Abstract
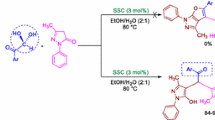
Solid base catalytic materials such as ZrO2, MgO, ZrO2–MgO were prepared by either precipitation or impregnation method and characterized by, BET, CO2-TPD, PXRD, FT-IR, ICP-OES and TEM techniques. These catalysts were used for the synthesis of bis(indolyl)methanes by the condensation of different benzaldehydes with indole under solvent free conditions in shorter reaction times (20 min) at moderate temperature (70 °C). ZrO2/MgO catalyst was found to be highly basic and also resulted in high yields of bis(indolyl)methanes up to ~99%. This methodology offers several advantages such as high quality yields, easy procedure, mild and environmentally benign conditions. TEM studies revealed that ZrO2–MgO is mesoporous (25–45 nm) in nature. ZrO2–MgO catalysts were found to be economical, efficient and were found to be highly active, recyclable and reusable up to six reaction cycles without much loss of their activity.





Similar content being viewed by others
References
T.M. Miller, V.H. Grassian, J. Am. Chem. Soc. 117, 10969 (1995)
N. Pernicone, F. Lazzerin, G. Liberti, G. Lanzavecchia, J. Catal. 14, 391 (1969)
N. Thimmaraju, S.Z.M. Shamshuddin, S.R. Pratap, S.R. Venkatesh, J. Mol. Catal A: Chem. 391, 55 (2014)
A.V. Biradar, S.B. Umbarkar, M.K. Dongare, Appl. Catal. A 285, 190 (2005)
A.P. Amrute, A. Bordoloi, N. Lucas, K. Palraj, S.B. Halligudi, Catal. Lett. 126, 286 (2008)
E.L.S. Ngee, Y. Gao, X. Chen, T. M. Lee, Z. Hu, D. Zhao, N. Yan, Ind. Eng. Chem. Res. 53, 14225 (2014)
N. Thimmaraju, S.Z.M. Shamshuddin, S. R. Pratap, K. Raja, RSC Adv. 5, 99517 (2015)
N. Thimmaraju, S.Z.M. Shamshuddin, S.R. Pratap, K. Shyam Prasad, Arab. J. Chem. (2014). doi:10.1016/j.arabjc.2014.12.011
H. Mohammed, W. Al-Hazmi, AW. Apblett, Catal. Sci. Tech. 1, 621 (2011)
X. Zhang, H. De-Hua, Q.J. Zhang, Y. Qing, X. Bo-qing, Z. Qi-ming, Appl. Catal. A Gen. 249, 107 (2003)
J.F. Shackelford, D.H. Doremus, Ceramic and glass materials: structure, properties and processing (Springer, New York, 2008)
M.B. Gawande, R.V. Jayaram, Catal. Commun. 7, 931 (2006)
M.B. Gawande, P.S. Branco, K. Parghi, J.J. Shrikhande, R.K. Pandey, C.A.A. Ghumman, N. Bundaleski, O.M.N.D. Teodoro, R.V. Jayaram, Catal. Sci. Technol. 1, 1653 (2011)
K. Tanabe, W.F. Hölderich, Appl. Catal. A 181, 399 (1999)
M. Shiri, M.A. Zolfigol, H.G. Kruger, Z. Tanbakouchian, Chem. Rev. 110, 2250 (2010)
M. Chakrabarty, N. Ghosh, R. Basak, Y. Harigaya, Tetrahedron Lett. 43, 4075 (2002)
H. Firouzabadi, N. Iranpoor, A. Jafari, J. Mol Catal A 244, 168 (2006)
C. Breitkopf, A. Garsuch, H. Papp, Appl. Catal. A 296, 148 (2005)
C. Praveen, Y.W. Sagayaraj, P.T. Perumal, Tetrahedron Lett. 50, 644 (2009)
H.-E. Qu, C. Xiao, N. Wang, K.-H. Yu, Q.-S. Hu, L.-X. Liu, Molecules. 16, 3855 (2011)
A.V. Reddy, K. Ravinder, V.L.N. Reddy, T.V. Goud, V. Ravikanth, Y. Venkateswaralu, Synth. Commun. 33, 3687 (2003)
M.L. Kantam, S. Laha, J. Yadav, B. Sreedhar, Tetrahedron Lett. 47, 6213 (2006)
A.C. Shaikh, C. Chen. Chin. J. Chem. Soc. 58, 899 (2011)
J.S. Yadav, B.V. Subba Reddy, C.V.S.R. Murthy, G.M. Kumar, C. Madan, Synthesis 5, 783 (2001)
C.C. Silveira, S.R. Mendes, F.M. Libero, E.J. Lenardao, G. Perin, Tetrahedron Lett. 50, 6060 (2009)
N. Kapuriya, R. Kakadiya, M.M. Savant, A.M. Pansuriya, C.V. Bhuva, A.S. Patel, P.V. Pipaliya, V.B. Audichya, S. Gangadharaiah, S.M. Anandalwar, J.S. Prasad, A. Shah, Y.T. Naliapara, Indian J. Chem. 51, 1032 (2012)
M. Shyamsundar, S.Z.M. Shamshuddin, J.N. Sahu, Korean J. Chem. Eng. 30, 2186 (2013)
L. Zhao, J. Zhao, M. Luo, G. Liu, J. song, Y. Zhang, Catal. Commun. 6, 617 (2005)
L.A. Perez-Maqueda, E. Matijevi, J. Mater. Res. 12, 3286 (1997)
K. Hadjiivanov, J.C. Lavalley, Catal. Commun. 2, 129 (2001)
M.H. Lee, D.G. Park, Bull. Korean Chem. Soc. 24, 1437 (2003)
M. Rekha, H.R. Manjunatha, N. Nagaraju, J. Ind. Eng. Chem. 19, 337 (2013)
Acknowledgements
The authors are grateful for the financial support given by the Vision Group of Science and Technology (VGST), Government of Karnataka (GRD-375). The authors acknowledge CENSE-IISc for TEM analysis and IISc-Bangalore for ICP-OES. The authors are thankful to Poornaprajna institute of scientific research, Bangalore PXRD, FTIR analysis. The authors are also thankful to the Department of Physical Chemistry, IISc- Bangalore for 1H NMR, 13C NMR, GC-MS.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shyamsundar, M., Shamshuddin, S.Z.M., Vasanth, V.T. et al. Simple but efficient synthesis of bis(indolyl)methanes by the condensation reaction of substituted benzaldehydes with indole over mesoporous ZrO2–MgO green catalysts under solvent free conditions. J Porous Mater 24, 1003–1011 (2017). https://doi.org/10.1007/s10934-016-0340-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-016-0340-7