Abstract
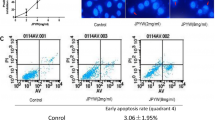
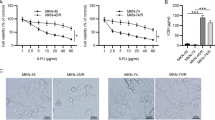
The aim of the present study was to determine the effect of AICAR, an AMPK activator, on apoptosis in gastric carcinoma cells (SGC-7901) with or without 5-fluorouracil (5-FU). SGC-7901 cells were treated with AICAR (0.2–5 mM, for 24–48 h) with or without 5-FU. Cell viability was determined using MTT assay, while apoptosis were measured through the evaluation of active caspase-3 activity and DNA fragmentation. Real-time PCR was employed to determine the expression of tumor suppressor and multi-drug resistant (mdr1) gene. Cleaved caspase-3 and phosphorylated AMPK (p-AMPK) were measured by Western blot. AICAR significant reduced cellular viability but increased apoptosis in a time- and dose-dependent manner, which is associated with an increase in p-AMPK levels. Importantly, AICAR enhanced the sensitivity to 5-FU-induced reduction of cellular viability and increased apoptosis in SGC-7901 cells. Furthermore, AICAR increased tumor suppressor genes [F-box and WD repeat domain containing 7 (FBXW7), semaphorin III/F (SEMA3F), and p21Cip1 (p21)] but reduced mdr1 expression. Finally, p-AMPK levels were reduced in 5-FU-resistant gastric cancer cells compared to human immortalized gastric epithelial cell line and 5-FU-sensitive gastric cancer cells. AICAR not only induces apoptosis alone but also enhances pro-apoptotic effect of 5-FU in SGC-7901 cells, which lays an experimental foundation to develop AICAR as a chemotherapeutic sensitizer against gastric cancer.




Similar content being viewed by others
References
Kamangar F, Dores GM, Anderson WF (2006) Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol 24:2137–2150. doi:10.1200/JCO.2005.05.2308
Cancer Genome Atlas Research Network (2014) Comprehensive molecular characterization of gastric adenocarcinoma. Nature 513:202–209. doi:10.1038/nature13480
Herrero R, Park JY, Forman D (2014) The fight against gastric cancer—the IARC Working Group report. Best Pract Res Clin Gastroenterol 28:1107–1114. doi:10.1016/j.bpg.2014.10.003
Mikulin T, Hardcastle JD (1987) Gastric cancer–delay in diagnosis and its causes. Eur J Cancer Clin Oncol 23:1683–1690
Tsai MM, Wang CS, Tsai CY, Chi HC, Tseng YH, Lin KH (2014) Potential prognostic, diagnostic and therapeutic markers for human gastric cancer. World J Gastroenterol 20:13791–13803. doi:10.3748/wjg.v20.i38.13791
Kim YH, Liang H, Liu X, Lee JS, Cho JY, Cheong JH, Kim H, Li M, Downey TJ, Dyer MD, Sun Y, Sun J, Beasley EM, Chung HC, Noh SH, Weinstein JN, Liu CG, Powis G (2012) AMPKalpha modulation in cancer progression: multilayer integrative analysis of the whole transcriptome in Asian gastric cancer. Cancer Res 72:2512–2521. doi:10.1158/0008-5472.CAN-11-3870
Jorgensen SB, Viollet B, Andreelli F, Frosig C, Birk JB, Schjerling P, Vaulont S, Richter EA, Wojtaszewski JF (2004) Knockout of the alpha2 but not alpha1 5′-AMP-activated protein kinase isoform abolishes 5-aminoimidazole-4-carboxamide-1-beta-4-ribofuranosidebut not contraction-induced glucose uptake in skeletal muscle. J Biol Chem 279:1070–1079. doi:10.1074/jbc.M306205200
Friedrichsen M, Mortensen B, Pehmoller C, Birk JB, Wojtaszewski JF (2013) Exercise-induced AMPK activity in skeletal muscle: role in glucose uptake and insulin sensitivity. Mol Cell Endocrinol 366:204–214. doi:10.1016/j.mce.2012.06.013
Viollet B, Andreelli F, Jorgensen SB, Perrin C, Geloen A, Flamez D, Mu J, Lenzner C, Baud O, Bennoun M, Gomas E, Nicolas G, Wojtaszewski JF, Kahn A, Carling D, Schuit FC, Birnbaum MJ, Richter EA, Burcelin R, Vaulont S (2003) The AMP-activated protein kinase alpha2 catalytic subunit controls whole-body insulin sensitivity. J Clin Invest 111:91–98. doi:10.1172/JCI16567
Kim JG, Lee SJ, Chae YS, Kang BW, Lee YJ, Oh SY, Kim MC, Kim KH, Kim SJ (2013) Association between phosphorylated AMP-activated protein kinase and MAPK3/1 expression and prognosis for patients with gastric cancer. Oncology 85:78–85. doi:10.1159/000351234
Luo L, Huang W, Tao R, Hu N, Xiao ZX, Luo Z (2013) ATM and LKB1 dependent activation of AMPK sensitizes cancer cells to etoposide-induced apoptosis. Cancer Lett 328:114–119. doi:10.1016/j.canlet.2012.08.034
Wang Z, Li W, Meng X, Jia B (2012) Resveratrol induces gastric cancer cell apoptosis via reactive oxygen species, but independent of sirtuin1. Clin Exp Pharmacol Physiol 39:227–232. doi:10.1111/j.1440-1681.2011.05660.x
Song W, Lu X, Feng Q (2000) Tumor necrosis factor-alpha induces apoptosis via inducible nitric oxide synthase in neonatal mouse cardiomyocytes. Cardiovasc Res 45:595–602
Zhu H, Zheng Z, Zhang J, Liu X, Liu Y, Yang W, Liu Y, Zhang T, Zhao Y, Liu Y, Su X, Gu X (2015) Anticancer effect of 2,7-dihydroxy-3-methylanthraquinone on human gastric cancer SGC-7901 cells in vitro and in vivo. Pharm Biol. doi:10.3109/13880209.2015.1033563
Yue L, Haroun S, Parent JL, de Brum-Fernandes AJ (2014) Prostaglandin D(2) induces apoptosis of human osteoclasts through ERK1/2 and Akt signaling pathways. Bone 60:112–121. doi:10.1016/j.bone.2013.12.011
Yue L, Durand M, Lebeau Jacob MC, Hogan P, McManus S, Roux S, de Brum-Fernandes AJ (2012) Prostaglandin D2 induces apoptosis of human osteoclasts by activating the CRTH2 receptor and the intrinsic apoptosis pathway. Bone 51:338–346. doi:10.1016/j.bone.2012.06.003
Wang Z, Si X, Xu A, Meng X, Gao S, Qi Y, Zhu L, Li T, Li W, Dong L (2013) Activation of STAT3 in human gastric cancer cells via interleukin (IL)-6-type cytokine signaling correlates with clinical implications. PLoS One 8:e75788. doi:10.1371/journal.pone.0075788
Calcagno DQ, Freitas VM, Leal MF, de Souza CR, Demachki S, Montenegro R, Assumpcao PP, Khayat AS, Smith Mde A, dos Santos AK, Burbano RR (2013) MYC, FBXW7 and TP53 copy number variation and expression in gastric cancer. BMC Gastroenterol 13:141. doi:10.1186/1471-230X-13-141
Onoyama I, Tsunematsu R, Matsumoto A, Kimura T, de Alboran IM, Nakayama K, Nakayama KI (2007) Conditional inactivation of Fbxw7 impairs cell-cycle exit during T cell differentiation and results in lymphomatogenesis. J Exp Med 204:2875–2888. doi:10.1084/jem.20062299
Mendes-da-Cruz DA, Brignier AC, Asnafi V, Baleydier F, Messias CV, Lepelletier Y, Bedjaoui N, Renand A, Smaniotto S, Canioni D, Milpied P, Balabanian K, Bousso P, Lepretre S, Bertrand Y, Dombret H, Ifrah N, Dardenne M, Macintyre E, Savino W, Hermine O (2014) Semaphorin 3F and neuropilin-2 control the migration of human T-cell precursors. PLoS One 9:e103405. doi:10.1371/journal.pone.0103405
Chen D, Jiao XL, Liu ZK, Zhang MS, Niu M (2013) Knockdown of PLA2G2A sensitizes gastric cancer cells to 5-FU in vitro. Eur Rev Med Pharmacol Sci 17:1703–1708
Park JB, Lee MS, Cha EY, Lee JS, Sul JY, Song IS, Kim JY (2012) Magnolol-induced apoptosis in HCT-116 colon cancer cells is associated with the AMP-activated protein kinase signaling pathway. Biol Pharm Bull 35:1614–1620
Yi B, Liu D, He M, Li Q, Liu T, Shao J (2013) Role of the ROS/AMPK signaling pathway in tetramethylpyrazine-induced apoptosis in gastric cancer cells. Oncol Lett 6:583–589. doi:10.3892/ol.2013.1403
Saitoh M, Nagai K, Nakagawa K, Yamamura T, Yamamoto S, Nishizaki T (2004) Adenosine induces apoptosis in the human gastric cancer cells via an intrinsic pathway relevant to activation of AMP-activated protein kinase. Biochem Pharmacol 67:2005–2011. doi:10.1016/j.bcp.2004.01.020
Khalil H, Peltzer N, Walicki J, Yang JY, Dubuis G, Gardiol N, Held W, Bigliardi P, Marsland B, Liaudet L, Widmann C (2012) Caspase-3 protects stressed organs against cell death. Mol Cell Biol 32:4523–4533. doi:10.1128/MCB.00774-12
Roos WP, Kaina B (2006) DNA damage-induced cell death by apoptosis. Trends Mol Med 12:440–450. doi:10.1016/j.molmed.2006.07.007
Wang JY (2001) DNA damage and apoptosis. Cell Death Differ 8:1047–1048. doi:10.1038/sj.cdd.4400938
Du J, Xu R (2012) RORalpha, a potential tumor suppressor and therapeutic target of breast cancer. Int J Mol Sci 13:15755–15766. doi:10.3390/ijms131215755
Xiong G, Wang C, Evers BM, Zhou BP, Xu R (2012) RORalpha suppresses breast tumor invasion by inducing SEMA3F expression. Cancer Res 72:1728–1739. doi:10.1158/0008-5472.CAN-11-2762
Geng M, Wang L, Chen X, Cao R, Li P (2013) The association between chemosensitivity and Pgp, GST-pi and Topo II expression in gastric cancer. Diagn Pathol 8:198. doi:10.1186/1746-1596-8-198
Hu WQ, Peng CW, Li Y (2009) The expression and significance of P-glycoprotein, lung resistance protein and multidrug resistance-associated protein in gastric cancer. J Exp Clin Cancer Res 28:144. doi:10.1186/1756-9966-28-144
Shi H, Lu D, Shu Y, Shi W, Lu S, Wang K (2008) Expression of multidrug resistance-related proteins p-glycoprotein, glutathione-s-transferases, topoisomerase-II and lung resistance protein in primary gastric cardiac adenocarcinoma. Hepatogastroenterology 55:1530–1536
Hien TT, Kim HG, Han EH, Kang KW, Jeong HG (2010) Molecular mechanism of suppression of MDR1 by puerarin from Pueraria lobata via NF-kappaB pathway and cAMP-responsive element transcriptional activity-dependent up-regulation of AMP-activated protein kinase in breast cancer MCF-7/adr cells. Mol Nutr Food Res 54:918–928. doi:10.1002/mnfr.200900146
Tran TP, Kim HG, Choi JH, Na MK, Jeong HG (2013) Reversal of P-glycoprotein-mediated multidrug resistance is induced by mollugin in MCF-7/adriamycin cells. Phytomedicine 20:622–631. doi:10.1016/j.phymed.2013.01.014
Kim HG, Hien TT, Han EH, Hwang YP, Choi JH, Kang KW, Kwon KI, Kim BH, Kim SK, Song GY, Jeong TC, Jeong HG (2011) Metformin inhibits P-glycoprotein expression via the NF-kappaB pathway and CRE transcriptional activity through AMPK activation. Br J Pharmacol 162:1096–1108. doi:10.1111/j.1476-5381.2010.01101.x
Qu C, Zhang W, Zheng G, Zhang Z, Yin J, He Z (2014) Metformin reverses multidrug resistance and epithelial-mesenchymal transition (EMT) via activating AMP-activated protein kinase (AMPK) in human breast cancer cells. Mol Cell Biochem 386:63–71. doi:10.1007/s11010-013-1845-x
Acknowledgments
This study was supported by the grants from the Natural Science Foundation of Anhui Province, China (No. 1301043054), the Educational Commission of Anhui Province (Grant No. kj2011Z210), and Affiliated Hospital of Anhui Medical University, Hefei, China.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wu, Y., Qi, Y., Liu, H. et al. AMPK activator AICAR promotes 5-FU-induced apoptosis in gastric cancer cells. Mol Cell Biochem 411, 299–305 (2016). https://doi.org/10.1007/s11010-015-2592-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-015-2592-y