Abstract
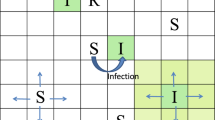
Many ecological and epidemiological studies occur in systems with mobile individuals and heterogeneous landscapes. Using a simulation model, we show that the accuracy of inferring an underlying biological process from observational data depends on movement and spatial scale of the analysis. As an example, we focused on estimating the relationship between host density and pathogen transmission. Observational data can result in highly biased inference about the underlying process when individuals move among sampling areas. Even without sampling error, the effect of host density on disease transmission is underestimated by approximately 50 % when one in ten hosts move among sampling areas per lifetime. Aggregating data across larger regions causes minimal bias when host movement is low, and results in less biased inference when movement rates are high. However, increasing data aggregation reduces the observed spatial variation, which would lead to the misperception that a spatially targeted control effort may not be very effective. In addition, averaging over the local heterogeneity will result in underestimating the importance of spatial covariates. Minimizing the bias due to movement is not just about choosing the best spatial scale for analysis, but also about reducing the error associated with using the sampling location as a proxy for an individual’s spatial history. This error associated with the exposure covariate can be reduced by choosing sampling regions with less movement, including longitudinal information of individuals’ movements, or reducing the window of exposure by using repeated sampling or younger individuals.





Similar content being viewed by others
References
Anderson RM, May RM (1991) Infectious diseases of humans: dynamics and control. Oxford University Press, Oxford
Bernardinelli L, Clayton D, Pascutto C, Montomoli C, Ghislandi M, Songini M (1995) Bayesian analysis of space–time variation in disease risk. Stat Med 14:2433–2443
Besag J, York J, Mollié A (1991) Bayesian image restoration, with two applications in spatial statistics. Ann Inst Stat Math 43(1):1–20
Bouma A, de Jong MCM, Kimman TG (1995) Transmission of pseudorabies virus within pig-populations is independent of the size of the population. Prev Vet Med 23(3–4):163–172
Cauchemez S, Valleron A, Boëlle P, Flahault A, Ferguson NM (2008) Estimating the impact of school closure on influenza transmission from sentinel data. Nature 452:750–754
Conner MM, Miller MW, Ebinger MR, Burnham KP (2007) A meta-BACI approach for evaluating management intervention on chronic wasting disease in mule deer. Ecol Appl 17(1):140–153
Cross PC, Edwards WH, Scurlock BM, Maichak EJ, Rogerson JD (2007) Effects of management and climate on elk brucellosis in the Greater Yellowstone Ecosystem. Ecol Appl 17(4):957–964
Cross PC, Heisey DM, Scurlock BM, Edwards WH, Ebinger MR, Brennan A (2010) Mapping brucellosis increases relative to elk density using hierarchical Bayesian models. PLoS One 5(4):e10322
Delahay RJ, Langton S, Smith GC, Clifton-Hadley RS, Cheeseman CI (2000) The spatio-temporal distribution of Mycobacterium bovis (bovine tuberculosis) infection in a high-density badger population. J Anim Ecol 69:428–441
Fenton A, Fairbairn JP, Norman R, Hudson PJ (2002) Parasite transmission: reconciling theory and reality. J Anim Ecol 71(5):893–905
Ferrari MJ, Perkins SE, Pomeroy LW, Bjørnstad ON (2011) Pathogens, social networks, and the paradox of transmission scaling. Interdiscip Perspect Infect Dis 267049
Fotheringham AS, Wong DWS (1991) The modifiable areal unit problem in multivariate statistical analysis. Environ Plan A 23:1025–1044
Gehlke CE, Biehl K (1934) Certain effects of grouping upon the size of the correlation coefficient in census tract material. J Am Stat Assoc 24:169–170
Gelman A, Hill J (2007) Data analysis using regression and multilevel/hierarchical models. Cambridge University Press, Cambridge
Getz WM, Pickering J (1983) Epidemic models: thresholds and population regulation. Am Nat 121:892–898
Glass K, Barnes B (2007) How much would closing schools reduce transmission during an influenza pandemic? Epidemiology 18(5):623–628
Greig-Smith P (1952) Use of random and contiguous quadrats in the study of the structure of plant communities. Ann Bot 16:293–316
Grenfell BT, Anderson RM (1985) The estimation of age-related rates of infection from case notifications and serological data. J Hyg 95(2):419–436
Halloran ME, Ferguson NM, Eubank S, Longini IM, Cummings DA, Lewis B, Xu S, Fraser C, Vullikanti A, Germann TC, Wagener D, Beckman R, Kadau K, Barrett C, Macken CA, Burke DS, Cooley P (2008) Modeling targeted layered containment of an influenza pandemic in the United States. Proc Natl Acad Sci USA 105(12):4639–4644
Hastings A (1993) Complex interactions between dispersal and dynamics: lessons from coupled logistic equations. Ecology 74(5):1362–1372
He D, Ionides EL, King AA (2010) Plug-and-play inference for disease dynamics: measles in large and small populations as a case study. J R Soc Interface 7:271–283
Heads PA, Lawton JH (1983) Studies on the natural enemy complex of the holly leaf-miner: the effects of scale on the detection of aggregative responses and the implications for biological control. Oikos 40(2):267–276
Heisey DM, Joly DO, Messier F (2006) The fitting of general force-of-infection models to wildlife disease prevalence data. Ecology 87(9):2356–2365
Heisey DM, Osnas EE, Cross PC, Joly DO, Langenberg JA, Miller MW (2010) Linking process to pattern: estimating spatiotemporal dynamics of a wildlife epidemic from cross-sectional data. Ecol Monogr 80(2):221–240
Ionides EL, Bretó C, King AA (2006) Inference for nonlinear dynamical systems. Proc Natl Acad Sci USA 103(49):18438–18443
Jelinski DE, Wu J (1996) The modifiable areal unit problem and implications for landscape ecology. Landscape Ecol 11:129–140
Kermack WO, McKendrick AG (1927) Contributions to the mathematical theory of epidemics. Proc R Soc Edinburgh 115:700–721
Knell RJ, Begon M, Thompson DJ (1998) Transmission of Plodia interpunctella granulosis virus does not conform to the mass action model. J Anim Ecol 67(4):592–599
Lloyd-Smith JO (2010) Modeling density dependence in heterogeneous landscapes: dispersal as a case study. J Theor Biol 265:160–166
Lloyd-Smith JO, Schreiber SJ, Kopp PE, Getz WM (2005) Superspreading and the effect of individual variation on disease emergence. Nature 438(7066):355–359
McCallum H, Barlow N, Hone J (2001) How should pathogen transmission be modelled? Trends Ecol Evol 16(6):295–300
Miller RE, Kaneene JB, Fitzgerald SD, Schmitt SM (2003) Evaluation of the influence of supplemental feeding of white-tailed deer (Odocoileus virginianus) on the prevalence of bovine tuberculosis in the Michigan wild deer population. J Wildl Dis 39(1):84–95
Openshaw S (1984) The modifiable areal unit problem. Concepts and techniques in modern geography 38. GeoBooks, Norwich
Pulliam HR (1988) Sources, sinks, and population regulation. Am Nat 132(5):652–661
R Development Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rachowicz LJ, Briggs CJ (2007) Quantifying the disease transmission function: effects of density on Batrachochytrium dendrobatidis transmission in the mountain yellow-legged frog Rana muscosa. J Anim Ecol 76:711–721
Ray C, Hastings A (1996) Density dependence: are we searching at the wrong spatial scale? J Anim Ecol 65(5):556–566
Rogers LM, Delahay RJ, Cheeseman CL, Smith GC, Clifton-Hadley RS (1999) The increase in badger (Meles meles) density at Woodchester Park, south-west England: a review of the implications for disease (Mycobacterium bovis) prevalence. Mammalia 63(2):183–192
Rudolph BA, Riley SJ, Hickling GJ, Frawley BJ, Garner MS, Winterstein SR (2006) Regulating hunter baiting for white-tailed deer in Michigan: biological and social considerations. Wildl Soc Bull 34(2):314–321
Schafer JL (1999) Multiple imputation: a primer. Stat Methods Med Res 8:3–15
Smith MJ, Telfer S, Kallio ER, Burthe S, Cook AR, Lambin X, Begon M (2009) Host-pathogen time series data in wildlife support a transmission function between density and frequency dependence. Proc Natl Acad Sci U S A 106(19):7905–7909
Soetaert K, Petzoldt T, Setzer RW (2010) Solving differential equations in R: package deSolve. J Stat Softw 33(9):1–25
Svancara LK, Garton EO, Chang KT, Scott JM, Zager P, Gratson M (2002) The inherent aggravation of aggregation: an example with elk aerial survey data. J Wildl Manage 66(3):776–787
Toni T, Welch D, Strelkowa N, Ipsen A, Stumpf MP (2009) Approximate Bayesian computation scheme for parameter inference and model selection in dynamical systems. J R Soc Interface 6(31):187–202
Turner J, Begon M, Bowers RG (2003) Modelling pathogen transmission: the interrelationship between local and global approaches. Proc R Soc Lond B 270(1510):105–112
Van Horn B (1983) Density as a misleading indicator of habitat quality. J Wildl Manage 47(4):893–901
Vazquez-Prokopec GM, Stoddard ST, Paz-Soldan V, Morrison AC, Elder JP, Kochel TJ, Scott TW, Kitron U (2009) Usefulness of commercially available GPS data-loggers for tracking human movement and exposure to dengue virus. Int J Health Geogr 8:68
Veldtman R, McGeoch MA (2004) Spatially explicit analyses unveil density dependence. Proc R Soc Lond B 271:2439–2444
Waller LA, Gotway CA (2004) Applied spatial statistics for public health data. Wiley, Hoboken
Woodroffe R, Donnelly CA, Wei G, Cox DR, Bourne FJ, Burke T, Butlin RK, Cheeseman CL, Gettinby G, Gilks P, Hedges S, Jenkins HE, Johnston WT, McInerney JP, Morrison WI, Pope LC (2009) Social group size affects Mycobacterium bovis infection in European badgers (Meles meles). J Anim Ecol 78(4):818–827
Acknowledgments
We thank M. Ebinger for help with the figures. PCC’s work was supported by U.S. Geological Survey, the NSF/NIH Ecology of Infectious Disease program DEB-1067129 and some ideas stem from working groups sponsored by the NIH/DHS funded RAPIDD program. DC’s work was supported by NSF Grant DEB-0749097 to L.A. Meyers. Any use of trade, product, or firm names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cross, P.C., Caillaud, D. & Heisey, D.M. Underestimating the effects of spatial heterogeneity due to individual movement and spatial scale: infectious disease as an example. Landscape Ecol 28, 247–257 (2013). https://doi.org/10.1007/s10980-012-9830-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-012-9830-4