Abstract
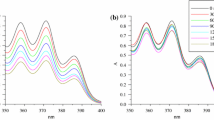
The irradiation of aqueous nitric acid solutions generates transient, reactive species that are known to oxidize neptunium. However, nitrous acid is also a long-lived product of nitric acid irradiation, which reduces neptunium. When we irradiated nitric acid solutions of neptunium and measured its speciation by UV/Vis spectroscopy, we found that at short irradiation times, oxidation of Np(V) to Np(VI) occurred due to reactions with radicals such as •OH, •NO3 and •NO2. However, at higher absorbed doses and after a sufficient amount of nitrous acid was produced, reduction of Np(VI) to Np(V) began to occur, eventually reaching an equilibrium distribution of these species depending on nitric acid concentration. Neptunium(IV) was not produced.




Similar content being viewed by others
References
Buxton GV, Greenstock CL, Helman WP, Ross AB (1988) J Phys Chem Ref Data 17:513–886
Katsumura Y, Jiang P-Y, Nagaishi R, Oishi T, Ishigure K (1991) J Phys Chem 95:4435–4439
Bugaenko LT, Roshchektaev BM (1971) Khim Vys Energy 5:472–474
Jiang P-Y, Nagaishi R, Yotsuyanagi T, Katsumura Y, Ishigure K (1994) J Chem Soc Faraday Trans 90:93–95
Elliot AJ (1989) Radiat Phys Chem 34:753–758
Gordon S, Hart EJ, Thomas JK (1964) J Phys Chem 68:1262–1264
Gogolev AV, Shilov VP, Fedoseev AM, Pikaev AK (1986) Bull Acad Sci USSR Div Chem Sci 35:422–424
Vladimirova MV (1995) Radiokhimiya 37:446–452
Siddall TH, Dukes EK (1959) J Am Chem Soc 81:790–794
Bhattacharyya PK, Saini RD (1973) Int J Radiat Phys Chem 5:91–99
Park J-Y, Lee Y-N (1988) J Phys Chem 92:6294–6302
Jiang P-Y, Katsumura Y, Ishigure K, Yoshida Y (1992) Inorg Chem 31:5135–5136
Acknowledgments
Work supported through the INL Laboratory Directed Research and Development (LDRD) program, under DOE Idaho Operations Office Contract DE-AC07-05ID14517.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mincher, B.J., Precek, M., Mezyk, S.P. et al. The role of oxidizing radicals in neptunium speciation in γ-irradiated nitric acid. J Radioanal Nucl Chem 296, 27–30 (2013). https://doi.org/10.1007/s10967-012-1937-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-012-1937-1