Abstract
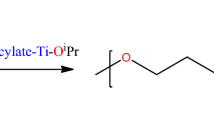
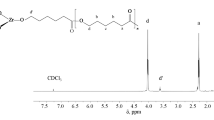
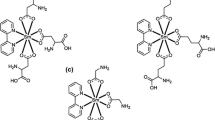
In this study, zirconium salicylate compounds (1–6) were prepared by reaction of zirconium(IV) propoxide with 5-chlorosalicylic acid, 4-hydroxysalicylic acid, and 5-nitrosalicylic acid in 1-propanol, respectively. All these compounds (1–6) were characterized by 1H, 13C NMR, FTIR, mass spectroscopy, elemental, and thermogravimetric analyses. These compounds were tested as catalysts in polymerization of ε-caprolactone and were effective. Polycaprolactone was characterized by 1H, 13C NMR, and gel permeation chromatography. The number of salicylate ligands (bonded to zirconium atom) effects and substituents (on salicylate ligands) effects on the polymerization reactions were investigated first time by this study.






Similar content being viewed by others
References
Gowda, R.R., Chakraborty, D., Ramkumar, V.: J. Organomet. Chem. 696, 572–580 (2011)
Vivani, R., Masci, S., Alberti, G.: Inorg. Chem. 43, 368–374 (2004)
Dinger, M.B., Henderson, W.: J. Organomet. Chem. 560, 233–243 (1998)
Yalcin, G., Yildiz, U., Kayan, A.: Appl. Catal. A Gen. 423, 205–210 (2012)
Gao, Y., Choudhury, N.R., Matisons, J., Schubert, U., Moraru, B.: Chem. Mater. 14, 4522–4529 (2002)
Yalcin, G., Kayan, A.: Appl. Catal. A Gen. 433, 223–228 (2012)
Piszczek, P., Radtke, A., Wojtczak, A., Muziol, T., Chojnacki, J.: Polyhedron 28, 279–285 (2009)
Lerot, L., Legrand, F., De Bruycker, P.: J. Mater. Sci. 26, 2353–2358 (1991)
Hubert-Pfalzgraf, L.G.: Appl. Organomet. Chem. 6, 627–643 (1992)
Kickelbick, G., Schubert, U.: J. Chem. Soc. Dalton Trans. 8, 1301–1305 (1999)
Chmura, A.J., Cousins, D.M., Davidson, M.G., Jones, M.D., Lunn, M.D., Mahon, M.F.: Dalton Trans. 11, 1437–1443 (2008)
Kayan, A.: J. Appl. Polym. Sci. 123, 3527–3534 (2012)
Chisholm, M.H.: Pure Appl. Chem. 82, 1647–1662 (2010)
Albertsson, A.-C., Varma, I.K.: Adv. Polym. Sci. 157, 1–40 (2002)
Kricheldorf, H.R.: J. Macromol. Sci. Pure Appl. Chem. 42, 4723–4742 (2004)
Darensbourg, D.J., Karroonnirun, O.: Macromolecules 43, 8880–8886 (2010)
Lee, W.Y., Hsieh, C.C., Hsu, J.W., Datta, A., Lin, Y.C., Huang, J.H., Lee, T.Y.: J. Organomet. Chem. 696, 3816–3821 (2011)
Gornshtein, F., Kapon, M., Botoshansky, M., Eisen, M.S.: Organometallics 26, 497–507 (2007)
Stavila, V., Thurston, J.H., Whitmire, K.H.: Inorg. Chem. 48, 6945–6951 (2009)
Cutler, A., Raja, M., Todaro, A.: Inorg. Chem. 26, 2877–2881 (1987)
Maggini, S., Girardi, F., Muller, K., Maggio, R.D.: J. Appl. Polym. Sci. 124, 2110–2117 (2012)
Li, J., Gao, Z., Han, L., Gao, L., Zhang, G., Tikkanen, W.: J. Organomet. Chem. 695, 1848–1851 (2010)
Schubert, U.: Acc. Chem. Res. 40, 730–737 (2007)
Stevels, W.M., Ankone, M.J.K., Dijkstra, P.J., Feijen, J.: Macromol. Chem. Phys. 196, 1153–1161 (1995)
Ruan, J.M., Xiao, A.G., Wu, H.W., Yang, H.L.: Des. Monomer Polym. 17, 345–355 (2014)
Chmura, A.J., Davidson, M.G., Jones, M.D., Lunn, M.D., Mahon, M.F., Johnson, A.F., Khunkamchoo, P., Roberts, S.L., Wong, S.S.F.: Macromolecules 39, 7250–7257 (2006)
Mert, O., Kayan, A.: Appl. Catal. A Gen. 464, 322–331 (2013)
Saha, T.K., Rajashekhar, B., Chakraborty, D.: RSC Adv. 2, 307–318 (2012)
Miranda, M.O., DePorre, Y., Vazquez-Lima, H., Johnson, M.A., Marell, D.J., Cramer, C.J., Tolman, W.B.: Inorg. Chem. 52, 13692–13701 (2013)
Reyes-Lopez, S.Y., Richa, A.M.: Macromol. Symp. 325, 21–37 (2013)
Jones, D., Cavell, K., Keim, W.: J. Mol. Catal. A: Chem. 138, 37–52 (1999)
Kricheldorf, H.R., Berl, M., Scharnagl, N.: Macromolecules 21, 286–293 (1988)
Mun, S.D., Hong, Y., Kim, Y.: Bull. Korean Chem. Soc. 28, 698–700 (2007)
Acknowledgments
This work was supported by the research foundation of Kocaeli University, (Project No: 2013/56).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mert, O., Kayan, A. Synthesis and characterization of substituted salicylate zirconium compounds and their catalytic activity over ε-caprolactone. J Incl Phenom Macrocycl Chem 80, 409–416 (2014). https://doi.org/10.1007/s10847-014-0429-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-014-0429-z