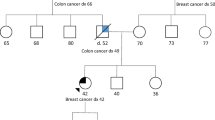
Abstract
Genetic testing recommendations for hereditary breast and ovarian cancer involve pedigree analysis and consultation of testing guidelines. The testing landscape for hereditary cancer syndromes is shifting as multiplex panel tests become more widely integrated into clinical practice. The purpose of the current study was to assess how genetic counselors utilize pedigrees to make recommendations for genetic testing, to determine consistency of these recommendations with National Comprehensive Cancer Network (NCCN) Guidelines and to explore current use of multiplex panel testing. Sixty-nine genetic counselors were recruited through the National Society of Genetic Counselors Cancer Special Interest Group’s Discussion Forum. Participation involved pedigree analysis and completion of an online questionnaire assessing testing recommendations and use of multiplex panel testing. Pedigree analysis and test recommendations were scored for consistency with NCCN guidelines. The average score was 12.83/15 indicating strong consistency with NCCN guidelines. Participants were more likely to consider multiplex testing when pedigrees demonstrated highly penetrant dominant inheritance but were not indicative of a particular syndrome. Participant concerns about multiplex panel testing include limited guidelines for both testing eligibility and medical management. This study demonstrates high utilization of pedigree analysis and raises new questions about its use in multiplex genetic testing.





Similar content being viewed by others
References
Ambry Genetics. (2012). BreastNext. Retrieved April 22, 2012 from https://doi.org/ambrygen.com/tests/breastnext.
American Society of Clinical Oncology. (2003). American Society of Clinical Oncology policy statement update: genetic testing for cancer susceptibility. Journal of Clinical Oncology, 21(12), 2397–2406. doi:https://doi.org/10.1200/JCO.2009.27.0660.
Amir, E., Freedman, O. C., Seruga, B., & Evans, D. G. (2010). Assessing women at high risk of breast cancer: a review of risk assessment models. Journal of the National Cancer Institute, 102(10), 680–691. doi:https://doi.org/10.1093/jnci/djq088.
Antoniou, A., Pharoah, P. D., Narod, S., Risch, H. A., Eyfjord, J. E., Hopper, J. L., et al. (2003). Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case series unselected for family history: a combined analysis of 22 studies. American Journal of Human Genetics, 72(5), 1117–1130.
Association for Molecular Pathology et al. v. Myriad Genetics, Inc, et al. 12–389 US. (2013). Retrieved October 12, 2013 from https://doi.org/www.supremecourt.gov/opinions/12pdf/12–398_1b7d.pdf.
Bennett, R. L. (2010). Practical guide to the genetic family history (2nd ed.). Hoboken: Wiley-Blackwell.
Bennett, R. L., Steinhaus, K. A., Uhrich, S. B., O’Sullivan, C. K., Resta, R. G., Lochner-Doyle, D., et al. (1995). Recommendations for standardized human pedigree nomenclature. Pedigree Standardization Task Force of the National Society of Genetic Counselors. American Journal of Human Genetics, 56, 745–752.
Berliner, J. L., Fay, A. M., Shelly, A., Cummings, S. A., Burnett, B., & Tillmanns, T. (2013). NSGC practice guideline: risk assessment and genetic counseling for hereditary breast and ovarian cancer. Journal of Genetic Counseling, 22, 155–163. doi:https://doi.org/10.1007/s10897-012-9547-1.
Breast Cancer Linkage Consortium. (1999). Cancer risks in BRCA2 mutation carriers. Journal of the National Cancer Institute, 91(15), 1310–1316.
Chompret, A., Abel, A., Stoppa-Lyonnet, D., Brugieres, L., Pages, S., Feunteun, J., et al. (2001). Sensitivity and predictive value of criteria for p53 germline mutation screening. Journal of Medical Genetics, 38, 43–47.
Culver, J. O., Lowstuter, K., & Bowling, L. (2006). Assessing breast cancer risk and BRCA1/2 carrier probability. Breast Disease, 27:5–20.
Domchek, S. M., Bradbury, A., Garber, J. E., Offit, K., & Robson, M. E. (2013). Multiplex genetic testing for cancer susceptibility: out on the high wire without a net? Journal of Clinical Oncology, 31(10), 1267–70. doi:https://doi.org/10.1200/JCO.2012.46.9403.
Euhus, D. M., Smith, K. C., Robinson, L., Stucky, A., Olopade, O. I., Cummings, S., et al. (2002). Pretest prediction of BRCA1 or BRCA2 mutation by risk counselors and the computer model BRCAPRO. Journal of the National Cancer Institute, 94(11), 844–851. doi: 10.1093.
Frezzo, T. M., Rubinstein, W. S., Dunham, D., & Ormond, K. E. (2003). The genetic family history as a risk assessment tool in internal medicine. Genetics in Medicine, 5(2), 84–91. doi: 10.1097.
Gonzalez, K., Buzin, C., Noltner, K., Gu, D., Li, W., Malkin, D., et al. (2009a). High frequency of de novo mutations in Li-Fraumeni syndrome. Journal of Medical Genetics, 46(10), 689–693. doi:https://doi.org/10.1136/jmg.2008.058958.
Gonzalez, K., Noltner, K., Buzin, C., Gu, D., Wen-Fong, C., Nguyen, V., et al. (2009b). Beyond Li Fraumeni Syndrome: clinical characteristics of families with p53 germline mutations. Journal of Clinical Oncology, 27(8), 1250–1256. doi:https://doi.org/10.1200/JCO.2008.16.6959.
Hampel, H., Stephens, J. A., Pukkala, E., Sankila, R., Aaltonen, L. A., Mecklin, J. P., et al. (2005). Cancer risk in hereditary nonpolyposis colorectal cancer syndrome: later age of onset. Gastroenterology, 129(2), 415–421.
Hemel, D., & Domchek, S. M. (2010). Breast cancer predisposition syndromes. Hematology/Oncology Clinics of North America, 24, 799–814. doi:https://doi.org/10.1016/j.hoc.2010.06.004.
McCuaig, J. M., Armel, S. R., Novokmet, A., Ginsburg, O. M., Demsky, R., Narod, S. A., et al. (2012). Routine TP53 testing for breast cancer under age 30: ready for prime time? Familial Cancer, 11(4), 607–613. doi:https://doi.org/10.1007/s10689-012-9557-z.
National Comprehensive Cancer Network Guidelines. (2013a). National Comprehensive Cancer Network. Genetic/Familial High-Risk Assessment: Breast and Ovarian (version 4.2013). Accessed 10/1/2013. https://doi.org/www.nccn.org.
National Comprehensive Cancer Network Guidelines. (2013b). National Comprehensive Cancer Network. Colorectal Cancer Screening (version 2.2013). Accessed 10/16/2013. https://doi.org/www.nccn.org.
National Society of Genetic Counselor (2012). Professional Status Survey: Work Environment. Retrieved January 28, 2013 from https://doi.org/www.nsgc.org/Publications/ProfessionalStatusSurvey/tabid/142/Default.aspx.
Nisbett, R. E., & Wilson, T. E. (1977). Telling more than we can know: verbal reports on mental processes. Psychological Review, 84(3), 231–259.
Pilarski, R. (2009). Cowden syndrome: a critical review of the clinical literature. Journal of Genetic Counseling, 18(1), 13–27. doi:https://doi.org/10.1007/s10897-008-9187-7.
Pyeritz, R. (2012). The family history: the first genetic test, and still useful after all those years? Genetic Medicine, 14(1), 3–9. doi:https://doi.org/10.1038/gim.0b013e3182310bcf.
Survey Monkey. (1999) Retrieved April 22, 2012 from https://doi.org/www.surveymonkey.com.
Tan, M. H., Mester, J. L., Ngeow, J., Rybicki, L. A., Orloff, M. S., & Eng, C. (2012). Lifetime cancer risks in individuals with germline PTEN mutations. Clinical Cancer Research, 18(2), 400–407. doi:https://doi.org/10.1158/1078-0432.CCR-11-2283.
Uhlmann, W., Schuette, J., & Yashar, B. (2009). A guide to genetic counseling (2nd ed.). Hoboken: John Wiley and Sons.
United States Preventive Services Task Force (USPSTF). (2005). Genetic risk assessment and BRCA mutation testing for breast and ovarian cancer susceptibility: recommendation statement. Annals of Internal Medicine, 143(5), 355–361. doi:https://doi.org/10.7326/0003-4819-143-5-200509060-00011.
Wang, G., Beattie, M. S., Ponce, N. A., & Phillips, A. (2011). Eligibility criteria in private and public coverage policies for BRCA genetic testing and genetic counseling. Genetics in Medicine, 13(12), 1045–1050. doi:https://doi.org/10.1097/GIM.0b013e31822a8113.
Weissman, S. M., Burt, R., Church, J., Erdman, S., Hampel, H., Holter, S., et al. (2012). Identification of individuals at risk for Lynch syndrome using targeted evaluations and genetic testing: National Society of Genetic Counselors and the Collaborative Group of the Americas on Inherited Colorectal Cancer joint practice guideline. Journal of Genetic Counseling, 21(4), 484–493. doi:https://doi.org/10.1007/s10897-011-9465-7.
Acknowledgments
We are grateful to the genetic counselors who took the time to participate in this survey, Laura Conway for her support and feedback, Steven Robbins for his input in design and interpretation, and Michael Kallan and Colleen Brensinger for their assistance with the statistical analysis.
Conflict of Interest
Meghan Lundy, Andrea Forman, Kathleen Valverde and Lisa Kessler declare they have no conflict of interest.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all participants for being included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lundy, M.G., Forman, A., Valverde, K. et al. An Investigation of Genetic Counselors’ Testing Recommendations: Pedigree Analysis and the Use of Multiplex Breast Cancer Panel Testing. J Genet Counsel 23, 618–632 (2014). https://doi.org/10.1007/s10897-014-9692-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10897-014-9692-9