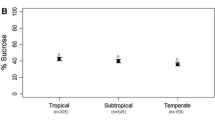
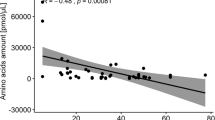
Abstract
High-pressure liquid chromatography analyses of 73 plant species showed that the nectars of phrygana (East Mediterranean garrigue) mainly contain sucrose, glucose, and fructose, and traces of 10 minor sugars. Although the sucrose/hexose ratio was not related to plant life habit, ecological constraints had a detectable effect in shaping sugar composition. This was detected by distinguishing the phryganic plant species into “spring–summer” and “winter” flowering, with the distinction made on the basis of the water deficit in the study area. Plants flowering in spring–summer had a higher rate of “high sucrose” (i.e., sucrose/hexose ratio ≥0.5; 60.8% of the plant species) vs. “low hexose” nectars (i.e., ratio <0.5; 39.2%). The ratio was reversed in winter flowering species (36.4% vs. 63.6% with “high sucrose” and “high hexose,” respectively). Sucrose/hexose ratios were associated with plant family. The highest values were those of Lamiaceae, which differed significantly from the “low sucrose” Liliaceae and Apiaceae. Based on recorded plant–pollinator interactions in the community, the present data provide evidence of a partitioning of nectar resources by the existing pollinator guilds within the community, based on the sugar profiles of nectar (all sucrose/hexose ratios for all interactions). Among all major groups, bees and wasps (aculeates) preferred “high sucrose” nectars, which differed significantly from syrphids, anthomyid a.o. flies, and beetles that visited “low sucrose” nectars. Similarly, butterflies visited “lower sucrose” nectars compared to bees. Within families, only Megachilidae could be clearly characterized as “high sucrose” consumers, differing in this respect from all the remaining insect groups including most other bee families. This confirms previous findings that Megachilidae have a key position in Mediterranean communities where they probably constitute a selective factor for “high sucrose” nectars.

Similar content being viewed by others
References
H. G. Baker I. Baker (1975) Studies of nectar constitution and pollinator–plant coevolution L. E. Gilbert P. H. Raven (Eds) Coevolution of Animals and Plants University of Texas Press Austin 100–140
H. G. Baker I. Baker (1979) ArticleTitleSugar ratios in nectars Phytochem. Bull. 12 43–45
H. G. Baker I. Baker (1982) Chemical constituents of nectar in relation to pollination mechanisms and phylogeny M. H. Nitecki (Eds) Biochemical Aspects of Evolutionary Biology—Proceedings of the 4th Annual Spring Systematics Symposium University of Chicago Press Chicago 131–172
H. G. Baker I. Baker (1983) Floral nectar sugar constituents in relation to pollinator type C. E. Jones R. J. Little (Eds) Handbook of Experimental Pollination Ecology Van Nostrand-Reinhold New York 117–141
H. G. Baker I. Baker (1986) ArticleTitleThe occurrence and significance of amino acids in floral nectar Plant Syst. Evol. 151 175–186
H. G. Baker I. Baker (1990) ArticleTitleThe predictive value of nectar chemistry to the recognition of pollinator types Isr. J. Botan. 39 157–166
J. C. Biesmeijer J. A. P. Richter M. A. J. P. Smets M. J. Sommeijer (1999) ArticleTitleNiche differentiation in nectar-collecting stingless bees: The influence of morphology, floral choice and interference competition Ecol. Entomol. 24 380–388
S. A. Corbet P. G. Willmer J. W. L. Beament D. M. Unwin O. E. Prys-Jones (1979) ArticleTitlePost-secretory determinants of sugar concentration in nectar Plant Cell Environ. 2 293–308
H. Dafni Y. Lensky A. Fahn (1988) ArticleTitleFlower and nectar characteristics of nine species of Labiatae and their influence on honeybee visits J. Apic. Res. 27 103–114
M. E. Dodd J. Silvertown M. W. Chase (1999) ArticleTitlePhylogenetic analysis of trait evolution and species diversity variation among angiosperm families Evolution 53 732–744
G. Gottsberger J. Schrauwen H. F. Linskens (1984) ArticleTitleAmino acids and sugars in nectar, and their putative evolutionary significance Plant Syst. Evol. 145 55–77
J. Herrera (1985) ArticleTitleNectar secretion patterns in southern Spanish Mediterranean scrublands Isr. J. Botan. 34 47–58
J. P. Kochmer S. N. Handel (1986) ArticleTitleConstraints and competition in the evolution of flowering phenology Ecol. Monogr. 56 303–325
C. D. Michener (1979) ArticleTitleBiogeography of the bees Ann. Mo. Bot. Gard. 66 277–347
C. D. Michener (2000) The Bees of the World The Johns Hopkins Univ. Press Baltimore
S. W. Nicolson (1998) ArticleTitleThe importance of osmosis in nectar secretion and its consumption by insects Am. Zool. 38 418–425
S. W. Nicolson (2002) ArticleTitlePollination by passerine birds: Why are the nectars so dilute? Comp. Biochem. Phys., B. 131 645–652
S. W. Nicolson B. E. Wyk ParticleVan (1998) ArticleTitleNectar sugars in Proteaceae: Patterns and processes Aust. J. Bot. 46 489–504
M. Pagano K. Gauvreau (1993) Principles of Biostatistics Duxbury Press Belmont, CA
J. S. Pate M. B. Peoples P. J. Storer C. A. Atkins (1985) ArticleTitleThe extrafloral nectaries of cowpea (Vigna unguiculata (L.) Walp.). II. Nectar composition, origin of nectar solutes, and nectary functioning Planta 166 28–38
M. S. Percival (1961) ArticleTitleTypes of nectar in angiosperms New Phytol. 60 235–281
M. S. Percival (1965) Floral Biology Pergamon Oxford
Petanidou, T. 1991. Pollination ecology in a phryganic ecosystem. Ph.D. thesis (in Greek, with English summary). Aristotle University, Thessaloniki.
T. Petanidou (1996) ArticleTitleLabiatae: A key family for wild bees and the pollination ecology in Mediterranean phryganic communities Lamiales Newsl. 4 4–6
T. Petanidou W. N. Ellis (1993) ArticleTitlePollinating fauna of a phryganic ecosystem: Composition and diversity Biodiversity Lett. 1 9–22
T. Petanidou W. N. Ellis (1996) Interdependence of native bee faunas and floras in changing Mediterranean communities A. Matheson S. L. Buchmann C. O’Toole P. Westrich I. H. Williams (Eds) The Conservation Biology of Bees. Linnean Society Symposium Series No. 18 Academic Press London 201–226
Petanidou, T. and Potts, S. G. 2005. Mutual use of resources in Mediterranean plant–pollinator communities: How specialised are pollination webs?, in N. Waser and J. Ollerton (eds.). Specialization and Generalization in Plant–Pollinator Interactions. University of Chicago Press, in press.
T. Petanidou E. Smets (1995) ArticleTitleThe potential of marginal lands for bees and apiculture: Nectar secretion in Mediterranean shrublands Apidologie 26 39–52
T. Petanidou D. Vokou (1990) ArticleTitlePollination and pollen energetics in Mediterranean ecosystems Am. J. Bot. 77 986–992
T. Petanidou D. Vokou (1993) ArticleTitlePollination ecology of Labiatae in a phryganic (East Mediterranean) ecosystem Am. J. Bot. 80 892–899
T. Petanidou W. N. Ellis N. S. Margaris D. Vokou (1995) ArticleTitleConstraints on flowering phenology in a phryganic (East Mediterranean shrub) community Am. J. Bot. 82 607–620
T. Petanidou A. Laere ParticleVan E. Smets (1996) ArticleTitleChange in floral nectar components from fresh to senescent flowers of Capparis spinosa L., a nocturnally flowering Mediterranean shrub Plant Syst. Evol. 199 79–92
T. Petanidou V. Goethals E. Smets (2000) ArticleTitleNectary structure of Labiatae in relation to their nectar secretion and characteristics in a Mediterranean shrub community—Does flowering time matter? Plant Syst. Evol. 225 103–118
G. H. Pyke N. M. Waser (1981) ArticleTitleOn the production of dilute nectars by hummingbird and honeyeater flowers Biotropica 13 260–270
H. P. Rusterholz A. Erhardt (1997) ArticleTitlePreferences for nectar sugars in the peacock butterfly, Inachis io Ecol. Entomol. 22 220–224
E. E. Southwick (1982) Nectar biology and pollinator attraction in the north temperate climate M. C. D. Breed C. Michener H. E. Evans (Eds) The Biology of Social Insects Westview Press Boulder 19–23
F. G. Stiles C. E. Freeman (1993) ArticleTitlePatterns in floral nectar characteristics of some bird-visited plant species from Costa Rica Biotropica 25 191–205
The International Plant Names Index. 2004. http://www.ipni.org.
Waser, N. and Ollerton, J. 2005. Specialization and Generalization in Plant–Pollinator Interactions. University of Chicago Press, in press.
H. Wells P. S. Hill P. H. Wells (1992) ArticleTitleNectarivore foraging ecology—Rewards differing in sugar types Ecol. Entomol. 17 280–288
G. R. Wykes (1952) ArticleTitleAn investigation of the sugars present in the nectar of flowers of various species New Phytol. 51 210–215
Acknowledgments
The study was supported by an EC grant (DG VI-contract AIR3-BM92-004). I thank Prof. E. Smets and Prof. A. Van Laere for offering laboratory facilities at the Catholic University of Leuven; E. Nackaerts and V. Goethals for technical assistance on sugar analyses; Prof. Sue Nicolson, Prof. Peter J. Davies, Dr. Ellen Lamborn and an anonymous reviewer for invaluable comments on earlier versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petanidou, T. Sugars in Mediterranean Floral Nectars: An Ecological and Evolutionary Approach. J Chem Ecol 31, 1065–1088 (2005). https://doi.org/10.1007/s10886-005-4248-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-005-4248-y