Abstract
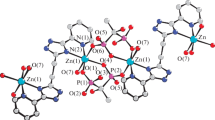
Mixed-ligand zinc(II) complex, {(pipzH2)[Zn(hypydc)(btc)0.5]·H2O} n , (1) was prepared with 4-hydroxypyridine-2,6-dicarboxylic acid (hypydcH2), benzene-1,2,4,5-tetracarboxylic acid (btcH4) and piperazine (pipz). This compound is composed of an anionic complex, [Zn(hypydc)(btc)0.5]2−, protonated piperazine as a counter-ion, (pipzH2)2+ and one uncoordinated water molecule. The three donor atoms of the (hypydc)2− and two donor atoms of bridged (btc)4− ligands form a distorted square pyramid arrangement around the ZnII center. In the crystal structure of the title compound, a wide range of non-covalent interactions consisting of hydrogen bonding (of the types of O–H···O, N–H···O and C–H···O with D···A ranging from 2.630(2) to 3.347(3) Å, ion pairing and C–O···π stacking interactions connect the various components into a supramolecular structure. The quaternary complex system including of hypydcH2–btcH4–pipz and Zn2+ ion, has been investigated. The stoichiometry and stability constants of binary complexes including each of ligands (hypydcH2, btcH4, pipz) in presence of Zn2+ ion, ternary complexes including each two of three ligands (hypydcH2–btcH4, hypydcH2–pipz, btcH4–pipz) in presence of metal ion and finally quaternary systems including three ligands (hypydcH2–btcH4–pipz) and Zn2+ ion were calculated in aqueous solutions by potentiometric pH titration method.
Graphical Abstract
A novel polymeric mixed-ligand zinc(II) complex, {(pipzH2)[Zn(hypydc)(btc)0.5]·H2O} n containing 4-hydroxypyridine-2,6-dicarboxylic acid, benzene-1,2,4,5-tetracarboxylic acid and piperazine was synthesized and characterized.






Similar content being viewed by others
References
Fabelo O, Pasán J, Lloret F, Julve M, Ruiz-Pérez C (2007) Cryst Eng Commun 9:815–827
Aghabozorg H, Manteghi F, Sheshmani S (2008) J Iran Chem Soc 5:184–227
Abu Bakr MS, Rageh HM, Hashem EY, Moustafa NH (1994) Monatsh Chem 125:1197–1205
Berl V, Hue I, Khoury RG, Lehn JM (2001) Chem Eur J 7:2798–2809
Boger DL, Hong J, Hikota M, Ishida M (1999) J Am Chem Soc 121:2471–2477
Fessmann T, Kilburn JD (1999) Angew Chem Int Ed 38:1993–1996
Ng SW (1999) J Organomet Chem 585:12–17
Nakatsuji Y, Bradshaw JS, Tse PK, Arena G, Wilson BE, Wilson NK, Dalley NK, Izatt RM (1985) Chem Commun 12:749–751
Searcey M, MeClean S, Madden B, McGown AT, Wakelin LPG (1998) Anti-cancer Drug Des 13:837–856
Ng SW (1998) Z Kristallogr 213:421–426
Jian-Ping Z, Mei-Hong C, Long-Zhu Z, Qiu-Ju X, Zhi-Qiang X (2011) J Chem Crystallogr 41:1820–1833
Mirzaei M, Eshtiagh-Hosseini H, Karrabi Z, Notash B (2013) Acta Cryst C69:1140–1143
Aghabozorg H, Firoozi N, Roshan L, Eshtiagh-Hosseini H, Salimi AR, Mirzaei M, Ghanbari M, Shamsipur M, Ghadermazi M (2011) J Iran Chem Soc 8:992–1005
Yang L, Cour AL, Anderson OP, Crans DC (2002) Inorg Chem 24:6322–6331
Zhou GW, Guo GC, Liu B, Wang MS, Cai LZ, Huang JS (2004) Bull Korean Chem Soc 25:676–680
Zhou GW, Lan YZ, Zheng FK, Zhang X, Lin MH, Guo GC, Huang JS (2006) Chem Phys Lett 426:341–344
Zhou GW, Wu AQ, Wang MS, Guo GC, Huang JS (2007) Acta Crystallogr E63:m2463–m2464
Canadillas-Delgado L, Pasan J, Delgado FS, Lloret F, Cano J, Julve M, Ruiz-Perez C (2009) Inorg Chem 48:11342–11351
Aghabozorg H, Ilaie N, Heidari M, Manteghi F, Pasdar H (2008) Acta Cryst E64:m1351–m1352
Mirzaei M, Lippolis V, Eshtiagh-Hosseini H, Mahjoobizadeh M (2012) Acta Cryst C68:m7–m11
Feng X, Liu J, Li TF, Lei PP (2011) Z Kristallogr NCS 226:80–82
Xing QJ, Chen MH, Xie Y, Dai SC, Zhang LZ, Zou JP (2010) Z Kristallogr NCS 225:671–672
Aghabozorg H, Bahrami Z, Tabatabaei M, Ghadermazi M, Attar-Gharamaleki J (2007) Acta Cryst E63:m2022–m2023
Aghabozorg H, Attar-Gharamaleki J, Motyeian E, Ghadermazi M (2007) Acta Cryst E63:m2793–m2794
Aghabozorg H, Ghadermazi M, Sheshmani S (2006) Anal Sci 22:x251–x252
Aghabozorg H, Mahfoozi F, Sharif MA, Shokrollahi A, Derki S, Shamsipur M, Khavasi HR (2010) J Iran Chem Soc 7:727–739
Jia YY, Chen B, Yuan YX (2012) Acta Cryst E68:m1150
Yao JC, Wang YF, Zhang L, Guo JB, Cao XX, Fan CP (2011) Chin J Struct Chem 30:170–175
Yuan JX, Xiao HP, Hu ML (2004) Z Kristallogr NCS 219:224–226
Pasban N, Esmhosseini M, Ahmadi M, Mohebbi M, Salkhordeh S, Vatani M (2012) Z Kristallogr NCS 227:265–266
Sharif MA, Najafi GR (2013) Acta Chim Slov 60:138–143
Bruker (2007) APEX2. SAINT & SADABS Bruker AXS Inc., Madison
Sheldrick GM (2008) Acta Cryst A64:112–122
Gans P, Sabatini A, Vacca A (1996) Talanta 43:1739–1753
Alderighi L, Gans P, Ienco A, Peters D, Sabatini A, Vacca A (1999) Coord Chem Rev 184:311–318
Harned HS, Owen BB (1958) The physical chemistry of electrolytic solutions, 3rd edn. Reinhold Publishing Corp, New York, pp 634–649
Addison AW, Rao TN, Reedijk J, van Rijn J, Verschoor GC (1984) J Chem Soc Dalton Trans 7:1349–1356
Moghimi A, Sharif MA, Shokrollahi A, Shamsipur M, Aghabozorg H (2005) Z Anorg Allg Chem 631:902–908
Aghajani Z, Sharif MA, Aghabozorg H, Naderpour A (2006) Acta Cryst E62:m830
Tabatabaee M, Aghabozorg H, Attar-Gharamaleki J, Sharif MA (2009) Acta Cryst E65:m473–m474
Derikvand Z, Olmstead MM, Mercado BQ, Shokrollahi A, Shahryari M (2013) Inorg Chim Acta 406:256–265
Aghabozorg H, Manteghi F, Ghadermazi M, Mirzaei M, Salimi AR, Shokrollahi A, Derki S, Eshtiagh-Hosseini H (2009) J Mol Struct 919:381–388
Derikvand Z, Nemati A, Shokrollahi A, Zarghampour F (2012) Inorg Chim Acta 392:362–373
Moghimi A, Sheshmani S, Shokrollahi A, Shamsipur M, Kickelbik G, Aghabozorg H (2005) Z Anorg Allg Chem 631:160–169
Martell AE, Motekaitis RJ (1992) Determination and use of stability constants, 2nd edn. VCH, New York
Norkus E, Stalnionien I, Crans DC (2003) Heteroat Chem 14:625–632
Shokrollahi A, Ghaedi M, Rajabi HR, Niband MS (2008) Spectrochim Acta A 71:655–662
Aghabozorg H, Ramezanipour F, Soleimannejad J, Sharif MA, Shokrollahi A, Shamsipur M, Moghimi A, Attar-Gharamaleki J, Lippolis V, Blake AJ (2008) Polish J Chem 82:487–507
Acknowledgments
The authors greatly acknowledge the scientific support from the Islamic Azad University, Qom Branch, to the first author in this study. This paper is a part of a project entitled “Synthesis and characterization of two valance metal complexes with mixed ligand of multifunctional carboxylic acids”.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
A view of the C–O···π stacking interaction between the carbonyl group of the 4-hydroxypyridine-2,6-dicarboxylate groups and the pyridine ring of hypydc2− fragments, with an O···π distance of 3. 659(2) Å for C7–O4···Cg1 (1/2-X, 1/2 + Y, 1/2-Z) [Cg1 is the centroid for the (N1/C1-C5) ring]. Supplementary material 1 (JPEG 91 kb)
Fig. S2
Distribution diagrams of hypydc–btc (a), hypydc–pipz (b), btc–pipz (c), hypydc–btc– pipz (d) proton transfer systems in aqueous solution at 25 ± 0.1 °C and μ = 0.1 M NaNO3. Supplementary material 2 (JPEG 1586 kb)
Fig. S3
Potentiometric titration curves of hypydc (a) and btc (b) in the absence and presence of Zn2+ions with NaOH 0.09613 M in aqueous solution at 25 ± 0.1 °C and μ = 0.1 M NaNO3 and distribution diagrams of hypydc–Zn2+ (c) and btc–Zn2+ (d). Supplementary material 3 (JPEG 293 kb)
Rights and permissions
About this article
Cite this article
Sharif, M.A., Tabatabaee, M., Shokrollahi, A. et al. The Zinc(II) Coordination Polymer with Mixed-Ligand of 4-Hydroxypyridine-2,6-dicarbolic Acid and Benzene-1,2,4,5-tetracarboxylic Acid in the Presence of Piperazine as a Counter Ion: Synthesis, Crystal Structure and Solution Study. J Chem Crystallogr 45, 103–113 (2015). https://doi.org/10.1007/s10870-015-0565-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-015-0565-3