Abstract
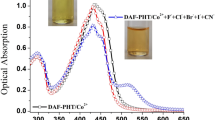
A novel 8-hydroxyquinoline-based fluorescent and colorimetric chemosensor was designed, synthesized and fully characterized. The sensor showed high selectivity and sensitivity toward Al3+ over other tested cations in EtOH/H2O (1:99, v/v) medium. The increase in fluorescence intensity was linearly proportional to the concentration of Al3+ with a detection limit of 7.38 × 10−6 M. Moreover, the sensor exhibited an obvious color change from yellow to black in the presence of Fe2+ and Fe3+ in EtOH/THF (99:1, v/v) solution. The absorbance changes showed a linear response to iron ions with the detection limits of 4.24 × 10−7 M and 5.60 × 10−7 M for Fe2+ and Fe3+, respectively. Thus, this chemosensor provides a novel approach for selectively recognition of Al3+, Fe3+ and Fe2+ among environmentally relevant metal ions.















Similar content being viewed by others
References
Banks WA, Kastin AJ (1989) Aluminum-Induced neurotoxicity: Alterations in membrane function at the blood-brain barrier. Neurosci Biobehav Rev 13(1):47–53
Nayak P (2002) Aluminum: impacts and disease. Environ Res 89(2):101–115
Good PF, Olanow CW, Perl DP (1992) Neuromelanin-containing neurons of the substantia nigra accumulate iron and aluminum in Parkinson’s disease: a LAMMA study. Brain Res 593(2):343–346
Walton JR (2007) An aluminum-based rat model for Alzheimer’s disease exhibits oxidative damage, inhibition of PP2A activity, hyperphosphorylated tau, and granulovacuolar degeneration. J Inorg Biochem 101(9):1275–1284
Darbre PD (2005) Aluminium, antiperspirants and breast cancer. J Inorg Biochem 99(9):1912–1919
Alvim MN, Ramos FT, Oliveira DC, Isaias RM, Franca MG (2012) Aluminium localization and toxicity symptoms related to root growth inhibition in rice (Oryza sativa L.) seedlings. J Biosci 37(6):1079–1088
Poléo ABS (1995) Aluminium polymerization — A mechanism of acute toxicity of aqueous aluminium to fish. Aquat Toxicol 31(4):347–356
Liu X, Theil EC (2005) Ferritins: Dynamic management of biological iron and oxygen chemistry. Acc Chem Res 38(3):167–175
Kustka A, Carpenter EJ, Sañudo-Wilhelmy SA (2002) Iron and marine nitrogen fixation: progress and future directions. Res Microbiol 153(5):255–262
Wójciak RW, Mojs E, Stanislawska-Kubiak M (2014) The occurrence of iron-deficiency anemia in children with type 1 diabetes. J Investig Med 62(6):865–867
Simcox JA, McClain DA (2013) Iron and diabetes risk. Cell Metab 17(3):329–341
Weinreb O, Mandel S, Youdim MBH, Amit T (2013) Targeting dysregulation of brain iron homeostasis in Parkinson’s disease by iron chelators. Free Radic Biol Med 62:52–64
Torti SV, Torti FM (2013) Iron and cancer: more ore to be mined. Nat Rev Cancer 13(5):342–355
Unsal YE, Soylak M, Tuzen M, Hazer B (2015) Determination of lead, copper, and iron in cosmetics, water, soil, and food using polyhydroxybutyrate-B-polydimethyl siloxane preconcentration and flame atomic absorption spectrometry. Anal Lett 48(7):1163–1179
Ulusoy Hİ, Gürkan R, Aksoy Ü, Akçay M (2011) Development of a cloud point extraction and preconcentration method for determination of trace aluminum in mineral waters by FAAS. Microchem J 99(1):76–81
Sooriyaarachchi M, Gailer J (2010) Removal of Fe3+ and Zn2+ from plasma metalloproteins by iron chelating therapeutics depicted with SEC-ICP-AES. Dalton Trans 39(32):7466–7473
Frankowski M, Zioła-Frankowska A, Kurzyca I, Novotný K, Vaculovič T, Kanický V, Siepak M, Siepak J (2011) Determination of aluminium in groundwater samples by GF-AAS, ICP-AES, ICP-MS and modelling of inorganic aluminium complexes. Environ Monit Assess 182(1–4):71–84
Das S, Dutta M, Das D (2013) Fluorescent probes for selective determination of trace level Al3+: Recent developments and future prospects. Anal Methods 5(22):6262–6285
Sahoo SK, Sharma D, Bera RK, Crisponi G, Callan JF (2012) Iron(III) selective molecular and supramolecular fluorescent probes. Chem Soc Rev 41(21):7195–7227
Yi C, Song B, Tian W, Cui X, Qi Q, Jiang W, Qi Z, Sun Y (2014) Fluorescent sensor of fluorene derivatives having phosphonic acid as a fluorogenic ionophore: Synthesis and static quenched properties for Fe(III). Tetrahedron Lett 55(37):5119–5123
Zhang G, Lu B, Wen Y, Lu L, Xu J (2012) Facile fabrication of a cost-effective, water-soluble, and electrosynthesized poly(9-aminofluorene) fluorescent sensor for the selective and sensitive detection of Fe(III) and inorganic phosphates. Sensors Actuators B Chem 171–172:786–794
Huang L, Hou F, Cheng J, Xi P, Chen F, Bai D, Zeng Z (2012) Selective off-on fluorescent chemosensor for detection of Fe3+ ions in aqueous media. Org Biomol Chem 10(48):9634–9638
Jiménez-Sánchez A, Ortiz B, Navarrete VO, Flores JC, Farfán N, Santillan R (2015) A dual-model fluorescent Zn2+/Cu2+ ions sensor with in-situ detection of S2−/(PO4)− and colorimetric detection of Fe2+ ion. Inorg Chim Acta 429:243–251
Santhoshkumar S, Velmurugan K, Prabhu J, Radhakrishnan G, Nandhakumar R (2016) A naphthalene derived Schiff base as a selective fluorescent probe for Fe2+. Inorg Chim Acta 439:1–7
Singh G, Singh J, Mangat SS (2015) Design of selective 8-methylquinolinol based ratiometric Fe2+ and Fe3+/H2PO4 − fluorescent chemosensor mimicking NOR and IMPLICATION logic gates. J Lumin 165:123–129
Kim KB, Kim H, Song EJ, Kim S, Noh I, Kim C (2013) A cap-type Schiff base acting as a fluorescence sensor for zinc(II) and a colorimetric sensor for iron(II), copper(II), and zinc(II) in aqueous media. Dalton Trans 42(47):16569–16577
Li CR, Liao ZC, Qin JC, Wang BD, Yang ZY (2015) Study on 2-acetylpyrazine (pyridine-2′-acetyl) Hydrazone as a fluorescent sensor for Al3+. J Lumin 168:330–333
Chen CH, Liao DJ, Wan CF, Wu AT (2013) A turn-on and reversible Schiff base fluorescence sensor for Al3+ ion. Analyst 138(9):2527–2530
Gou C, Qin SH, Wu HQ, Wang Y, Luo J, Liu XY (2011) A highly selective chemosensor for Cu2+ and Al3+ in two different ways based on salicylaldehyde Schiff. Inorg Chem Commun 14(10):1622–1625
Wang G-q, Qin J-c, Li C-R, Yang Z-y (2015) A highly selective fluorescent probe for Al3+ based on quinoline derivative. Spectrochim Acta A 150:21–25
An J, Li T, Wang B, Yang Z, Yan M (2014) An off-on fluorescent sensor with high selectivity and sensitivity for Fe(III). J Coord Chem 67(5):921–928
Qin JC, Cheng XY, Fang R, Wang MF, Yang ZY, Li TR, Li Y (2016) Two Schiff-base fluorescent sensors for selective sensing of aluminum (III): Experimental and computational studies. Spectrochim Acta A 152:352–357
Fan L, Li TR, Wang BD, Yang ZY, Liu CJ (2014) A colorimetric and turn-on fluorescent chemosensor for Al(III) based on a chromone Schiff-base. Spectrochim Acta A 118:760–764
Qin J-c, Li T-r, Wang B-d, Yang Z-y, Fan L (2014) Fluorescent sensor for selective detection of Al3+ based on quinoline–coumarin conjugate. Spectrochim Acta A 133:38–43
Sen S, Sarkar S, Chattopadhyay B, Moirangthem A, Basu A, Dhara K, Chattopadhyay P (2012) A ratiometric fluorescent chemosensor for iron: discrimination of Fe2+ and Fe3+ and living cell application. Analyst 137(14):3335–3342
Aydin Z, Wei Y, Guo M (2012) A highly selective rhodamine based turn-on optical sensor for Fe3+. Inorg Chem Commun 20:93–96
Devaraj S, Tsui YK, Chiang CY, Yen YP (2012) A new dual functional sensor: Highly selective colorimetric chemosensor for Fe3+ and fluorescent sensor for Mg2+. Spectrochim Acta A 96:594–599
Choi YW, Park GJ, Na YJ, Jo HY, Lee SA, You GR, Kim C (2014) A single schiff base molecule for recognizing multiple metal ions: A fluorescence sensor for Zn(II) and Al(III) and colorimetric sensor for Fe(II) and Fe(III). Sensors Actuators B Chem 194:343–352
Bronson RT, Bradshaw JS, Savage PB, Fuangswasdi S, Lee SC, Krakowiak KE, Izatt RM (2001) Bis-8-hydroxyquinoline-armed diazatrithia-15-crown-5 and diazatrithia-16-crown-5 ligands: Possible fluorophoric metal ion sensors. J Organomet Chem 66(14):4752–4758
Farruggia G, Iotti S, Prodi L, Montalti M, Zaccheroni N, Savage PB, Trapani V, Sale P, Wolf FI (2006) 8-Hydroxyquinoline derivatives as fluorescent sensors for magnesium in living cells. J Am Chem Soc 128(1):344–350
Song EJ, Park GJ, Lee JJ, Lee S, Noh I, Kim Y, Kim S-J, Kim C, Harrison RG (2015) A fluorescence sensor for Zn2+ that also acts as a visible sensor for Co2+ and Cu2+. Sensors Actuators B Chem 213:268–275
Vaswani KG, Keränen MD (2009) Detection of aqueous mercuric ion with a structurally simple 8-hydroxyquinoline derived on-off fluorosensor. Inorg Chem 48(13):5797–5800
Zhao Y, Lin Z, Liao H, Duan C, Meng Q (2006) A highly selective fluorescent chemosensor for Al3+ derivated from 8-hydroxyquinoline. Inorg Chem Commun 9(9):966–968
Jiang XH, Wang BD, Yang ZY, Liu YC, Li TR, Liu ZC (2011) 8-Hydroxyquinoline-5-carbaldehyde Schiff-base as a highly selective and sensitive Al3+ sensor in weak acid aqueous medium. Inorg Chem Commun 14(8):1224–1227
Fan L, Jiang XH, Wang BD, Yang ZY (2014) 4-(8′-Hydroxyquinolin-7′-yl)methyleneimino-1-phenyl-2,3-dimethyl-5-pyzole as a fluorescent chemosensor for aluminum ion in acid aqueous medium. Sensors Actuators B Chem 205:249–254
Ramos ML, Justino LLG, Salvador AIN, De Sousa ARE, Abreu PE, Fonseca SM, Burrows HD (2012) NMR, DFT and luminescence studies of the complexation of Al(III) With 8-hydroxyquinoline-5-sulfonate. Dalton Trans 41(40):12478–12489
Pierre J-L, Baret P, Serratrice G (2003) Hydroxyquinolines as iron chelators. Curr Med Chem 10:1077–1084
Li Z, Li H, Shi C, Zhang W, Zhou W, Wei L, Yu M (2016) Naked-eye-based highly selective sensing of Fe3+ and further for PPi with nano copolymer film. Sensors Actuators B Chem 226:127–134
Zarabadi-Poor P, Badiei A, Yousefi AA, Barroso-Flores J (2013) Selective optical sensing of Hg(II) in aqueous media by H-Acid/SBA-15: A combined experimental and theoretical study. J Phys Chem C 117(18):9281–9289
Karimi M, Badiei A, Mohammadi Ziarani G (2015) A novel naphthalene-immobilized nanoporous SBA-15 as a highly selective optical sensor for detection of Fe3+ in water. J Fluoresc 25(5):1297–1302
Afshani J, Badiei A, Karimi M, Lashgari N, Mohammadi Ziarani G (2016) A single fluorescent sensor for Hg2+ and discriminately detection of Cr3+ and Cr(VI). J Fluoresc 26:263–270
Karimi M, Badiei A, Mohammadi Ziarani G (2015) A single hybrid optical sensor based on nanoporous silica type SBA-15 for detection of Pb2+ and I− in aqueous media. RSC Adv 5(46):36530–36539
Wang F, Peng R, Sha Y (2008) Selective dendritic fluorescent sensors for Zn(II). Molecules 13(4):922–930
Maity D, Govindaraju T (2010) Conformationally constrained (coumarin − triazolyl − bipyridyl) click fluoroionophore as a selective Al3+ sensor. Inorg Chem 49(16):7229–7231
Kim S, Noh JY, Kim KY, Kim JH, Kang HK, Nam SW, Kim SH, Park S, Kim C, Kim J (2012) Salicylimine-based fluorescent chemosensor for aluminum ions and application to bioimaging. Inorg Chem 51(6):3597–3602
Qin JC, Yang ZY (2015) Selective fluorescent sensor for Al3+ using a novel quinoline derivative in aqueous solution. Synth Metals 209:570–576
Bardez E, Devol I, Larrey B, Valeur B (1997) Excited-state processes in 8-hydroxyquinoline: Photoinduced tautomerization and solvation effects. J Phys Chem B 101(39):7786–7793
Huston ME, Haider KW, Czarnik AW (1988) Chelation enhanced fluorescence in 9,10-bis[[(2-(dimethylamino)ethyl)methylamino]methyl]anthracene. J Am Chem Soc 110(13):4460–4462
Bronson RT, Montalti M, Prodi L, Zaccheroni N, Lamb RD, Dalley NK, Izatt RM, Bradshaw JS, Savage PB (2004) Origins of ‘on–off’ fluorescent behavior of 8-hydroxyquinoline containing chemosensors. Tetrahedron 60(49):11139–11144
Erdemir S, Kocyigit O, Karakurt S (2015) A new perylene bisimide-armed calix[4]-aza-crown as "turn on" fluorescent sensor for Hg2+ ion and its application to living cells. Sensors Actuators B Chem 220:381–388
Afshani J, Badiei A, Lashgari N, Mohammadi Ziarani G (2016) A simple nanoporous silica-based dual mode optical sensor for detection of multiple analytes (Fe3+, Al3+ and CN−) in water mimicking XOR logic gate. RSC Adv 6(7):5957–5964
Meier MAR, Schubert US (2005) Fluorescent sensing of transition metal ions based on the encapsulation of dithranol in a polymeric core shell architecture. Chem Commun. doi:10.1039/b505409e
Hao E, Meng T, Zhang M, Pang W, Zhou Y, Jiao L (2011) Solvent dependent fluorescent properties of a 1,2,3-triazole linked 8-hydroxyquinoline chemosensor: tunable detection from zinc(II) to iron(III) in the CH3CN/H2O system. J Phys Chem A 115(29):8234–8241
Acknowledgments
The authors thank the research council of University of Tehran for financial support.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
ESM 1
(DOCX 742 kb)
Rights and permissions
About this article
Cite this article
Lashgari, N., Badiei, A. & Mohammadi Ziarani, G. A Fluorescent Sensor for Al(III) and Colorimetric Sensor for Fe(III) and Fe(II) Based on a Novel 8-Hydroxyquinoline Derivative. J Fluoresc 26, 1885–1894 (2016). https://doi.org/10.1007/s10895-016-1883-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1883-3