Abstract
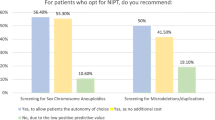
Using cell-free DNA in maternal serum to detect fetal aneuploidy has been shown to have high sensitivity and specificity. The purpose of this study was to assess attitudes and knowledge of Maternal-Fetal Medicine (MFM) fellows regarding noninvasive prenatal testing (NIPT). A 13 question survey was sent via listserv to US-based MFM fellows. One hundred sixteen fellows responded, a 42.3 % response rate, with >75 % reporting they are comfortable ordering NIPT. Most (82 %) preferred that a patient discuss options with a provider or genetic counselor. Three common methods used to learn about NIPT were: formal educational activities (n = 78, 69 %), self-review of the literature (n = 76, 67 %), and discussions with peers (n = 73, 65 %). On questions related to trisomy 21, accuracy was >70 %. However, accuracy was lower regarding use in twin pregnancies (42 %) and monosomy X screening (50 %).

Similar content being viewed by others
References
American College of Obstetricians and Gynecologists Committee on Genetics. Committee opinion no. 545. (2012). Noninvasive prenatal testing for fetal aneuploidy. Obstetrics and Gynecology, 120(6), 1532–1534.
Benn, P. A., & Chapman, A. R. (2010). Ethical challenges in providing noninvasive prenatal diagnosis. Current Opinion in Obstetrics and Gynecology, 22(2), 128–134.
Bianchi, D. W., Platt, L. D., Goldberg, J. D., Abuhamad, A. Z., Sehnert, A. J., Rava, R. P., et al. (2012). Genome-wide fetal aneuploidy detection by maternal plasma DNA sequencing. Obstetrics and Gynecology, 119(5), 890–901.
Bianchi, D. W., Prosen, T., Platt, L. D., Goldberg, J. D., Abuhamad, A. Z., Rava, R. P., et al. (2013). Massively parallel sequencing of maternal plasma DNA in 113 cases of fetal nuchal cystic hygroma. Obstetrics and Gynecology, 121(5), 1057–1062.
Bianchi, D. W., Parker, R. L., Wentworth, J., Madankumar, R., Saffer, C., Das, A. F., et al. (2014). DNA sequencing versus standard prenatal aneuploidy screening. New England Journal of Medicine, 370(9), 799–808.
Brierley, K. L., Blouch, E., Cogswell, W., Homer, J. P., Pencarinha, D., Stanislaw, C. L., et al. (2012). Adverse events in cancer genetic testing: medical, ethical, legal, and financial implications. Cancer Journal, 18(4), 303–309.
Madan, K., & Breuning, M. H. (2014). Impact of prenatal technologies on the sex ratio in India: an overview. Genetics in Medicine, 16(6), 425–32.
Palomaki, G. E., Kloza, E. M., Lambert-Messerlian, G. M., Haddow, J. E., Neveux, L. M., Ehrich, M., et al. (2011). DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study. Genetics in Medicine, 13(11), 913–920.
Plon, S. E., Cooper, H. P., Parks, B., Dhar, S. U., Kelly, P. A., Weinberg, A. D., et al. (2011). Genetic testing and cancer risk management recommendations by physicians for at-risk relatives. Genetics in Medicine, 13(2), 148–154.
Sayres, L. C., Allyse, M., Norton, M. E., & Cho, M. K. (2011). Cell-free fetal DNA testing: a pilot study of obstetric healthcare provider attitudes toward clinical implementation. Prenatal Diagnosis, 31(11), 1070–1076.
Selkirk, C. G., Weissman, S. M., Anderson, A., & Hulick, P. J. (2013). Physicians’ preparedness for integration of genomic and pharmacogenetic testing into practice within a major healthcare system. Genetic Testing and Molecular Biomarkers, 17(3), 219–225.
Vora, N. L., & O’Brien, B. M. (2014). Noninvasive prenatal testing for microdeletion syndromes and expanded trisomies: Proceed with caution. Obstetrics and Gynecology, 123(5), 1097–1099.
Wapner RJ, Babiarz JE, Levy B, et al. (2015) Expanding the scope of noninvasive prenatal testing: detection of fetal microdeletion syndromes. American journal of obstetrics and gynecology, 212(3): 332.e1-9
You, Y., Sun, Y., Li, X., Li, Y., Wei, X., Chen, F., et al. (2013). Integration of targeted sequencing and NIPT into clinical practice in a Chinese family with maple syrup urine disease. Genetics in Medicine, 16, 594–600.
Acknowledgments
We would like to acknowledge the Society of Maternal Fetal Medicine and Dr. Suneet Chauhan for sending the survey to the MFM fellows.
Funding Sources
None.
Conflict of Interest:
Paul Swaney, Emily Hardisty, Lauren Sayres, Samantha Wiegand and Neeta Vora declare that they have no conflict of interest.
Human Studies and Informed Consent:
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 (5). Informed consent was obtained from all patients for being included in the study.
Animal Studies:
This article does not contain any studies with animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Swaney, P., Hardisty, E., Sayres, L. et al. Attitudes and Knowledge of Maternal-Fetal Medicine Fellows Regarding Noninvasive Prenatal Testing. J Genet Counsel 25, 73–78 (2016). https://doi.org/10.1007/s10897-015-9844-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10897-015-9844-6