Abstract
Purpose
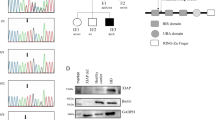
X-linked inhibitor of apoptosis (XIAP) deficiency caused by mutations in BIRC4 was originally described in male patients with X-linked lymphoproliferative syndrome type 2 (XLP2). Recent observations have highlighted a critical role of XIAP for the regulation of NOD2 signaling and are probably the molecular basis for increasingly recognized further immune dysregulatory symptoms of XIAP deficient patients, such as inflammatory bowel disease (IBD). We describe a large Caucasian family in which IBD and erythema nodosum (EN) also manifested in female carriers of XIAP mutations.
Methods
Clinical data and laboratory findings including flow cytometric analysis of XIAP protein expression and sequencing of the BIRC4 gene. NOD2 signaling was investigated by determination of TNFα production in monocytes upon L18-MDP stimulation in vitro.
Results
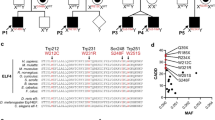
The BIRC4 nonsense mutation p.P225SfsX226 was identified as the genetic cause of XIAP deficiency in our family. Surprisingly, clinical symptoms were not restricted to male patients, but also occurred in several female carriers. The most severely affected carrier demonstrated random X-inactivation, leading to a significant expression of mutated XIAP protein in monocytes, and consequently to impaired NOD2 responses in vitro.
Conclusion
Our report provides further evidence that clinical symptoms of XIAP deficiency are not restricted to male patients. Random X-inactivation may be associated with EN and mild IBD also in female carriers of BIRC4 mutations. Analysis of the X-inactivation pattern reflected by XIAP protein expression can identify such carriers and the analysis of NOD2 signaling by flow cytometry can confirm the functional significance. XIAP expression patterns should be investigated in female patients with a family history of EN and/or IBD.


Similar content being viewed by others
References
Uhlig HH. Monogenic diseases associated with intestinal inflammation: implications for the understanding of inflammatory bowel disease. Gut. 2013;62(12):1795–805. doi:10.1136/gutjnl-2012-303956.
Rigaud S, Fondaneche MC, Lambert N, Pasquier B, Mateo V, Soulas P, et al. XIAP deficiency in humans causes an X-linked lymphoproliferative syndrome. Nature. 2006;444(7115):110–4. doi:10.1038/nature05257.
Pachlopnik Schmid J, Canioni D, Moshous D, Touzot F, Mahlaoui N, Hauck F, et al. Clinical similarities and differences of patients with X-linked lymphoproliferative syndrome type 1 (XLP-1/SAP deficiency) versus type 2 (XLP-2/XIAP deficiency). Blood. 2011;117(5):1522–9. doi:10.1182/blood-2010-07-298372.
Speckmann, Ehl S. XIAP deficiency is a mendelian cause of late-onset IBD. Gut. 2013:gutjnl-2013-306474. doi:10.1136/gutjnl-2013-306474
Worthey EA, Mayer AN, Syverson GD, Helbling D, Bonacci BB, Decker B, et al. Making a definitive diagnosis: successful clinical application of whole exome sequencing in a child with intractable inflammatory bowel disease. Genet Med. 2011;13(3):255–62. doi:10.1097/GIM.0b013e3182088158.
Yang X, Kanegane H, Nishida N, Imamura T, Hamamoto K, Miyashita R, et al. Clinical and genetic characteristics of XIAP deficiency in Japan. J Clin Immunol. 2012;32(3):411–20. doi:10.1007/s10875-011-9638-z.
Zeissig Y, Petersen B-S, Milutinovic S, Bosse E, Mayr G, Peuker K et al. XIAP variants in male Crohn's disease. Gut. 2014:gutjnl-2013-306520. doi:10.1136/gutjnl-2013-306520.
Aguilar C, Lenoir C, Lambert N, Bègue B, Brousse N, Canioni D, et al. Characterization of Crohn disease in X-linked inhibitor of apoptosis-deficient male patients and female symptomatic carriers. J Allergy Clin Immunol. 2014. doi:10.1016/j.jaci.2014.04.031.
Speckmann C, Lehmberg K, Albert MH, Damgaard RB, Fritsch M, Gyrd-Hansen M, et al. X-linked inhibitor of apoptosis (XIAP) deficiency: the spectrum of presenting manifestations beyond hemophagocytic lymphohistiocytosis. Clin Immunol (Orlando Fla). 2013;149(1):133–41. doi:10.1016/j.clim.2013.07.004.
Marsh RA, Rao K, Satwani P, Lehmberg K, Muller I, Li D, et al. Allogeneic hematopoietic cell transplantation for XIAP deficiency: an international survey reveals poor outcomes. Blood. 2013;121(6):877–83. doi:10.1182/blood-2012-06-432500.
Damgaard RB, Fiil BK, Speckmann C, Yabal M, Stadt UZ, Bekker-Jensen S, et al. Disease-causing mutations in the XIAP BIR2 domain impair NOD2-dependent immune signalling. EMBO Mol Med. 2013;5(8):1278–95. doi:10.1002/emmm.201303090.
Ammann S, Elling R, Gyrd-Hansen M, Dückers G, Bredius R, Burns SO et al. A new functional assay for the diagnosis of X-linked inhibitor of apoptosis (XIAP) deficiency. Clinical and experimental immunology. 2014:n/a-n/a. doi:10.1111/cei.12306.
Marsh RA, Madden L, Kitchen BJ, Mody R, McClimon B, Jordan MB, et al. XIAP deficiency: a unique primary immunodeficiency best classified as X-linked familial hemophagocytic lymphohistiocytosis and not as X-linked lymphoproliferative disease. Blood. 2010;116(7):1079–82. doi:10.1182/blood-2010-01-256099.
Yabal M, Müller N, Adler H, Knies N, Groß CJ, Damgaard RB, et al. XIAP Restricts TNF- and RIP3-Dependent Cell Death and Inflammasome Activation. Cell Rep. 2014. doi:10.1016/j.celrep.2014.05.008.
Marsh RA, Bleesing JJ, Filipovich AH. Flow cytometric measurement of SLAM-associated protein and X-linked inhibitor of apoptosis. Methods Mol Biol. 2013;979:189–97. doi:10.1007/978-1-62703-290-2_15.
Acknowledgments
Prof. Stephan Ehl from the CCI in Freiburg, who pointed us towards the possible involvement of XIAP in this family. We sincerely thank all our patients and their family members who made this study possible and the referring consultants (Dr Claire Dolling and Dr Yusuf Karim) for their help. We also thank the technicians of the Center of Chronic Immunodeficiency Advanced Diagnostic for excellent technical assistance.
Funding
This study was supported by the German Federal Ministry of Education and Research (BMBF 01 EO 0803 and and BMBF 01GM1111B). The authors are responsible for the contents of this publication.
Financial Disclosure
There is no financial disclosure.
Conflict of Interest
The authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Magdalena Dziadzio, Sandra Ammann, Carsten Speckmann and Bodo Grimbacher contributed equally and are considered aequo loco.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Colonic biopsies from index patient V.24 A) Pre-BMT: H&E staining of the large bowel mucosa with preservation of crypt architecture. There is patchy active inflammation and focal increase in mixed inflammatory cells in the lamina propria; no granulomas. The appearance is consistent with mild focal active colitis. B): Post-BMT: H&E staining of the large bowel mucosa showing hypocellularity of the lamina propria and some mild distortion of the crypt architecture. There is no evidence of GVHD and no viral inclusions. (JPEG 115 kb)
Table S1
Summary of the phenotypes of patients and carriers. (DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Dziadzio, M., Ammann, S., Canning, C. et al. Symptomatic Males and Female Carriers in a Large Caucasian Kindred with XIAP Deficiency. J Clin Immunol 35, 439–444 (2015). https://doi.org/10.1007/s10875-015-0166-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-015-0166-0