Abstract
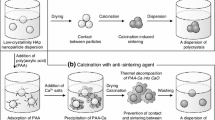
Pechini method is a materials synthesis method based on the preparation of a polymeric matrix. The advantage of this method is the ability to obtain materials with different particle sizes depending on the synthesis condition with a homogeneous distribution. In this work, carbonated hydroxyapatite (c-OHAp) nanoparticles were obtained by a modified Pechini method. To obtain the polymeric precursor of the c-OHAp, the polymeric matrix was prepared through a polyesterification reaction between citric acid and ethylene-glycol. Adding calcium hydroxide and ortophosphoric acid in aqueous solutions, raising the temperature up to 140 °C/2 h and keeping constant the pH at 8. The polymeric matrix was calcinated at different ranges of temperature from 200 to 600 °C in order to obtain the c-OHAp powder. The results show the presence of c-OHAp a as unique phase. The thermal analysis indicates that the c-OHAp phase was obtained at 600 °C. The particle size of the obtained material was <50 nm.









Similar content being viewed by others
References
Amin Alavia M, Morsalia A (2010) Ultrasonic-assisted synthesis of Ca(OH)2 and CaO nanostructures. J Exp Nanosci 5(2):93–105
Rama Krishna DS, Siddharthan A, Seshadri SK, Sampath Kumar TS (2007) A novel route for synthesis of nanocrystalline hydroxyapatite from eggshell waste. J Mater Sci Mater Med 18:1735–1743
Silva CC, Pinheiro AG, Miranda MAR, Góes JC, Sombra ASB (2003) Structural, properties of hydroxyapatite obtained by mechanosynthesis. Solid State Sci 5:553–558
Li C, Meng F (2008) Nano-crystallinite hydroxyapatite synthesized by neutralization with the assist of citric acid. Mater Lett 62:932–934
Gross KA, Rodríguez-Lorenzo LM (2004) Biodegradable composites scaffolds with an interconnected spherical pore network for bone tissue engineering. Biomaterials 25(20):4955–4962
Hutmacher DW, Schantz JT, Lam CXF, Tan KC, Lim TC (2007) State of the art and future directions of scaffold-based bone engineering from a biomaterials perspective. J.Tiss. Eng Regen Med
Mahabole MP et al (2005) Synthesis, characterization and gas sensing property of hydroxyapatite ceramic. Bull Mater Sci 28(6):535–545
Martins MA, Santos C, Margarida Almeida M, Costa MEV (2008) Hydroxyapatite micro and nanoparticles: nucleation and growth mechanisms in the presence of citrate species. J Colloid Interface Sci 318:210–216
Fernández-Montes Moraleda B, San Román J, Rodríguez-Lorenzo LM (2013) Influence of surface features of hydroxyapatite on the adsorption of proteins relevant to bone regeneration. J Biomed Mater Res 8:2332–2339. doi:10.10002/jbm.a.34528101
De Campos M, Muller FA, Bressiani AHA, Bressiani JC, Greil P (2007) Sonochemical synthesis of calcium phosphate powders. J Mater Sci Mater Med 18:669–675
Sadat-Shojai M (2009) Preparation of hydroxyapatite nanoparticles: comparison between hydrothermal and solvo-treatment processes and colloidal stability of produced nanoparticles in a dilute experimental adhesive. J Iran Chem Soc 6(2):386–392
Santos MH et al (2004) Synthesis control and characterization of hydroxyapatite prepared by wet precipitation process. Mater Res 7(4):625–630
Jaworski R, Pierlot Ch, Pawlowski L, Bigan M, Martel M (2009) Design of the synthesis of fine HA powder for suspension plasma spraying. Surf Coat Technol 203:2092–2097
Cengiz B, Gokce Y, Yildiz N, Aktas Z, Calimli A (2008) Synthesis and characterization of hydroxyapatite nanoparticles. Colloids Surf A: PhysicoChem Eng Asp 322:29–33
Bigi A, Boanini E, Rubini K (2004) Hydroxyapatite gels and nanocrystals prepared through a sol–gel process. J Solid State Chem 177:3092–3098
Bezzi G, Celotti G, Landib E, La Torretta TMG, Sopyan I, Tampieri A (2003) A novel sol–gel technique for hydroxyapatite preparation. Mater Chem Phys 78:816–824
Chu CL, Lin PH, Dong YS, Guo DY (2002) Influences of citric acid as a chelating reagent on the characteristics of nanophase hydroxyapatite powders fabricated by a sol–gel method. J Mater Sci Lett 21:1793–1795
Pechini M P (1967) Method of preparing lead and alkaline earth titanates and niobates and coating method using the same to form a capacitor, 3,330,697 Patented July 11
Segal D (1997) Chemical synthesis of ceramic materials. J Mater Chem 7(8):1297–1305
Peña J, Vallet-Regí M (2003) Hydroxyapatite, tricalcium phosphate and biphasic materials prepared by a liquid mix technique. J Eur Ceram Soc 23:1687–1696
Galceran M, Pujol MC, Aguiló M, Díaz F (2007) Sol–gel modified Pechini method for obtaining nanocrystalline KRE(WO4)2 (RE = Gd and Yb). J Sol–Gel Techn 42:78–88
Zhang Y, Li A, Yan Z, Xu G, Liao C, Yan C (2003) (ZrO2)0.85(REO1.5)0.15 (RE = Sc, Y) solid solutions prepared via THREE Pechini-type gel routes: 1-gel formation and calcinations behaviors. J Sci Sci Comm 17:1434–1438
Mosquera A, Varela JA, Rodríguez-Páez JE (2007) Varistores de SnO2 obtenidos por el método de precursor polimérico (Pechini). Rev Acad Colomb Cienc 31(118):89–96
Cho SG, Johnson PF, Condrate RA (1990) Thermal decomposition of (Sr, Ti) organic precursors during the Pechini process. J Mater Sci 25:4738–4744
Tsay J, Fang T (1999) Effects of molar ratio of citric acid to cations and of pH value on the formation and thermal-decomposition behavior of barium titanium citrate. J Am Ceram Soc 82(6):1409–1415
Razpotnik T, Franceti V, Maek J (2006) Preparation of NiO/YSZ powders using a Pechini-type method. MTAEC 40(2):69–72
Lee H, Hong M, Bae S, Lee H, Park E, Kim K (2003) A novel approach to preparing nano-size Co3O4-coated Ni powder by the Pechini method for MCFC cathodes. J Mater Chem 13:2626–2632
Costa ACFM, Vilar MA, Lira HL, Kiminami RHGA, Gama L (2006) Synthesis and characterization of TiO2 nanoparticles. Cerâmica 52:255–259
Farhikhteh S, Maghsoudipour A, Raissi B, Mozaffari B (2009) Synthesis of high specific surface area YSZ (ZrO2–8Y2O3) nanocrystalline powder by modified polymerized complex method. J Sol–Gel Sci Tech 49:60–65
Alessandra F, de Jesus A, Santos da Silva R, Soares Z (2010) Synthesis of Bi4Ge3O12 ceramic scintillators by the polymeric precursos method. J Therm Anal Calorim 100:537–541
Voicu G, Ghitulica CD, Andronescu E (2012) Modified Pechini synthesis of tricalcium aluminate powder. Mater Charact 73:89–95
Xu Z, Li Y, Liu Z, Xiong Z (2001) Low-temperature synthesis of nanocrystalline ZnGa2O4:Tb3+ phosphors via the Pechini method. Mater sci eng 110:302–306
Gutiérrez D, Tartaj J, Durán P, Moure C (2001) Síntesis, sinterización y propiedades eléctricas de soluciones sólidas YNixMn1−xO3 preparadas a partir de Precursores Poliméricos. Bol Soc Esp Cer Vid 40(5):339–343
NIST Chemistry Webook (http://webook.nist.gov/chemistry)
Laberty-Robert Ch, Ansart F, Deloget C, Gaudon M, Rousset A (2001) Powder synthesis of nanocristalline ZrO2-8%y2o3 via a polymerization route. Mater Res Bul 36:2083–2101
Mosquera A, Rodríguez-Páez JE (2008) Obtención de nano-estructuras bi-dimensionales de SnO2 utilizando el método pechini: estudio de la conformación de la resina. Bol Soc Esp Ceram 47(5):278–286
Solís Moré Y, García Carrodeguas R, Davidenko N, Peniche Covas C, Camerón R (2008) Preparación y caracterización de composites de hidroxiapatita deficiente en calcio y quitosana. CENIC Ciencias Químicas 39:17–22
Xiao X-F, Liu R-F, Gao Y-J (2008) Hydrothermal preparation of nanocarbonated hydroxyapatite crystallites. Mater Sci Technol 24(10):1199–1203
Liu WB, Qu SX, Shen R, Jian CX, Li XH, Feng B, Weng J (2006) Influence of pH values on preparation of hydroxyapatite/gelatin composites. J Mater Sci 41:1851–1853
Vargas-Becerril N, Patiño-Carachure C, Tellez-Jurado L, Rodriguez-Lorenzo LM (2013) Synthesis of hybrid compounds apatite–alendronate by reactive milling and effects on the structure and morphology of the apatite phase. Ceram Int 39:3921–3929
Enrique Pérez Alberaez, WALCO S.A. Vol.1.0;27-VII-1997
Fathi MH, Hanifi A (2007) Evaluation and characterization of nanostructure hydroxyapatite powder prepared by simple sol–gel method. Mater Lett 61:3978–3983
Ferraz MP, Monteiro FJ, Manuel CM (2004) Hydroxyapatite nanoparticles: a review of preparation methodologies. J Appl Biomater Biomech 2:74–80
Siva Rama Krishna D, Siddharthan A, Seshadri SK, Sampath Kumar TS (2007) A novel route for synthesis of nanocrystalline hydroxyapatite from eggshell waste. J Mater Sci Mater Med 18:1735–1743
Fernández-Montes Moraleda B, San Román J, Rodríguez-Lorenzo LM (2013) Influence of surface features of hydroxyapatite on the adsorption of proteins relevant to bone regeneration. J Biomed Mater Res 101A:2332–2339
Bracci B, Torricelli P, Panzavolta S, Boanini E, Giardino R, Bigi A (2009) Effect of Mg2+, Sr2+ and Mn2+ on the chemico-physical and in vitro biological properties of calcium phosphate biomimetic coatings. J Inorg Biochem 1013:1666–1674
Acknowledgments
The authors thanks to UNAM-DGAPA for postdoctoral scholarship supporting to NVB during the course of this study. We appreciate the technical support from S. Tehuacanero Núñez, P. Mexia Hernández This study was partially conducted with the financial support granted by DGAPA-UNAM through projects IN106710 and IN213912, by IPN through SIP project 20130452 and by CICYT, Spain through project MAT2010-18155.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vargas-Becerril, N., Téllez-Jurado, L., Reyes-Gasga, J. et al. Synthesis of nanosized carbonated apatite by a modified Pechini method: hydroxyapatite nucleation from a polymeric matrix. J Sol-Gel Sci Technol 72, 571–580 (2014). https://doi.org/10.1007/s10971-014-3478-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-014-3478-2