Abstract
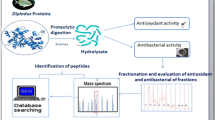
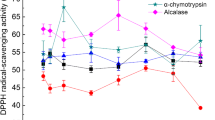
Soft tissue from cultured farm fresh oysters (Crassostrea madrasensis) was subjected to two standard enzymatic peptide extraction procedures using pepsin and papain. The crude extracts obtained were partially purified by column chromatography and were freeze-dried. The hydrolysates obtained were compared with respect to their degree of hydrolysis (DH), antioxidant potential (AP) and total phenolic content (TPC). The hydrolysate showing better antioxidant property was further subjected to purification by high performance liquid chromatography and characterized by LC-MS/MS. Papain-digested oyster protein (OPHpap) hydrolysate showed higher DH, AP and TPC. OPHpap was further subjected to ultrafiltration and fractionated into 3 sizes namely, above 10, 3–10 and 1–3 kDa according to the molecular size. Antioxidant capacity of <3 kDa fraction OPHpap-3 evaluated by DPPH free radical scavenging assay, metal chelating activity, linoleic acid autoxidation assay showed maximum effectiveness. Of the seven fractions collected by purification of OPH-pap-3 on semi-preparative RP-HPLC, fraction 7 that showed the highest antioxidant activity was further characterized by LC-ESI-MS/MS and its sequence determined. An antioxidant peptide molecule with thirteen amino acids was identified in oyster protein hydrolysate obtained by papain digestion that may find application as a nutraceutical or may be utilized in food industry for prevention of rancidity in foods.










Similar content being viewed by others
References
Anderson RS, Beaven AE (2001) Antibacterial activities of oyster (Crassostrea virginica) and mussel (Mytilus edulis and Geukensia demissa) plasma. Aquat Living Resour 14:343–349
Asha KK, Anandan R, Mathew S, Lakshmanan PT (2014) Biochemical profile of oyster Crassostrea madrasensis and its nutritional attributes. Egypt J Aquat Res 40:35–41
Bougatef A, Hajji M, Balti R, Lassoued I, Triki-Ellouz Y, Nasri M (2009) Antioxidant and free radical-scavenging activities of smooth hound (Mustelus mustelus) muscle protein hydrolysates obtained by gastrointestinal proteases. Food Chem 114:1198–1205
Bougatef A, Nedjar-Arroume N, Manni L, Ravallec R, Barkia A, Guillochon D, Nasri M (2010) Purification and identification of novel antioxidant peptides from enzymatic hydrolysates of sardinelle (Sardinella aurita) by-products proteins. Food Chem 118:559–565
Chay Pak Ting BP, Mine Y, Juneja LR, Okubo T, Gauthier SF, Pouliot Y (2001) Comparative composition and antioxidant activity of peptide fractions obtained by ultrafiltration of egg yolk protein enzymatic hydrolysates. Membranes 1:149–161
Cheng Z, Ren J, Li Y, Chang W, Chen Z (2003) Establishment of a quantitative structure-activity relationship model for evaluating and predicting the protective potentials of phenolic antioxidants on lipid peroxidation. J Pharm Sci 92:475–484
Cinq-Mars CD, Hu C, Kitts DD, Li-Chan EC (2008) Investigations into inhibitor type and mode, simulated gastrointestinal digestion, and cell transport of the angiotensin I-converting enzyme-inhibitory peptides in Pacific hake (Merluccius productus) fillet hydrolysate. J Agric Food Chem 56:410–419
Dinis TCP, Madeira VMC, Almeida LM (1994) Action of phenolic derivatives (acetaminophen, salicylate, and 5-aminosalicylate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch of Biochem Biophys 315:161–169
Dong SY, Zeng MY, Wang DF, Liu ZY, Zhao YH, Yang HC (2008) Antioxidant and biochemical properties of protein hydrolysates prepared from Silver carp (Hypophthalmichthys molitrix). Food Chem 107:1485–1493
Fruton JS, Fujii S, Knappenberger MH (1961) The mechanism of pepsin action. Proc Natl Acad Sci USA 15(47):759–761
Halliwell B, Grootveld M, Gutteridge JM (1988) Methods for the measurement of hydroxyl radicals in biomedical systems: deoxyribose degradation and aromatic hydroxylation. Methods Biochem Anal 33:59–90
Hsu KC (2010) Purification of antioxidative peptides prepared from enzymatic hydrolysates of tuna dark muscle by-product. Food Chem 122:42–48
Ichikawa K, Okabayashi T, Shima Y, Iiyama T, Takezaki Y, Munekage M, Namikawa T, Sugimoto T, Kobayashi M, Mimura T, Hanazaki K (2012) Branched-chain amino acid-enriched nutrients stimulate antioxidant DNA repair in a rat model of liver injury induced by carbon tetrachloride. Mol Biol Rep 39:10803–10810
Iris F, Benzie F, Strain JJ (1999) Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. In: Packer L (ed) Methods in enzymology. Academic Press, New York, pp 15–27
Ishida Y, Fujita T, Asai K (1981) New detection and separation method for amino acids by high-performance liquid chromatography. J Chromatogr 204:143–148
Je JY, Park PJ, Kim SK (2005) Antioxidant activity of a peptide isolated from Alaska pollack (Theragra chalcogramma) frame protein hydrolysate. Food Res Int 38:45–50
Kim SY, Je JY, Kim SK (2007) Purification and characterization of antioxidant peptide from hoki (Johnius belengerii) frame protein by gastrointestinal digestion. J Nutri Biochem 18:31–38
Klompong V, Benjakul S, Kantachote D, Hayes KD, Shahidi F (2008) Comparative study on antioxidative activity of yellow stripe trevally protein hydrolysate produced from Alcalase and Flavourzyme. Int J Food Sci Technol 43:1019–1026
Klompong V, Benjakul S, Yachai M, Visessanguan W, Shahidi F, Hayes K (2009) Amino acid composition and antioxidative peptides from protein hydrolysates of yellow stripe trevally (Selaroides leptolepis). J Food Sci 74:C126–C133
Kumar NSS, Nazeer RA, Jaiganesh R (2011) Purification and biochemical characterization of antioxidant peptide from horse mackerel (Magalaspis cordyla) viscera protein. Peptides 32:1496–1501
Levine RL, Bertlet BS, Moskovitz J, Mosoni L (1999) Methionine residues may protect proteins from critical oxidative damage. Mech Ageing Dev 107:323–332
Li Y-W, Li B (2013) Characterization of structure-antioxidant activity relationship of peptides in free radical systems using QSAR models: key sequence positions and their amino acid properties. J Theor Biol 318:29–43
Liu Z, Dong S, Xu J, Zeng M, Song H, Zhao Yuanhui (2008) Production of cysteine-rich antimicrobial peptide by digestion of oyster (Crassostrea gigas) with alcalase and bromelin. Food Control 19:231–235
Maralani MN, Movahedian A, Javanmard SH (2012) Antioxidant and cytoprotective effects of l-serine on human endothelial cells. Res Pharm Sci 7:209–215
Marques C, Licks F, Zattoni I, Borges B, de Souza LER, Marroni CA, Marroni NP (2013) Antioxidant properties of glutamine and its role in VEGF-Akt pathways in portal hypertension gastropathy. World J Gastroenterol 19:4464–4474
Mendis E, Rajapakse N, Kim SK (2004) Antioxidant properties of a radical-scavenging peptide purified from enzymatically prepared fish skin gelatin hydrolysate. J Agric Food Chem 53:581–587
Mitsuda H, Yasumoto K, Iwami I (1966) Antioxidative action of indole compounds during the autoxidation of linoleic acid. Eiyoto Shokuryo 19:210–214
Nalinanon S, Benjakul S, Kishimura H, Shahidi F (2001) Functionalities and antioxidant properties of protein hydrolysates from the muscle of ornate threadfin bream treated with pepsin from skipjack tuna. Food Chem 124:1354–1362
Nielsen PM, Petersen D, Dambmann C (2001) Improved method for determining food protein degree of hydrolysis. J Food Sci 66:642–646
Palashoff MH (2008) Determining the specificity of pepsin for proteolytic digestion. Department of Chemistry and Chemical Biology, Northeastern University, Boston, MS thesis, p. 78
Pham-Huy LA, He H, Pham-Huy C (2008) Free radicals, antioxidants in disease and health. Inter J Biomed Sci 4:89–96
Picot L, Bordenave S, Didelot S, Fruitier-Arnaudin I, Sannier F, Thorkelsson G et al (2006) Antiproliferative activity of fish protein hydrolysates on human breast cancer cell lines. Process Biochem 41:1217–1222
Qian ZJ, Jung WK, Byun HG, Kim SK (2008) Protective effect of an antioxidative peptide purified from gastrointestinal digests of oyster. Crassostrea gigas against free radical induced DNA damage. Bioresour Technol 99:3365–3371
Raghavan S, Kristinsson HG, Leeuwenburgh C (2008) Radical scavenging and reducing ability of tilapia (Oreochromis niloticus) protein hydrolysates. J Agric Food Chem 56:10359–10367
Rajapakse N, Mendis E, Byun HG, Kim SK (2005) Purification and in vitro antioxidative effects of giant squid muscle peptides on free radical-mediated oxidative systems. J Nutr Biochem 16:562–569
Ramarathnam N, Osawa T, Namiki M, Kawakishi S (1988) Chemical studies on novel rice hull antioxidants. I. Isolation, fractionation partial characterization. J Agric Food Chem 36:732–737
Sastry CSP, Tammuru MK (1985) Spectrophotometric determination of tryptophan in proteins. J Food Sci Tech 22:146–147
Sathivel S, Smiley S, Prinyawiwatkul W, Bechtel PJ (2005) Functional and nutritional properties of red salmon (Oncorhynchus nerka) enzymatic hydrolysates. J Food Sci 70:401–406
Shahidi F, Zhong Y (2008) Bioactive peptides. J AOAC Int 91:914–931
Shan L, Wang B, Gao G, Cao W, Zhang Y (2013) l-Arginine supplementation improves antioxidant defenses through l-arginine/nitric oxide pathways in exercised rats. J Appl Physiol 115:1146–1155
Sharp JS, Becker JM, Hettich RL (2003) Protein surface mapping by chemical oxidation: structural analysis by mass spectrometry. Anal Biochem 313:216–225
Singleton VL, Rossi JA Jr (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Souissi N, Bougatef A, Triki-Ellouz Y, Nasri M (2007) Biochemical and functional properties of Sardinella (Sardinella aurita) by-product hydrolysates. Food Technol Biotechnol 45:187–194
Stawikowski M, Fields GB (2002) Introduction to peptide synthesis. In: Coligan JE et al (eds) Current protocols in protein science. Wiley, New York
Tanaka H, Fukahori S, Baba S, Ueno T, Sivakumar R, Yagi M, Asagiri K, Ishii S, Tanaka Y (2014) Branched-chain amino acid-rich supplements containing microelements have antioxidant effects on nonalcoholic steatohepatitis in mice. J Parenter Enteral Nutr
Udenigwe CC, Aluko RE (2011) Chemometric analysis of the amino acid requirements of antioxidant food protein hydrolysates. Int J Molecular Sci 12:3148–3161
von Gadow A, Joubert E, Hansmann CF (1997) Comparison of the antioxidant activity of aspalathin with that of other plant phenols of rooibos tea (Aspalathus linearis), alpha-tocopherol, BHT, and BHA. J Agric Food Chem 45:632–638
Wallner S, Hermetter A, Mayer B, Wascher TC (2001) The alpha-amino group of l-arginine mediates its antioxidant effect. Eur J Clin Invest 31:98–102
Wang Y, Zhu F, Han F, Wang H (2008) Purification and characterization of antioxidative peptides from salmon protamine hydrolysate. J Food Biochem 32:654–671
Wu HC, Chen HM, Shiau CY (2003) Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackerel (Scomber austriasicus). Food Res Int 36:949–957
Zhang Z, Smith DL (1993) Determination of amide hydrogen exchange by mass spectrometry: a new tool for protein structure elucidation. Protein Sci 2:522–553
Zhu K, Zhou H, Qian H (2006) Antioxidant and free radical scavenging activities of wheat germ protein hydrolysates WGPH prepared with Alcalase. Process Biochem 41:1296–1302
Acknowledgment
Funding
This study was funded by Indian Council of Agricultural research (ICAR), New Delhi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Each of the authors, namely, Asha K. K., Remyakumari K. Raghavan, Ashok Kumar K., Niladri S. Chatterjee, R. Anandan and Seseela Mathew declare that he/she has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. This article also does not contain any studies with animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Asha, K.K., Remya Kumari, K.R., Ashok Kumar, K. et al. Sequence Determination of an Antioxidant Peptide Obtained by Enzymatic Hydrolysis of Oyster Crassostrea madrasensis (Preston). Int J Pept Res Ther 22, 421–433 (2016). https://doi.org/10.1007/s10989-016-9521-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-016-9521-0