Abstract
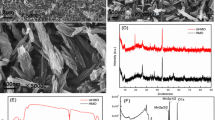
Adsorption is one of important factors associated with ore-forming process of uranium. In present paper, pyrite crystals were prepared with a hydrothermal method. Uranium adsorption was investigated with a batch method. At pH 6, the maximum sorption capacity is found to be 42.57 mg g−1. The sorption kinetics has been successfully modeled by pseudo-second-order kinetic model. Langmuir adsorption fits well with the experimental data. Results show that U(VI) is chemically concentrated onto pyrite to form uranium mineral, and sorbed U(VI) can be partially reduced to U(V). The study provides basic data for revealing the ore-forming mechanism of uranium in the presence pyrite.











Similar content being viewed by others
References
Ponomarev SNN, Kuznetsov VV, Gagarinskii AY, Moniz EJ, Gottemoeller R, Poneman D (2002) The future of nuclear power: energy, ecology, and safety. At Energy 93:855–865
Gajda D, Kiegiel K, Zakrzewska KG, Chajduk E, Bartosiewicz I, Wolkowicz S (2015) Mineralogy and uranium leaching of ores from Triassic Peribaltic sandstones. J Radioanal Nucl Chem 303:521–529
American Association of Petroleum Geologists, Energy Minerals Division (2014) Unconventional energy resources: 2013 review. Nat Resour Res 23:19–98
Specter H (2009) A sustainable U.S. energy plan. Nat Resour Res 18:285–333
Li ZY, Fang XH, Xia YL, Xiao XJ, Sun Y, Chen AP, Jiao YQ, K Zhang (2005) Metallogenetic conditions and exploration criteria of the Dongsheng sandstone type uranium deposit in Inner Mongolia, China. Mineral Deposit Research: Meeting the Global Challenge. Springer, Berlin, pp 291–294
Li ZY, Fang XH, Chen AP, Ou GX, Xiao XJ, Sun Y, Liu CY, Wang Y (2007) Origin of gray-green sandstone in ore bed of sandstone type uranium deposit in north Ordos Basin. Sci China Ser D 50:165–173
Hu H, Wang RC, Chen WF, Chen PR, Ling HF, Liu GN (2013) Timing of hydrothermal activity associated with the Douzhashan uranium-bearing granite and its significance for uranium mineralization in northeastern Guangxi, China. Chin Sci Bull 58:4319–4328
Li JW, Zhou MF, Li XF, Fu ZR, Li ZJ (2002) Structural control on uranium mineralization in south China: implications for fluid flow in continental strike slip faults. Sci China Ser D 45:851–864
Dam DV, Heil GW, Bobbink R, Heijne B (1990) Atmospheric deposition to grassland canopies: lysimeter budgets discriminating between interception deposition, mineral weathering and mineralization. Water Air Soil Pollut 53:83–101
Song SM, Hu K, Wen HJ, Zhang YX, Li K, Fan HF (2011) Molybdenum isotopic composition as a tracer for low-medium temperature hydrothermal ore-forming systems: a case study on the Dajiangping pyrite deposit, western Guangdong Province, China. Chin Sci Bull 56:2221–2228
Mersie W, Seybold C, Tsegaye T (1999) Movement, adsorption and mineralization of atrazine in two soils with and without switchgrass (Panicum virgatum) roots. Eur J Soil Sci 50:343–349
Vaughan DJ, Lennie AR (1991) The iron sulfide minerals—their chemistry and role in nature. Sci Prog 75:371–388
Zhao KD, Jiang SY, Chen WF, Chen PR, Ling HF (2014) Mineralogy, geochemistry and ore genesis of the Dawan uranium deposit in southern Hunan Province, South China. J Geochem Explor 38:59–71
Bruggeman C, Maes N (2010) Uptake of Uranium(VI) by pyrite under boom clay conditions: influence of dissolved organic carbon. Environ Sci Technol 44:4210–4216
Noubactep C, Sonnefeld J, Merten D, Heinrichs T, Sauter M (2006) Effects of the presence of pyrite and carbonate minerals on the kinetics of the uranium release from a natural rock. J Radioanal Nucl Chem 270:325–333
Descostes M, Schlegel M, Eglizaud N, Descamps F, Miserque F, Simoni E (2010) Uptake of uranium and trace elements in pyrite (FeS2) suspensions. Geochim Cosmochim Acta 74:1551–1562
Jiang L, Ma LL, Zheng YF (2009) Hydrothemal synthesis of pyrite FeS2 powder. J Shanxi Datong Univ (Nat Sci) 25:33–36 (in Chinese)
Liu ST, Wu J, Yu P, Ding QH, Zhou ZH, Li HD, Lai CC, Chueh YL, Wang ZM (2014) Phase-pure iron pyrite nanocrystals for low-cost photodetectors. Nanoscal Res Lett 9:549
Zhang YL, Zhang K, Dai CM, Zhou XF (2014) Performance and mechanism of pyrite for nitrobenzene removal in aqueous solution. Chem Eng Sci 111:135–141
Bi Y, Yuan YB, Exstrom CL, Darveau SA, Huang JS (2011) Air stable, photosensitive, phase pure iron pyrite nanocrystal thin films for photovoltaic application. Nano Lett 11:4953–4957
Li Y, Han ZL, Jiang LX, Su ZH, Liu FY, Lai YQ, Liu YX (2014) Template-directed synthesis of ordered iron pyrite (FeS2) nanowires and nanotubes arrays. J Sol–Gel Sci Technol 72:100–105
Bonato M, Ragnarsdottir KV, Allen GC (2012) Removal of uranium(VI), lead(II) at the surface of TiO2 nanotubes studied by X-ray photoelectron spectroscopy. Water Air Soil Pollut 223:3845–3857
Borah D, Senapati K (2006) Adsorption of Cd(II) from aqueous solution onto pyrite. Fuel 85:1929–1934
Kim EJ, Batchelor B (2009) Macroscopic and X-ray photoelectron spectroscopic investigation of interactions of arsenic with synthesized pyrite. Environ Sci Technol 43:2899–2904
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. J Am Chem Soc 38:2221–2295
Freundlich HMF (1906) Uber die adsorption in losungen. J Phys Chem 57:385–470
Oladoja NA, Aboluwoye CO, Oladimeji YB (2008) Kinetics and isotherm studies on methylene blue adsorption onto ground palm kernel coat. Turk J Eng Environ Sci 32:303–312
Lin SH, Juang RS (2002) Heavy metal removal from water by sorption using surfactant-modified montmorillonite. J Hazard Mater B92:315–326
Ding P, Huang KL, Li GY, Zeng WW (2007) Mechanisms and kinetics of chelating reaction between novel chitosan derivatives and Zn(II). J Hazard Mater 146:58–64
Bhattacharyya KG, Gupta SS (2008) Kaolinite and montmorillonite as adsorbents for Fe(III), Co(II) and Ni(II) in aqueous medium. Appl Clay Sci 41:1–9
Crini G, Peindy HN, Gimbert F, Robert C (2007) Removal of CI Basic Green 4 (Malachite Green) from aqueous solutions by adsorption using cyclodextrin—based adsorbent: kinetic and equilibrium studies. Sep Purif Technol 53:97–110
Rani R, Sasidhar P (2012) Geochemical and thermodynamic aspects of sorption of strontium on kaolinite dominated clay samples at Kalpakkam. Environ Earth Sci 65:1265–1274
Alkan M, Demirbas O, Dogan M (2007) Adsorption kinetics and thermodynamics of an anionic dye onto sepiolite. Microporous Mesoporous Mater 101:388–396
Zhang HX, Niu ZW, Liu Z, Wen ZD, Li WP, Wang XY, Wu WG (2015) Equilibrium, kinetic and thermodynamic studies of adsorption of Th(IV) from aqueous solution onto kaolin. J Radioanal Nucl Chem 303:87–97
Scott TB, Riba Tort O, Allen GC (2007) Aqueous uptake of uranium onto pyrite surfaces; reactivity of fresh versus weathered material. Geochim Cosmochim Acta 71:5044–5053
Yuan K, Renock D, Ewing RC, Becker U (2015) Uranium reduction on magnetite: probing for pentavalent uranium using electrochemical methods. Geochim Cosmochim Acta 156:194–206
Acknowledgments
The authors are grateful for the financial support from the National Natural Science Foundation of China (No: 21361001)and Jiangxi Educational Committee Foundation (GJJ13471). Radiation Facilities support was provided by the foundation of state key laboratory breeding base of nuclear resources and environment (Z201402) and Fundamental Science on Radioactive Geology and Exploration Technology Laboratory of East China Institute of Technology (2011RGET012). The authors would also like to thank Profs. Fengjun Nie and Jianhua Wu for their skilled technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luo, M., Liu, S., Li, J. et al. Uranium sorption characteristics onto synthesized pyrite. J Radioanal Nucl Chem 307, 305–312 (2016). https://doi.org/10.1007/s10967-015-4269-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4269-0