Abstract
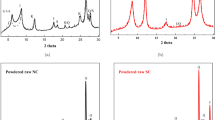
Kinetics of Pu(V) sorption on clays was studied under conditions similar to those expected at the waste repository. Batch sorption, sequential extraction (SE), oxidation state analysis and modeling were applied to study sorption mechanisms of Pu. The composition of clay coatings and their transformation were investigated using the Mössbauer spectroscopy and X-ray diffraction. In spite of the Pu(V) fast reduction its bonding to the clay was found to be different as compared to the experiments with Pu(IV). The largest portion of Pu was associated with exchangeable and carbonate fractions indicating its possible higher mobility in the environment.






Similar content being viewed by others
References
Choppin GR (2007) Actinide speciation in the environment. J Radioanal Nucl Chem 273:695–703
Choppin GR (2006) Actinide speciation in aquatic systems. Mar Chem 99:83–92
Powell BA, Fjeld RA, Kaplan DI, Coates JT, Serkiz SM (2005) Pu(V)O2 + adsorption and reduction on synthetic Goethite (α-FeOOH) and Hematite (α-Fe2O3). Env Sci Technol 39:2107–2114
Keeney-Kennicutt WL, Morse JW (1985) The redox chemistry of Pu(V)O2 + intercation with common mineral surfaces in dilute solutions and seawater. Geochim Cosmochim Acta 49:2577–2588
Sanchez AL, Murray JW, Sibley TH (1985) The adsorption of plutonium IV and V on goethite. Geochim Cosmochim Acta 49:2297–2307
Powell BA, Fjeld RA, Kaplan DI, Coates JT, Serkiz SM (2004) Pu(V)O2 + adsorption and reduction by synthetic magnetite (Fe3O4). Environ Sci Technol 38:6016–6024
Zavarin M, Powell BA, Bourbin M, Zhao PH, Kersting AB (2012) Np(V) and Pu(V) ion exchange and surface-mediated reduction mechanisms on montmorillonite. Environ Sci Technol 46:2692–2698
Lee W, Batchelor B (2003) reductive capacity of natural reductants. Environ Sci Technol 37:535–541
EPA Report (1999) 402-R-99-004A, Vol. I “Kd Model, Measurement Methods, and Applications of Chemical Reaction Codes,” Environmental Protection Agency (USA)
Lujanienė G, Beneš P, Štamberg K, Jokšas K, Kulakauskaitė I (2013) Pu and Am sorption to the baltic sea bottom sediments. J Radioanal Nucl Chem 295:1957–1967
Lujanienė G, Beneš P, Štamberg K, Ščiglo T (2012) Kinetics Of plutonium and americium sorption to natural clay. J Environ Radioact 108:41–49
Lujanienė G, Beneš P, Štamberg K, Šapolaitė J, Vopalka D, Radžiūtė E, Ščiglo T (2010) Effect of natural clay components on sorption of Cs, Pu and Am by the clay. J Radioanal Nucl Chem 286:353–359
Lujanienė G, Motiejūnas S, Šapolaitė J (2007) Sorption of Cs, Pu, Am on clay minerals. J Radioanal Nucl Chem 274:345–353
Outola I, Inn K, Ford R, Markham S, Outola P (2009) Optimizing standard sequential extraction protocol with lake and ocean sediments. J Radioanal Nucl Chem 282:321–327
Lujanienė G, Šapolaitė J, Amulevičius AS, Mažeika K, Motiejūnas S (2006) Retention of cesium, plutonium and americium by engineered and natural barriers. Czech J Phys 56:D103–D110
Lujanienė G, Šapolaitė J, Radžiūtė E, Aninkevičius V (2009) Plutonium oxidation state distribution in natural clay and goethite. J Radioanal Nucl Chem 282:793–797
Bertrand PA, Choppin GR (1982) Separation of actinides in different oxidation states by solvent extraction. Radiochim Acta 31:135–137
Andre C, Choppin GR (2000) Reduction of Pu(V) by humic acid. Radiochim Acta 88:613–616
Xia Y, Rao L, Rai D, Felmy AR (2001) Determining the distribution of Pu, Np and U oxidation states in diluted NaCl and synthetic brine solutions. J Radioanal Nucl Chem 250:27–37
Lujanienė G (2011) Determination of Pu, Am and Cm in environmental samples. In Proceeding International symposium on isotopes in hydrology, marine ecosystems, and climate change studies: Monaco, March 27-April 1, 2011, IAEA, Vienna, p. 411–418
Beneš P, Štamberg K, Štegmann R (1994) Study of the kinetics of interaction of Cs137 and Sr85 with soils using a batch method: methodological problems. Radiochim Acta 66(67):315–321
Herbelin AL and Westall JC (1996) FITEQL–A computer Programm for determination of chemical equilibrium constants from experimental data, Version 3.2, Report 96-01. Cornvallis, Oregon, Department of Chemistry, Oregon State University
Powell BA, Dai Z, Zavarin M, Zhao P, Kerstin AB (2011) Stabilization of plutonium nano–colloids by epitaxial distortion on mineral surfaces. Environ Sci Technol 45:2698–2703
Gok C, Turkozu DA, Aytas S (2011) Removal of Th(IV) ions from aqueous solution using bi-functionalized algae-yeast biosorbent. J Radioanal Nucl Chem 287:533–541
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is dedicated to the memory of Prof. Peter Beneš.
Rights and permissions
About this article
Cite this article
Lujanienė, G., Štamberg, K., Pakštas, V. et al. Study of Pu sorption behavior in natural clay. J Radioanal Nucl Chem 304, 53–59 (2015). https://doi.org/10.1007/s10967-014-3585-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-014-3585-0