Abstract
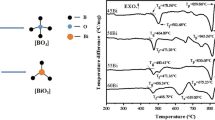
xB2O3-(1 − x) (60SiO2–15Al2O3–15MgO–10CaO) glasses with various x around 5–30 mol% at the step of 5 mol% are prepared by the melting-quenching method at 1,450 °C for 3 h. The IR data indicate that replacing other components by B2O3 levels up the amount of [BO4] tetrahedron and boron-oxygen rings, forcing [AlO4] tetrahedron to drop gradually. The glass transition temperature (Tg), crystallization temperature (Tc) and thermal stability (∆T) rise when B2O3 is present in lower concentrations (≤20 mol%); accordingly the glass network enhances. However, further increase in concentration of B2O3 (>20 mol%) reduces the Tg, Tc and ∆T. We utilize temperature-gradient furnace to measure the G6 glass, while the re-melt sample and crystallized phase are observed from polarizing microscope destroying microstructure. As B2O3 content move up, falls in dielectric constant (εr) and loss (tanδ) can be associated with drop of cations polarizability as well as concentration, along with glass network rigidity ascending.






Similar content being viewed by others
References
X. Zhang, Y. Yue, H. Wu, J. Mater. Sci. 24, 8 (2013)
A. Dahshan, J. Non-Cryst. Solids 354, 26 (2008)
J. Wu, J.F. Stebbins, J. Non-Cryst. Solids. 362, (2013)
N.O. Dantas, W.E. Ayta, A.C. Silva, N.F. Cano, S.W. Silva, P.C. Morais, Spectrochim Acta. A 81, 1 (2011)
A. Efimov, J. Non-Cryst. Solids. 203, (1996)
Y. Cheng, H. Xiao, W. Guo, W. Guo, Ceram. Int. 33, 7 (2007)
H. Darwish, M. Gomaa, J. Mater. Sci. 17, 1 (2006)
Y. Cheng, H. Xiao, C. Shuguang, B. Tang, Phys. B 404, 8–11 (2009)
T. Sun, H. Xiao, W. Guo, X. Hong, Ceram. Int. 36, 2 (2010)
M. Arora, S. Baccaro, G. Sharma, D. Singh, K.S. Thind, D.P. Singh, Nucl. Instrum. Meth. B 267, 5 (2009)
G. El-Damrawi, K. El-Egili, Phys. B 299, 1 (2001)
E. Kamitsos, A. Patsis, G.D. Chryssikos, J. Non-Cryst. Solids 152, 2 (1993)
L.-S. Du, J.F. Stebbins, J. Non-Cryst. 351, 43–45 (2005)
V. Gerasimov, O. Spirina, Glass Ceram. 61, 5–6 (2004)
M. Reben, H. Li, Int. J. Appl. Glass Sci. 2, 2 (2011)
A. Hrubý, Czech. J. Phys. B 22, 11 (1972)
S. Mahadevan, A. Giridhar, A. Singh, J. Non-Cryst. Solids 88, 1 (1986)
A.S. Weber, R. Hodyss, P.V. Johnson, K. Willacy, I. Kanik, Astrophys. J. 703, 1 (2009)
P.A. Bingham, R.J. Hand, S.D. Forder, Mater. Res. Bull. 41, 9 (2006)
K. Joseph, K.V.G. Kutty, M.C. Goswami, P.R.V. Rao, Thermochim. Acta 587, (2014)
J. Chovanec, M. Chromčíková, P. Pilný, J. Shánělová, J. Málek, M. Liška, J. Therm. Anal. Calorim. 114, 3 (2013)
X. Zhang, Z. Du, H. Wu, Y. Yue, Surf. Rev. Lett. 20, 03 (2013)
S.-P. Szu, C.-Y. Lin, Mater. Chem. Phys. 82, 2 (2003)
S. Sindhu, S. Sanghi, A. Agarwal, V. Seth, N. Kishore, Mater. Chem. Phys. 90, 1 (2005)
V. Dimitrov, T. Komatsu, J. Solid State Chem. 163, 1 (2002)
T. Inoue, T. Honma, V. Dimitrov, T. Komatsu, J. Solid State Chem. 183, 12 (2010)
B.H. Jung, D.N. Kim, H.-S. Kim, J. Non-Cryst. Solids 351, 40 (2005)
Y. Yue, X. Zhang, Y. Xu, S. Huang, P. Chen, Mater. Lett. (2014)
M. Gomaa, H. Abo-Mosallam, H. Darwish, J. Mater. Sci. 20, 6 (2009)
G. Srinivasarao, N. Veeraiah, J. Solid State Chem. 166, 1 (2002)
G. Srinivasarao, N. Veeraiah, J. Phys. Chem. Solids 63, 4 (2002)
B.-H. Jung, S.-J. Hwang, H.-S. Kim, J. Eur. Ceram. Soc. 25, 13 (2005)
J. Lumeau, L. Glebova, G.P. Souza, E.D. Zanotto, L.B. Glebov, J. Non-Cryst. Solids 354, 42 (2008)
T. Zhang, M.-Q. Wu, S.-R. Zhang, J. Xiong, J.-M. Wang, D.-H. Zhang, F.-M. He, Z.-P. Li, Chin. Phys. B 21, 7 (2012)
S. Ramesh, A. Yahaya, A. Arof, Solid State Ionics 152, (2002)
R.S. Gedam, D.D. Ramteke, J. Phys. Chem. Solids 74, 7 (2013)
F. Salman, A. Mekki, J. Non-Cryst. Solids 357, 14 (2011)
E. Ozel, H. Yurdakul, S. Turan, M. Ardit, G. Cruciani, M. Dondi, J. Eur. Ceram. Soc. 30, 16 (2010)
Acknowledgments
This work is supported by Natural Science Foundation of Shandong Province (Nos. ZR2012EMM019), Natural Science Foundation of Shandong Province (Nos. ZR2011EMQ005) and Natural Science Foundation (Nos. 51172093, 51042009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, S., Li, S., Wu, F. et al. Effect of B2O3 on Structure and Properties of CaO–MgO–B2O3–Al2O3–SiO2 Glasses. J Inorg Organomet Polym 25, 816–822 (2015). https://doi.org/10.1007/s10904-015-0164-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-015-0164-y