Abstract
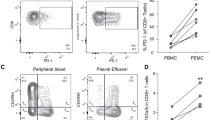
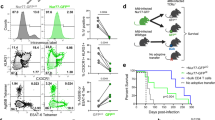
Natural killer T (NKT) cells from mouse and human play a protective role in the immune responses against the infection of Mycobacterium tuberculosis. However, the characteristic of CD3+TCRvβ11+ NKT cells at the local site of M. tuberculosis infection remains poorly defined. In the present study, we found that the numbers of CD3+TCRvβ11+ NKT cells in pleural fluid mononuclear cells (PFMCs) were significantly lower than those in peripheral blood mononuclear cells (PBMCs). However, CD3+TCRvβ11+ NKT cells from PFMCs spontaneously expressed high levels of CD69 and CD25 and effector memory phenotypes of CD45ROhighCD62LlowCCR7low. After stimulation with the antigens of M. tuberculosis, CD3+TCRvβ11+ NKT cells from PFMCs produced high levels of IFN-γ. Sorted CD3+TCRvβ11+ NKT cells from PFMCs cultured with antigen presenting cells (APCs) produced IFN-γ protein and mRNA. The production of IFN-γ could be completely inhibited by AG490 and Wortmannin. In addition, CD3+TCRvβ11+ NKT cells from PFMCs expressed higher levels of Fas (CD95), FasL (CD178) and perforin but lower levels of granzyme B compared with those from PBMCs. Taken together, our data demonstrated for the first time that M. tuberculosis-specific CD3+TCRvβ11+ NKT cells participated in the local immune responses against M. tuberculosis through the production of IFN-γ and the secretion of cytolytic molecules.








Similar content being viewed by others
References
World Health Organization. Global tuberculosis report. 2012.
Ahmad S. New approaches in the diagnosis and treatment of latent tuberculosis infection. Respir Res. 2010;11:169.
Dye C, Scheele S, Dolin P, Pathania V, Raviglione MC. Consensus statement. Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. WHO global surveillance and monitoring project. JAMA. 1999;282(7):677–86.
Landry J, Menzies D. Preventive chemotherapy. Where has it got us? Where to go next? Int J Tuberc Lung Dis. 2008;12(12):1352–64.
Kaufmann SH. Tuberculosis: back on the immunologists’ agenda. Immunity. 2006;24(4):351–7.
Sada-Ovalle I, Chiba A, Gonzales A, Brenner MB, Behar SM. Innate invariant NKT cells recognize mycobacterium tuberculosis-infected macrophages, produce interferon-gamma, and kill intracellular bacteria. PLoS Pathog. 2008;4(12):e1000239.
Godfrey DI, MacDonald HR, Kronenberg M, Smyth MJ, Van Kaer L. NKT cells: what’s in a name? Nat Rev Immunol. 2004;4(3):231–7.
Van Kaer L. alpha-Galactosylceramide therapy for autoimmune diseases: prospects and obstacles. Nat Rev Immunol. 2005;5(1):31–42.
Godfrey DI, Kronenberg M. Going both ways: immune regulation via CD1d-dependent NKT cells. J Clin Invest. 2004;114(10):1379–88.
Wermeling F, Lind SM, Jordö ED, Cardell SL, Karlsson MC. Invariant NKT cells limit activation of autoreactive CD1d-positive B cells. J Exp Med. 2010;207(5):943–52.
Montoya CJ, Cataño JC, Ramirez Z, Rugeles MT, Wilson SB, Landay AL. Invariant NKT cells from HIV-1 or mycobacterium tuberculosis-infected patients express an activated phenotype. Clin Immunol. 2008;127(1):1–6.
Sallusto F, Geginat J, Lanzavecchia A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol. 2004;22:745–63.
Li ZT, Yang BY, Wu CY. Differences between CD3+ TCRvα24+ NKT cell and CD3+ TCRvβ11+ NKT cell in PBMC. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2011;27(9):949–53.
Berzins SP, Smyth MJ, Baxter AG. Presumed guilty: natural killer T cell defects and human disease. Nat Rev Immunol. 2011;11(2):131–42.
Sutherland JS, Jeffries DJ, Donkor S, Walther B, Hill PC, Adetifa IM, et al. High granulocyte/lymphocyte ratio and paucity of NKT cells defines TB disease in a TB-endemic setting. Tuberculosis (Edinb). 2009;89(6):398–404.
Chackerian A, Alt J, Perera V, Behar SM. Activation of NKT cells protects mice from tuberculosis. Infect Immun. 2002;70(11):6302–9.
Gansert JL, Kiessler V, Engele M, Wittke F, Röllinghoff M, Krensky AM, et al. Human NKT cells express granulysin and exhibit antimycobacterial activity. J Immunol. 2003;170(6):3154–61.
Sada-Ovalle I, Sköld M, Tian T, Besra GS, Behar SM. Alpha-galactosylceramide as a therapeutic agent for pulmonary mycobacterium tuberculosis infection. Am J Respir Crit Care Med. 2010;182(6):841–7.
Venkataswamy MM, Baena A, Goldberg MF, Bricard G, Im JS, Chan J, et al. Incorporation of NKT cell-activating glycolipids enhances immunogenicity and vaccine efficacy of Mycobacterium bovis bacillus Calmette-Guerin. J Immunol. 2009;183(3):1644–56.
Flynn JL, Chan J, Triebold KJ, Dalton DK, Stewart TA, Bloom BR. An essential role for interferon gamma in resistance to mycobacterium tuberculosis infection. J Exp Med. 1993;178(6):2249–54.
Newport MJ, Huxley CM, Huston S, Hawrylowicz CM, Oostra BA, Williamson R, et al. A mutation in the interferon-gamma-receptor gene and susceptibility to mycobacterial infection. N Engl J Med. 1996;335(26):1941–9.
Jouanguy E, Altare F, Lamhamedi S, Revy P, Emile JF, Newport M, et al. Interferon-gamma-receptor deficiency in an infant with fatal bacille Calmette-Guérin infection. N Engl J Med. 1996;335(26):1956–61.
Jouanguy E, Lamhamedi-Cherradi S, Altare F, Fondanèche MC, Tuerlinckx D, Blanche S, et al. Partial interferon-gamma receptor 1 deficiency in a child with tuberculoid bacillus Calmette-Guérin infection and a sibling with clinical tuberculosis. J Clin Invest. 1997;100(11):2658–64.
Lee PT, Putnam A, Benlagha K, Teyton L, Gottlieb PA, Bendelac A. Testing the NKT cell hypothesis of human IDDM pathogenesis. J Clin Invest. 2002;110(6):793–800.
Chan AC, Neeson P, Leeansyah E, Tainton K, Quach H, Prince HM, et al. Testing the NKT cell hypothesis in lenalidomide-treated myelodysplastic syndrome patients. Leukemia. 2010;24(3):592–600.
Lucas M, Gadola S, Meier U, Young NT, Harcourt G, Karadimitris A, et al. Frequency and phenotype of circulating Valpha24/Vbeta11 double-positive natural killer T cells during hepatitis C virus infection. J Virol. 2003;77(3):2251–7.
van der Vliet HJ, Molling JW, von Blomberg BM, Kölgen W, Stam AG, de Gruijl TD, et al. Circulating Valpha24 + Vbeta11+ NKT cell numbers and dendritic cell CD1d expression in hepatitis C virus infected patients. Clin Immunol. 2005;114(2):183–9.
Ward SM, Jonsson JR, Sierro S, Clouston AD, Lucas M, Vargas AL, et al. Virus-specific CD8+ T lymphocytes within the normal human liver. Eur J Immunol. 2004;34(6):1526–31.
Baev DV, Peng XH, Song L, Barnhart JR, Crooks GM, Weinberg KI, et al. Distinct homeostatic requirements of CD4+ and CD4- subsets of Valpha24-invariant natural killer T cells in humans. Blood. 2004;104(13):4150–6.
Berzins SP, Cochrane AD, Pellicci DG, Smyth MJ, Godfrey DI. Limited correlation between human thymus and blood NKT cell content revealed by an ontogeny study of paired tissue samples. Eur J Immunol. 2005;35(5):1399–407.
Kim CH, Rott L, Kunkel EJ, Genovese MC, Andrew DP, Wu L, et al. Rules of chemokine receptor association with T cell polarization in vivo. J Clin Invest. 2001;108(9):1331–9.
Fujiwara H, Tsuyuguchi I. Frequency of tuberculin-reactive T-lymphocytes in pleural fluid and blood from patients with tuberculous pleurisy. Chest. 1986;89(4):530–2.
Barnes PF, Fong SJ, Brennan PJ, Twomey PE, Mazumder A, Modlin RL. Local production of tumor necrosis factor and IFN-gamma in tuberculous pleuritis. J Immunol. 1990;145(1):149–54.
Li L, Qiao D, Fu X, Lao S, Zhang X, Wu C. Identification of M. tuberculosis-specific Th1 cells expressing CD69 generated in vivo in pleural fluid cells from patients with tuberculous pleurisy. PLoS One. 2011;6(8):e23700.
Li L, Qiao D, Fu X, Lao S, Zhang X, Wu C. Identification of Mycobacterium tuberculosis-specific Th1, Th17 and Th22 cells using the expression of CD40L in tuberculous pleurisy. PLoS One. 2011;6(5):e20165.
Li L, Qiao D, Zhang X, Liu Z, Wu C. The immune responses of central and effector memory BCG-specific CD4+ T cells in BCG-vaccinated PPD + donors were modulated by Treg cells. Immunobiology. 2011;216(4):477–84.
Qiao D, Li L, Guo J, Lao S, Zhang X, Zhang J, et al. Mycobacterium tuberculosis culture filtrate protein 10-specific effector/memory CD4+ and CD8+ T cells in tubercular pleural fluid, with biased usage of T cell receptor Vβ chains. Infect Immun. 2011;79(8):3358–65.
Feng L, Li L, Liu Y, Qiao D, Li Q, Fu X, et al. B lymphocytes that migrate to tuberculous pleural fluid via the SDF-1/CXCR4 axis actively respond to antigens specific for Mycobacterium tuberculosis. Eur J Immunol. 2011;41(11):3261–9.
Wu SH, Chu JJ, Chiang CD. Increased soluble Fas ligand concentration in tuberculous pleural effusion. J Formos Med Assoc. 2001;100(1):32–4.
Fayyazi A, Eichmeyer B, Soruri A, Schweyer S, Herms J, Schwarz P, et al. Apoptosis of macrophages and T cells in tuberculosis associated caseous necrosis. J Pathol. 2000;191(4):417–25.
Acknowledgments
We would like to thank the patients and healthy volunteers for their perseverance and commitment to this study. This study was supported by a grant from Program of China during the twelfth Five-Year Plan Period (2013ZX10003007-002-003) and the grant from the introduction of innovative R&D team program of Guangdong Province (2009010058).
Disclosure
The authors declare that they have no conflict of interest related to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 40 kb)
Rights and permissions
About this article
Cite this article
Li, Z., Yang, B., Zhang, Y. et al. Mycobacterium Tuberculosis-Specific Memory NKT Cells in Patients with Tuberculous Pleurisy. J Clin Immunol 34, 979–990 (2014). https://doi.org/10.1007/s10875-014-0090-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-014-0090-8