Abstract
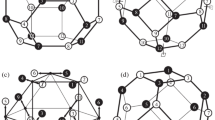
Isomeric diamondoids with the same number \(n\) of adamantane units (or cells), which share the same molecular formula \(\text{ C}_\mathrm{Q}(\text{ CH})_\mathrm{T}(\text{ CH}_{2})_\mathrm{S}\), can be divided into valence isomers by partitioning the number \(C \) of their carbon atoms according to whether they are Quaternary, Tertiary, or Secondary: \(C =Q +T +S\). Each [\(n\)]diamondoid has a dualist (or inner dual) with \(n\) vertices (situated at centers of adamantane units), and edges connecting vertices of adjacent adamantane units sharing a chair-shaped hexagon of carbon atoms. Such a dualist is characterized by a quadruplet of indices (denoted as p, s, t, q for primary, secondary, tertiary, or quaternary) specifying again the connectivity of each vertex by assimilating it with a virtual carbon atom. The diamond lattice is self-dual. Dualists help in classifying diamondoids as catamantanes with acyclic dualists, perimantanes with dualists having chair-shaped six-membered rings, or coronamantanes with dualists having only higher-membered rings. In turn, catamantanes can be either regular when they have formulas \(\text{ C}_{4n+6}\text{ H}_{4n+12}\), or irregular when the numbers of carbon and hydrogen atoms are lower than the above values for the given numbers \(n\) of adamantane units. Regular catamantanes can have branched or non-branched dualists and they are isomeric when having the same \(n\). Partitioned formulas reflect the branching patterns, encoded in their dualists. Partition formulas and codes are presented for all possible diamondoids with up to 7 adamantane units. A remarkable symmetry is observed for the table of partition periodic table of regular catamantanes with up to 7 adamantane units. Isomeric irregular catamantanes with six or more adamantane units may be valence-isomeric (or homomeric, sharing both the molecular and the partitioned formulas), or heteromeric when they have different partitioned formulas.







Similar content being viewed by others
References
R. Todeschini, V. Consonni, Molecular Descriptors for Chemoinformatics, 2nd edn (Wiley-VCH, Weinheim, 2009)
A.T. Balaban, M. Banciu, V. Ciorba, Annulenes, Benzo-, Hetero-, Homo-Derivatives and Their Valence Isomers (CRC Press, Boca Raton, 1986)
A.T. Balaban, F. Harary, Chemical graphs. V. Enumeration and proposed nomenclature of benzenoid cata-condensed polycyclic aromatic hydrocarbons. Tetrahedron 24, 2505–2516 (1968)
A.T. Balaban, Chemical graphs. VII. Proposed nomenclature of branched cata-condensed benzenoid polycyclic hydrocarbons. Tetrahedron 25, 2949–2956 (1969)
A.T. Balaban, M. Pompe, QSPR for physical properties of cata-condensed benzenoids using two simple dualist-based descriptors. J. Phys. Chem. A 111, 2448–2454 (2007)
A.T. Balaban, S. Aziz, A.D. Manikpuri, P.V. Khadikar, Simple correlations of the \(\pi \)-electron energy and other properties of cata-condensed benzenoids. J. Indian Chem. Soc. 88, 87–97 (2011)
A.T. Balaban, D. Biermann, W. Schmidt, Chemical graphs. 41. Dualist graph approach for correlating Diels-Alder reactivites of polycyclic aromatic hydrocarbons. Nouv. J. Chim. 9, 443–449 (1985)
D.J. Klein, Rigorous results for branched polymer models with excluded volume. J. Chem. Phys. 75, 5186–5189 (1981)
L. Bytautas, D.J. Klein, Formula periodic table for acyclic hydrocarbon isomer classes: combinatorially averaged graph invariants. Phys. Chem. Chem. Phys. 1, 5565–5572 (1999)
L. Bytautas, D.J. Klein, T.G. Schmalz, All acyclic hydrocarbons: formula periodic table and average properties via chemical combinatorics. New J. Chem. 24, 329–336 (2000)
A.T. Balaban, Chemical graphs. XXVII. Enumeration and codification of staggered conformations of alkanes. Rev. Roum. Chim. 21, 1049–1071 (1976)
A.T. Balaban, Enumeration of catafusenes, diamondoid hydrocarbons and staggered alkane C-rotamers. MATCH Commun. Math. Comput. Chem. 2, 51–61 (1976)
A.T. Balaban, V. Baciu, Chemical graphs. XXXV. Application of Polya’s theorem to catamantanes. MATCH Commun. Math. Comput. Chem. 4, 131–159 (1978)
A.T. Balaban, P. von R. Schleyer, Systematic classification and nomenclature of diamond hydrocarbons. I. Graph-theoretical enumeration of polymantanes. Tetrahedron 34, 3599–3609 (1978)
A.T. Balaban, Partitioned-formula tables for diamond hydrocarbons (diamondoids). J. Chem. Inf. Model. 52, 2856–2869 (2012)
J.E.P. Dahl, J.M. Moldowan, Z. Wei, P.A. Lipton, P. Denisevich, R. Gat, S. Liu, P.R. Schreiner, R.M.K. Carlson, Synthesis of higher diamondoids and implication for their formation in petroleum. Angew. Chem. Int. Ed. 49, 9881–9885 (2010)
S. Landa, V. Mahacek, Sur l’adamantane, nouvel hydrocarbure extrait du naphte. Coll. Czech. Chem. Commun. 5, 1–5 (1933)
V. Prelog, B. Seiwerth, Über die synthese des adamantans. Ber. Dtsch. Chem. Ges. 74, 1644–1648 (1941)
V. Prelog, B. Seiwerth, Über eine neue, ergiebigere darstellung des adamantans. Ber. Dtsch. Chem. Ges. 74, 1679–1772 (1941)
P. von R. Schleyer, A simple preparation of adamantane. J. Am. Chem. Soc. 79, 3292 (1957)
C.A. Cupas, P. von R. Schleyer, D.J. Trecker, Congressane. J. Am. Chem. Soc. 87, 917–918 (1965)
V.Z. Williams Jr, P. von R. Schleyer, G.J. Gleicher, L.B. Rodewald, Triamantane. J. Am. Chem. Soc. 88, 3862–3863 (1966)
H.W. Whitlock Jr, M.W. Siefken, Tricyclo [4.4.0.0\(^{3,8}\)] decane to adamantane rearrangement. J. Am. Chem. Soc. 90, 4929–4939 (1968)
E.M. Engler, M. Farcasiu, A. Sevin, J.M. Cense, P. von R. Schleyer, On the mechanism of adamantane rearrangements. J. Am. Chem. Soc. 95, 5769–5771 (1973)
M.A. McKervey, Adamantane rearrangements. Chem. Soc. Rev. 3, 479–512 (1974)
P. von R. Schleyer, My thirty years in hydrocarbon cages: from adamantane to dodecahedrane, in Cage Hydrocarbons, ed. by G.A. Olah (Wiley-Interscience, New York, 1990), pp. 1–38
R.C. Fort Jr, P. von R. Schleyer, Adamantane. Consequences of the diamondoid structure. Chem. Rev. 64, 277–300 (1964)
T.M. Gund, P. von R. Schleyer, P.H. Gund, W.T. Wipke, Computer assisted graph-theoretical analysis of complex mechanistic problems in polycyclic hydrocarbons. The mechanism of diamantane formation from various pentacyclodecanes. J. Am. Chem. Soc. 97, 751 (1975)
M.A. McKervey, Synthetic approaches to large diamondoid hydrocarbons. Tetrahedron 36, 971–992 (1980)
M.A. McKervey, J.J. Rooney, Catalytic routes to adamantane and its homologues, in Cage Hydrocarbons, ed. by G.A. Olah (Wiley-Interscience, New York, 1990), pp. 39–64
W. Burns, T.R.B. Mitchell, M.A. McKervey, J.J. Rooney, G. Ferguson, P. Roberts, Gas-phase reactions on platinum. Synthesis and crystal structure of anti-tetramantane, a large diamondoid fragment. Chem. Commun. 893–895 (1976)
R.C. Fort Jr, Adamantane. The Chemistry of Diamond Molecules (Marcel Dekker, New York, 1976)
P.L.B. Araujo, E.S. Araujo, G.A. Mansoori, Diamondoids: occurrence in fossil fuels, applications in petroleum exploration and fouling in petroleum production. A reveiew paper. Int. J. Oil Gas Coal Technol. 5, 316–367 (2012)
A.T. Balaban, Diamond hydrocarbons and related structures, in Diamond and Related Nanostructures, ed. by M.V. Diudea, C.L. Nagy (Springer, Berlin, 2013)
G.A. Mansoori, P.L.B. de Araujo, E.S. de Araujo, Diamondoid Molecules with Applications in Biomedicine, Materials Science Nanotechnology and Petroleum Science (Wiley, New York, 2012)
J.E. Dahl, J.M. Moldowan, K.E. Peters, G.E. Claypool, M.A. Rooney, G.E. Michael, M.R. Mello, M.L. Kohnen, Diamondoid hydrocarbons as indicators of natural oil cracking. Nature 399, 54–47 (1999)
J.E. Dahl, S.G. Liu, R.M.K. Carlson, Isolation and structure of higher diamondoids, nanometer-sized diamond molecules. Science 299, 96–99 (2003)
J.W. Dahl, J.M. Moldowan, T.M. Peakman, J.C. Clardy, E. Lobkowski, M.M. Olmstead, P.W. My, T.J. Davis, J.W. Steeds, K.E. Peters, A. Pepper, A. Ekuan, R.M.K. Carlson, Isolation and structural proof of the large diamond molecule, cyclohexamantane \((\text{ C}_{26}\text{ H}_{30})\). Angew. Chem. Int. Ed. 42, 2040–2044 (2003)
A.T. Balaban, D.J. Klein, J.E. Dahl, R.M.K. Carlson, Molecular descriptors for natural diamondoid hydrocarbons and quantitative structure-property relationships for their chromatographic data. Open Org. Chem. J. 1, 13–31 (2007)
P.R. Schreiner, A.A. Fokin, H.P. Reisenauer, B.A. Tkachenko, E. Vass, M.M. Olmstead, D. Bläser, R. Boese, J.E.P. Dahl, R.M.K. Carlson, [123]Tetramantane: parent of a new family of \(\sigma \)-helicenes. J. Am. Chem. Soc. 131, 11292–11293 (2009)
H. Schwertfeger, C. Würtele, H. Hausmann, J.E.P. Dahl, R.M.K. Carlson, A.A. Fokin, P.R. Schreiner, Selective preparation of diamondoid fluorides. Adv. Synth. Catal. 451, 1041–1054 (2009)
A.A. Fokin, P.A. Gunchenko, A.A. Novikovsky, T.E. Shubina, B.V. Chernyaev J.E.P. Dahl, R.M.K. Carlson, A.G. Yurchenko, P.R. Schreiner, Photoacetylation of diamondoids: selectivities and mechanism. Eur. J. Org. Chem. 5153–5161 (2009)
A.A. Fokin, B.A. Tkachenko, N.A. Fokina, H. Hausmann, M. Serafin, J.E.P. Dahl, R.M.K. Carlson, P.R. Schreiner, Reactivities of the prism-shaped diamondoids [1(2)3]tetramantane and [12312]hexamantane (cyclohexamantane). Chem. Eur. J. 15, 3851–3862 (2009)
A.A. Fokin, A. Merz, N.A. Fokina, H. Schwertfeger, S.L. Liu, J.E.P. Dahl, R.M.K. Carlson, P.R. Schreiner, Synthetic routes to aminotriamantanes, topological analogues of the neuroprotector memantine. Synthesis, 909–912 (2009)
A.A. Fokin, T.S. Zhuk, A.E. Pashenko, P.O. Dral, P.A. Gunchenko, J.E.P. Dahl, R.M.K. Carlson, T.V. Koso, M. Serafin, P.R. Schreiner, Oxygen-doped nanodiamonds: synthesis and functionalizations. Org. Lett. 11, 3068–3071 (2009)
A.A. Fokin, P.R. Schreiner, Band gap tuning in nanodiamonds: first principle computational studies. Mol. Phys. 107, 823–830 (2009)
A.A. Fokin, B.A. Tkachenko, P.A. Gunchenko, D.V. Gusev, P.R. Schreiner, Functionalized nanodiamonds. Part I. An experimental assessment of diamantane and computational predictions for higher diamondoids. Chem. Eur. J. 11, 7091–7101 (2005)
A.T. Balaban, J. Brunvoll, J. Cioslowski, B.N. Cyvin, S.J. Cyvin, I. Gutman, H. Wenchen, H. Wenjie, J.V. Knop, M. Kovacević, W.R. Müller, W.R. Szymanski, R. Tosić, N. Trinajstić, Enumeration of benzenoid and coronoid hydrocarbons. Z. Naturforsch. 42c, 863–870 (1987)
G. Caporossi, P. Hansen, Enumeration of polyhex hydrocarbons to h = 21. J. Chem. Inf. Model. 38, 610–619 (1998)
I. Gutman, How many benzenoid hydrocarbons? Bull. Chem. Technol. Maced. 21, 53–56 (2002)
G. Brinkmann, G. Caporossi, P. Hansen, A survey and new results on computer enumeration of polyhex and fusene hydrocarbons. J. Chem. Inf. Comput. Sci. 43, 842–851 (2003)
G. Brinkmann, C. Grothaus, I. Gutman, Fusenes and benzenoids with perfect matchings. J. Math. Chem. 42, 909–924 (2007)
M. Vöge, A.J. Gutmann, I. Jensen, On the number of benzenoid hydrocarbons. J. Chem. Inf. Comput. Sci. 42, 456–466 (2002)
I. Jensen, A parallel algorithm for the enumeration of benzenoid hydrocarbons. J. Stat. Mech. P02065 (2009)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balaban, A.T. Diamond hydrocarbons revisited: partitioned formula tables of diamondoids. J Math Chem 51, 1043–1055 (2013). https://doi.org/10.1007/s10910-012-0130-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10910-012-0130-7