Abstract
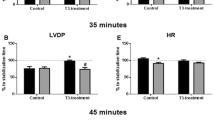
We have previously shown that acute thyroid hormone treatment could limit reperfusion injury and increase post-ischemic recovery of function. In the present study, we further explore potential initiating mechanisms of this response. Thus, isolated rat hearts were subjected to 30 min zero-flow global ischemia (I) followed by 60-min reperfusion (R). Reperfusion injury was assessed by post-ischemic recovery of left ventricular developed pressure (LVDP%) and LDH release. T3 at a dose of 60nM which had no effect on contractile function of non-ischemic myocardium, significantly increased LVDP% [48% (2.9) vs. 30.2% (3.3) for untreated group, P < 0.05] and reduced LDH release [8.3 (0.3) vs. 10 (0.42) for untreated group, P < 0.05] when administered at R. T4 (60 and 400 nM) had no effect on contractile function either in non-ischemic or ischemic myocardium. Administration of debutyl-dronedarone (DBD), a TRα1 antagonist abolished the T3-limiting effect on reperfusion injury: Thus, co-administration of T3 and DBD resulted in significantly lower LVDP%, [23% (4.7) vs. 48% (2.9) for T3 group, P < 0.05] and higher LDH release [9.9 (0.3) vs. 8.3 (0.3), for T3 group, P < 0.05]. In conclusion, acute T3 and not T4 treatment will be able to protect against reperfusion injury. T3 can exert this beneficial effect on ischemic myocardium at a dose that has no effects on non-ischemic myocardium. Acute T3-limiting effect on reperfusion injury is mediated, at least in part, via TRα1 receptor.



Similar content being viewed by others
References
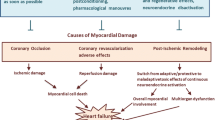
Pantos C, Mourouzis I, Cokkinos DV (2010) Rebuilding the post-infarcted myocardium by activating ‘physiologic’ hypertrophic signaling pathways: the thyroid hormone paradigm. Heart Fail Rev 15:143–154
Pantos C, Mourouzis I, Cokkinos DV (2010) New insights into the role of thyroid hormone in cardiac remodeling: time to reconsider? Heart Fail Rev 16:79–96
Axelband F, Dias J, Ferrao FM, Einicker-Lamas M (2010) Nongenomic signaling pathways triggered by thyroid hormones and their metabolite 3-iodothyronamine on the cardiovascular system. J Cell Physiol 226:21–28
Pantos C, Mourouzis I, Cokkinos DV (2010) Thyroid hormone as a therapeutic option for treating ischaemic heart disease: from early reperfusion to late remodelling. Vascul Pharmacol 52:157–165
Pantos C, Cokkinos DV (2010) Thyroid hormone: an old drug to new indications. Vascul Pharmacol 52:101
Pantos C, Mourouzis I, Xinaris C, Papadopoulou-Daifoti Z, Cokkinos D (2008) Thyroid hormone and “cardiac metamorphosis”: potential therapeutic implications. Pharmacol Ther 118:277–294
Pantos C, Malliopoulou V, Varonos DD, Cokkinos DV (2004) Thyroid hormone and phenotypes of cardioprotection. Basic Res Cardiol 99:101–120
Pantos CI, Malliopoulou VA, Mourouzis IS, Karamanoli EP, Paizis IA, Steimberg N, Varonos DD, Cokkinos DV (2002) Long-term thyroxine administration protects the heart in a pattern similar to ischemic preconditioning. Thyroid 12:325–329
Pantos C, Mourouzis I, Saranteas T, Clave G, Ligeret H, Noack-Fraissignes P, Renard PY, Massonneau M, Perimenis P, Spanou D, Kostopanagiotou G, Cokkinos DV (2009) Thyroid hormone improves postischaemic recovery of function while limiting apoptosis: a new therapeutic approach to support hemodynamics in the setting of ischaemia-reperfusion? Basic Res Cardiol 104:69–77
Ranasinghe AM, Quinn DW, Pagano D, Edwards N, Faroqui M, Graham TR, Keogh BE, Mascaro J, Riddington DW, Rooney SJ, Townend JN, Wilson IC, Bonser RS (2006) Glucose-insulin-potassium and tri-iodothyronine individually improve hemodynamic performance and are associated with reduced troponin I release after on-pump coronary artery bypass grafting. Circulation 114:I245–I250
Cao X, Kambe F, Yamauchi M, Seo H (2009) Thyroid-hormone-dependent activation of the phosphoinositide 3-kinase/Akt cascade requires Src and enhances neuronal survival. Biochem J 424:201–209
Kenessey A, Ojamaa K (2006) Thyroid hormone stimulates protein synthesis in the cardiomyocyte by activating the Akt-mTOR and p70S6 K pathways. J Biol Chem 281:20666–20672
Hiroi Y, Kim HH, Ying H, Furuya F, Huang Z, Simoncini T, Noma K, Ueki K, Nguyen NH, Scanlan TS, Moskowitz MA, Cheng SY, Liao JK (2006) Rapid nongenomic actions of thyroid hormone. Proc Natl Acad Sci USA 103:14104–14109
Saelim N, John LM, Wu J, Park JS, Bai Y, Camacho P, Lechleiter JD (2004) Nontranscriptional modulation of intracellular Ca2 + signaling by ligand stimulated thyroid hormone receptor. J Cell Biol 167:915–924
Pantos C, Mourouzis I, Malliopoulou V, Paizis I, Tzeis S, Moraitis P, Sfakianoudis K, Varonos DD, Cokkinos DV (2005) Dronedarone administration prevents body weight gain and increases tolerance of the heart to ischemic stress: a possible involvement of thyroid hormone receptor alpha1. Thyroid 15:16–23
Pantos C, Mourouzis I, Paizis I, Malliopoulou V, Xinaris C, Moraitis P, Cokkinos AD, Cokkinos DV (2007) Pharmacological inhibition of TRalpha1 receptor potentiates the thyroxine effect on body weight reduction in rats: potential therapeutic implications in controlling body weight. Diabetes Obes Metab 9:136–138
Van Beeren HC, Jong WM, Kaptein E, Visser TJ, Bakker O, Wiersinga WM (2003) Dronerarone acts as a selective inhibitor of 3, 5, 3′-triiodothyronine binding to thyroid hormone receptor-alpha1: in vitro and in vivo evidence. Endocrinology 144:552–558
Pantos C, Xinaris C, Mourouzis I, Perimenis P, Politi E, Spanou D, Cokkinos DV (2008) Thyroid hormone receptor alpha 1: a switch to cardiac cell “metamorphosis”? J Physiol Pharmacol 59:253–269
Pantos C, Mourouzis I, Delbruyere M, Malliopoulou V, Tzeis S, Cokkinos DD, Nikitas N, Carageorgiou H, Varonos D, Cokkinos D, Nisato D (2002) Effects of dronedarone and amiodarone on plasma thyroid hormones and on the basal and postischemic performance of the isolated rat heart. Eur J Pharmacol 444:191–196
Pantos C, Mourouzis I, Markakis K, Tsagoulis N, Panagiotou M, Cokkinos DV (2008) Long-term thyroid hormone administration re-shapes left ventricular chamber and improves cardiac function after myocardial infarction in rats. Basic Res Cardiol 103(4):308–318
Shulga A, Blaesse A, Kysenius K, Huttunen HJ, Tanhuanpaa K, Saarma M, Rivera C (2009) Thyroxin regulates BDNF expression to promote survival of injured neurons. Mol Cell Neurosci 42:408–418
Farach-Carson MC, Davis PJ (2003) Steroid hormone interactions with target cells: cross talk between membrane and nuclear pathways. J Pharmacol Exp Ther 307:839–845
Wehling M, Losel R (2006) Non-genomic steroid hormone effects: membrane or intracellular receptors? J Steroid Biochem Mol Biol 102:180–183
Kress E, Rezza A, Nadjar J, Samarut J, Plateroti M (2008) The thyroid hormone receptor-alpha (TRalpha) gene encoding TRalpha1 controls deoxyribonucleic acid damage-induced tissue repair. Mol Endocrinol 22:47–55
Pantos C, Mourouzis I, Galanopoulos G, Gavra M, Perimenis P, Spanou D, Cokkinos DV (2010) Thyroid hormone receptor alpha1 downregulation in postischemic heart failure progression: the potential role of tissue hypothyroidism. Horm Metab Res 42:718–724
Ocasio CA, Scanlan TS (2006) Design and characterization of a thyroid hormone receptor alpha (TRalpha)-specific agonist. ACS Chem Biol 1:585–593
Kaptein EM, Sanchez A, Beale E, Chan LS (2010) Clinical review: thyroid hormone therapy for postoperative nonthyroidal illnesses: a systematic review and synthesis. J Clin Endocrinol Metab 95:4526–4534
Pingitore A, Galli E, Barison A, Iervasi A, Scarlattini M, Nucci D, L’Abbate A, Mariotti R, Iervasi G (2008) Acute effects of triiodothyronine (T3) replacement therapy in patients with chronic heart failure and low-T3 syndrome: a randomized, placebo-controlled study. J Clin Endocrinol Metab 93:1351–1358
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pantos, C., Mourouzis, I., Saranteas, T. et al. Acute T3 treatment protects the heart against ischemia-reperfusion injury via TRα1 receptor. Mol Cell Biochem 353, 235–241 (2011). https://doi.org/10.1007/s11010-011-0791-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-0791-8