
Freedman MS et al (2005) Recommended standard of cerebrospinal fluid analysis in the diagnosis of multiple sclerosis: a consensus statement. Arch Neurol 62:865–870. doi:10.1001/archneur.62.6.865
Gonsette RE (2003) Mitoxantrone in progressive multiple sclerosis: when and how to treat? J Neurol Sci 206:203–208
Huan J et al (2004) Monomeric recombinant TCR ligand reduces relapse rate and severity of experimental autoimmune encephalomyelitis in SJL/J mice through cytokine switch. J Immunol 172:4556–4566
Huitinga I, van Rooijen N, de Groot CJ, Uitdehaag BM, Dijkstra CD (1990) Suppression of experimental allergic encephalomyelitis in Lewis rats after elimination of macrophages. J Exp Med 172:1025–1033
Jasanoff A, Song S, Dinner AR, Wagner G, Wiley DC (1999) One of two unstructured domains of Ii becomes ordered in complexes with MHC class II molecules. Immunity 10:761–768
King G, Dixon AM (2010) Evidence for role of transmembrane helix-helix interactions in the assembly of the Class II major histocompatibility complex. Mol BioSyst 6:1650–1661. doi:10.1039/c002241a
Koch N, Zacharias M, Konig A, Temme S, Neumann J, Springer S (2011) Stoichiometry of HLA class II-invariant chain oligomers. PLoS ONE 6:e17257. doi:10.1371/journal.pone.0017257
Kowarik MC, Grummel V, Wemlinger S, Buck D, Weber MS, Berthele A, Hemmer B (2014) Immune cell subtyping in the cerebrospinal fluid of patients with neurological diseases. J Neurol 261:130–143. doi:10.1007/s00415-013-7145-2
Lamb CA, Cresswell P (1992) Assembly and transport properties of invariant chain trimers and HLA-DR-invariant chain complexes. J Immunol 148:3478–3482
Leng L et al (2003) MIF signal transduction initiated by binding to CD74. J Exp Med 197:1467–1476. doi:10.1084/jem.20030286
Lennon VA et al (2004) A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet 364:2106–2112. doi:10.1016/S0140-6736(04)17551-X
Liu L et al (2010) CXCR2-positive neutrophils are essential for cuprizone-induced demyelination: relevance to multiple sclerosis. Nat Neurosci 13:319–326. doi:10.1038/nn.2491
Meza-Romero R et al (2014) HLA-DRalpha1 constructs block CD74 expression and MIF effects in experimental autoimmune encephalomyelitis. J Immunol 192:4164–4173. doi:10.4049/jimmunol.1303118
Neumann J, Koch N (2005) Assembly of major histocompatibility complex class II subunits with invariant chain. FEBS Lett 579:6055–6059. doi:10.1016/j.febslet.2005.09.070
Polman CH et al (2010) Recommendations for clinical use of data on neutralising antibodies to interferon-beta therapy in multiple sclerosis. Lancet Neurol 9:740–750. doi:10.1016/S1474-4422(10)70103-4
Roche PA, Marks MS, Cresswell P (1991) Formation of a nine-subunit complex by HLA class II glycoproteins and the invariant chain. Nature 354:392–394. doi:10.1038/354392a0
Schwartz V et al (2009) A functional heteromeric MIF receptor formed by CD74 and CXCR4. FEBS Lett 583:2749–2757. doi:10.1016/j.febslet.2009.07.058
Shi X et al (2006) CD44 is the signaling component of the macrophage migration inhibitory factor-CD74 receptor complex. Immunity 25:595–606. doi:10.1016/j.immuni.2006.08.020
Sinha S et al (2007) A promising therapeutic approach for multiple sclerosis: recombinant T-cell receptor ligands modulate experimental autoimmune encephalomyelitis by reducing interleukin-17 production and inhibiting migration of encephalitogenic cells into the CNS. J Neurosci Off J Soc Neurosci 27:12531–12539. doi:10.1523/JNEUROSCI. 3599-07.2007
Sinha S et al (2009) Cytokine switch and bystander suppression of autoimmune responses to multiple antigens in experimental autoimmune encephalomyelitis by a single recombinant T-cell receptor ligand. J Neurosci Off J Soc Neurosci 29:3816–3823. doi:10.1523/JNEUROSCI. 5812-08.2009
Sinha S et al (2010a) Binding of recombinant T cell receptor ligands (RTL) to antigen presenting cells prevents upregulation of CD11b and inhibits T cell activation and transfer of experimental autoimmune encephalomyelitis. J Neuroimmunol 225:52–61. doi:10.1016/j.jneuroim.2010.04.013
Sinha S et al (2010b) Recombinant TCR ligand reverses clinical signs and CNS damage of EAE induced by recombinant human MOG. J Neuroimmune Pharmacol Off J Soc Neuroimmune Pharmacol 5:231–239. doi:10.1007/s11481-009-9175-1
Sospedra M, Martin R (2005) Immunology of multiple sclerosis. Annu Rev Immunol 23:683–747. doi:10.1146/annurev.immunol.23.021704.115707
Steinman L (2001) Multiple sclerosis: a two-stage disease. Nat Immunol 2:762–764. doi:10.1038/ni0901-762
Vandenbark AA et al (2003) Recombinant TCR ligand induces tolerance to myelin oligodendrocyte glycoprotein 35–55 peptide and reverses clinical and histological signs of chronic experimental autoimmune encephalomyelitis in HLA-DR2 transgenic mice. J Immunol 171:127–133
Vandenbark AA et al (2013) A novel regulatory pathway for autoimmune disease: binding of partial MHC class II constructs to monocytes reduces CD74 expression and induces both specific and bystander T-cell tolerance. J Autoimmun 40:96–110. doi:10.1016/j.jaut.2012.08.004
Wang C et al (2006) Antigen-specific therapy promotes repair of myelin and axonal damage in established EAE. J Neurochem 98:1817–1827. doi:10.1111/j.1471-4159.2006.04081.x
Yadav V et al (2012) Recombinant T-cell receptor ligand (RTL) for treatment of multiple sclerosis: a double-blind, placebo-controlled, phase 1, dose-escalation study. Autoimmune Dis 2012:954739. doi:10.1155/2012/954739
Acknowledgments
This work was supported by NIH grants NS47661 (to AAV), National Multiple Sclerosis Society grant RG3794-B-6 (to AAV), postdoctoral fellowship from the National Multiple Sclerosis Society (to GB) and the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development. The contents do not represent the views of the Department of Veterans Affairs or the United States Government.
Conflict of interest
Drs. Vandenbark, Benedek, Meza-Romero and OHSU have a significant financial interest in Artielle ImmunoTherapeutics, Inc., a company that may have a commercial interest in the results of this research and technology. This potential conflict of interest has been reviewed and managed by the OHSU and VAMC Conflict of Interest in Research Committees.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Benedek, G., Meza-Romero, R., Bourdette, D. et al. The use of flow cytometry to assess a novel drug efficacy in multiple sclerosis. Metab Brain Dis 30, 877–884 (2015). https://doi.org/10.1007/s11011-014-9634-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-014-9634-0