Abstract
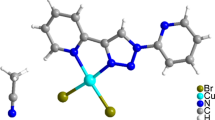
Three new nickel clusters [Ni3(hmpm)4(DMF)2(X)2]·Y (1, X = HCOO−, Y = 0; 2, X = CH3COO−; Y = C5H8; 3, X = F3CCOO−, Y = 0.5H2O, Hhmpm = (2-hydroxy-4-methoxy-phenyl)-phenyl-methanone) have been prepared via solvothermal method. They were characterized by elemental analysis, IR, UV–Vis, fluorescence and X-ray single-crystal diffraction. All three compounds are defined as trinuclear clusters with three nickel atoms linked in line. Herein, we have used three different carboxylate radicals. The results indicate that the steric hindrance effects in carboxylate radicals do not influence the structures of the clusters. The magnetic investigation shows that complexes 1–3 exhibit a ferromagnetic coupling between NiII ions.







Similar content being viewed by others
References
M. Fujita, A. Powell, and C. Creutz From the Molecular to the Nanoscale: Synthesis, Structure and Properties, vol. 7 (Elsevier, Oxford, 2004).
L. Bogani and W. Wernsdorfer (2008). Nat. Mater. 7, 179.
C. J. Milios, R. Inglis, A. Vinslava, R. Bagai, W. Wernsdorfer, S. Parsons, S. P. Perlepes, G. Christou, and E. K. Brechin (2007). J. Am. Chem. Soc. 129, 12505.
D. Gatteschi and R. Sessoli (2003). Angew. Chem. Int. Ed. 42, 268.
A. M. Ako, V. Mereacre, Y. H. Lan, W. Wernsdorfer, R. Clérac, C. E. Anson, and A. K. Powell (2010). Inorg. Chem. 49, 1.
G. Aromı, S. Parsons, W. Wernsdorfer, E. K. Brechin, and E. J. L. McInnes (2005). Chem. Commun. 5038.
P. J. Hagrman, D. Hagrman, and J. Zubieta (1999). Angew. Chem. Int. Ed. 38, 2638.
T. Taguchi, W. Wernsdorfer, K. A. Abboud, and G. Christou (2010). Inorg. Chem. 49, 199.
M. T. Gamer, Y. H. Lan, P. W. Roesky, A. K. Powell, and R. Clérac (2008). Inorg. Chem. 47, 6581.
P. Alborés and E. Rentschler (2009). Angew. Chem. Int. Ed. 48, 9366.
L. Yang, Q. P. Huang, C. L. Zhang, R. X. Zhao, and S. H. Zhang (2014). Supramol. Chem. 26, 81.
S. H. Zhang, M. F. Tang, and C. M. Ge (2009). Z. Anorg. Allg. Chem. 635, 1442.
W. Wang, H. Hai, S. H. Zhang, L. Yang, and C. L. Zhang (2014). J. Cluster Sci. 25, 357.
S.-H. Zhang, N. Li, C. M. Ge, C. Feng, and L. F. Ma (2011). Dalton Trans. 40, 3000.
L. F. Ma, L. Y. Wang, X. K. Huo, Y. Y. Wang, Y. T. Fan, J. G. Wang, and S. H. Chen (2008). Cryst. Grow. Des. 8, 620.
S. H. Zhang, Y. L. Zhou, X. J. Sun, L. Q. Wei, M. H. Zeng, and H. Liang (2009). J. Solid State Chem. 182, 2991.
M. Yoneya, T. Yamaguchi, S. Sato, and M. Fujita (2012). J. Am. Chem. Soc. 134, 14401.
D. Fujita, K. Suzuki, S. Sato, M. Yagi-Utsumi, Y. Yamaguchi, N. Mizuno, T. Kumasaka, M. Takata, M. Noda, S. Uchiyama, K. Kato, and M. Fujita (2012). Nat. Commun. 3, 1093.
S. Ulrich, A. Petitjean, and J. M. Lehn (2010). Eur. J. Inorg. Chem. 1913.
S. M. Biros, R. M. Yeh, and K. N. Raymond (2008). Angew. Chem. Int. Ed. 47, 6062.
C. J. Brown, G. M. Miller, M. W. Johnson, R. G. Bergman, and K. N. Raymond (2011). J. Am. Chem. Soc. 133, 11964.
R. E. P. Winpenny (2002). J. Chem. Soc., Dalton Trans. 1.
G. Aromí, A. R. Bell, M. Helliwell, J. Raftery, S. J. Teat, G. A. Timco, O. Roubeau, and R. E. P. Winpenny (2003). Chem. Eur. J. 9, 3024.
P. Mukherjee and S. Mukherjee (2013). Acc. Chem. Res. 46, 2556.
S. H. Zhang, Y. D. Zhang, H. H. Zou, J. J. Guo, H. P. Li, Y. Song, and H. Liang (2013). Inorg. Chim. Acta. 396, 119.
G. M. Sheldrick (2008). Acta Cryst. A64, 112.
A mixture of Ni(HCOOH)2·2H2O (0.184 g, 1 mmol), Hhmpm (0.228 g, 1 mmol), H2O (8 mL) with a pH adjusted to 7.5 by addition of triethylamine was poured into a Teflon-lined autoclave (15 mL) and then heated at 140°C for 3 days. Green crystals of a were collected by filtration, washed with water and dried in air. Phase pure crystals of a were obtained by manual separation (yield: 161.6 mg, ca. 63.1 % based on Hhmpm ligand). Crystal data for the mononuclear nickel complex a: C28H22NiO6 (Ni(hmpm)2), Mr = 1026.33 g∙mol−1, monoclinic, P21/c, a = 11.267(2), b = 10.496(2), c = 20.600(3) Å, β = 98.030(2)°, V = 2412.1(7) Å3, θ = 25.01°, λ = 0.71073 Å, T = 296(2) K, μ(Mo Kα) = 0.846 mm−1. 3339 reflections were collected of which 4254 were unique (Rint = 0.0303). The structure was solved by direct methods and refined by full-matrix least squares of F 2, R 1 = 0.1789. Max/min residual electron density 0.902/–0.998 e Å3 and the structure of (a) see Figure S1.
Y. Xiao, S. H. Zhang, G. Z. Li, Y. G. Wang, and C. Feng (2011). Inorg. Chim. Acta. 366, 39.
K. O. Kongshaug and H. Fjellvåg (2003). Solid State Sci. 5, 303.
T. C. Higgs and C. J. Carrano (1997). Inorg. Chem. 36, 298.
J. Cano, G. D. Munno, F. Lloret, and M. Julve (2000). Inorg. Chem. 39, 1611.
R. Biswas, S. Mukherjee, P. Kar, and A. Ghosh (2012). Inorg. Chem. 51, 8150.
M. Shebl (2014). Spectrochim. Acta A 117, 127.
H. Naeimi and M. Moradian (2010). J. Coord. Chem. 63, 156.
M. Shebl (2009). Spectrochim. Acta A 73, 313.
K. Nakamoto Infrared and Raman Spectra of Inorganic and Coordination Compounds, 5th ed (Wiley, New York, 1997).
S. Mishra, S. Daniele, G. Ledoux, E. Jeanneau, and M. F. Joubert (2010). Chem. Commun. 46, 3756.
L. E. Valenti, M. B. Paci, C. P. D. Pauli, and C. E. Giacomelli (2011). Anal. Biochem. 410, 118.
S. Basak, S. Sen, S. Banerjee, S. Mitra, G. Rosair, and M. T. G. Rodriguez (2007). Polyhedron 26, 5104.
B. Shafaatian, A. Soleymanpour, N. Kholghi Oskouei, B. Notash, and S. A. Rezvani (2014). Spectrochim. Acta A 128, 363.
Y. Chen, X.-J. Zhao, X. Gan, and W.-F. Fu (2008). Inorg. Chim. Acta 361, 2335.
D. Das, B. G. Chand, K. K. Sarker, J. Dinda, and C. Sinha (2006). Polyhedron 25, 2333.
Acknowledgments
This work is financially supported by the National Natural Science Foundation of China (No. 21161006), and Program for Excellent Talents in Guangxi Higher Education Institutions (Gui Jiao Ren [2012]41).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Weng, Z., Zhang, SH., Wang, W. et al. Syntheses, Structures and Properties of Three New Trinuclear Nickel Clusters with (2-Hydroxy-4-methoxyphenyl)-phenyl-methanone. J Clust Sci 26, 1129–1142 (2015). https://doi.org/10.1007/s10876-014-0802-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-014-0802-5