Abstract
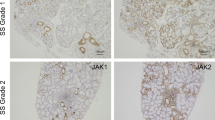
Tight junctions (TJs) are necessary for salivary gland function and may serve as indicators of salivary gland epithelial dysfunction. IgG4-related disease (IgG4-RD) is a newly recognized fibro-inflammatory condition which disrupts the TJ associated epithelial barrier. The salivary glands are one of the most frequently involved organs in IgG4-RD, however, changes of the TJ associated epithelial barrier in salivary gland duct epithelium is poorly understood. Here, we investigated the regulation and function of TJs in human submandibular gland ductal epithelial cells (HSDECs) in normal and IgG4-RD. We examined submandibular gland (SMG) tissue from eight control individuals and 22 patients with IgG4-RD and established an HSDEC culture system. Immunohistochemistry, immunocytochemistry, western blotting, and measurement of transepithelial electrical resistance (TER) were performed. Claudin-4, claudin-7, occludin, and JAM-A were expressed at the apical side of the duct epithelium in submandibular gland (SMG) tissue and at the cell borders in HSDECs of normal and IgG4-RD. The expression and distribution of TJs in SMG tissue were not different in control individuals and patients with IgG4-RD in vivo and in vitro. Although interferon-gamma (IFNγ) generally disrupts the integrity and function of TJs, as manifested by decreased epithelial barrier function, IFNγ markedly increased the epithelial barrier function of HSDECs via upregulation of claudin-7 expression in HSDECs from patients with IgG4-RD. This is the first report showing an IFNγ-dependent increase in epithelial barrier function in the salivary gland duct epithelium. Our results provide insights into the functional significance of TJs in salivary gland duct epithelium in physiological and pathological conditions, including IgG4-RD.




Similar content being viewed by others
References
Alexandre MD, Lu Q, Chen YH (2005) Overexpression of claudin-7 decreases the paracellular Cl-conductance and increases the paracellular Na+ conductance in LLC-PK1 cells. J Cell Sci 118:2683–2693. doi:10.1242/jcs.02406
Balda MS, Matter K (2009) Tight junctions and the regulation of gene expression. Biochim Biophys Acta 1788:761–767. doi:10.1016/j.bbamem.2008.11.024
Baron S, Poast J, Cloyd MW (1999) Why is HIV rarely transmitted by oral secretions? Saliva can disrupt orally shed, infected leukocytes. Arch Intern Med 159:303–310. doi:10.1001/archinte.159.3.303
Boivin MA, Roy PK, Bradley A, Kennedy JC, Rihani T, Ma TY (2009) Mechanism of interferon-gamma-induced increase in T84 intestinal epithelial tight junction. J Interf Cytokine Res 29:45–54. doi:10.1089/jir.2008.0128
Cereijido M, Valdes J, Shoshani L, Contreras RG (1998) Role of tight junctions in establishing and maintaining cell polarity. Ann Rev Physiol 60:161–177. doi:10.1146/annurev.physiol.60.1.161
Cereijido M, Contreras RG, Flores-Benítez D, Flores-Maldonado C, Larre I, Ruiz A, Shoshani L (2007) New diseases derived or associated with the tight junction. Arch Med Res 38:465–478. doi:10.1016/j.arcmed.2007.02.003
Deshpande V, Zen Y, Chan JK et al (2012) Consensus statement on the pathology of IgG4-related disease. Mod Pathol 25:1181–1192. doi:10.1038/modpathol.2012.72
Ewert P, Aguilera S, Alliende C et al (2010) Disruption of tight junction structure in salivary glands from Sjogren’s syndrome patients is linked to proinflammatory cytokine exposure. Arthritis Rheum 62:1280–1289. doi:10.1002/art.27362
Fitzner B, Holzhueter SA, Ibrahim S, Nizze H, Jaster R (2009) Interferon-gamma treatment accelerates and aggravates autoimmune pancreatitis in the MRL/Mp-mouse. Pancreatology 9:233–239. doi:10.1159/000199434
Fujita K, Katahira J, Horiguchi Y, Sonoda N, Furuse M, Tsukita S (2000) Clostridium perfringens enterotoxin binds to the second extracellular loop of claudin-3, a tight junction integral membrane protein. FEBS Lett 476:258–261. doi:10.1016/S0014-5793(00)01744-0
Gumbiner BM (1993) Breaking through the tight junction barrier. J Cell Biol 123:1631–1633. doi:10.1016/j.febslet.2008.11.032
Himi T, Takano K, Yamamoto M, Naishiro Y, Takahashi H (2012) A novel concept of Mikulicz’s disease as IgG4-related disease. Auris Nasus Larynx 39:9–17. doi:10.1007/s10165-011-0508-6
Kamisawa T, Zen Y, Pillai S, Stone JH (2015) IgG4-related disease. Lancet 385:1460–1471. doi:10.1016/S0140-6736(14)60720-0
Kawano Y, Kobune M, Yamaguchi M et al (2003) Ex vivo expansion of human umbilical cord hematopoietic progenitor cells using a coculture system with human telomerase catalytic subunit (hTERT)-transfected human stromal cells. Blood 101:532–540. doi:10.1182/blood-2002-04-1268
Kawedia JD, Nieman ML, Boivin GP et al (2007) Interaction between transcellular and paracellular water transport pathways through Aquaporin 5 and the tight junction complex. PNAS 104:3621–3626. doi:10.1073/pnas.0608384104
Kojima T, Yamamoto T, Murata M, Chiba H, Kokai Y, Sawada N (2003) Regulation of the blood-biliary barrier: interaction between gap and tight junctions in hepatocytes. Med Electron Microsc 36:157–164. doi:10.1007/s00795-003-0220-5
Kojima T, Takano K, Yamamoto T et al (2008) Transforming growth factor-beta induces epithelial to mesenchymal transition by down-regulation of claudin-1 expression and the fence function in adult rat hepatocytes. Liver Int 28:534–545. doi:10.1111/j.1478-3231.2007.01631.x
Kojima T, Go M, Takano K et al (2013) Regulation of tight junctions in upper airway epithelium. Biomed Res Int 2013:947072. doi:10.1155/2013/947072
Krug SM, Günzel D, Conrad MP, Lee IF, Amasheh S, Fromm M, Yu AS (2012) Charge-selective claudin channels. Ann N Y Acad Sci 1257:20–28. doi:10.1111/j.1749-6632.2012.06555.x
Lutz KL, Siahaan TJ (1997) Molecular structure of the apical junction complex and its contribution to the paracellular barrier. J Pharm Sci 86:977–984. doi:10.1021/js970134j
Mahajan VS, Mattoo H, Deshpande V, Pillai SS, Stone JH (2014) IgG4-related disease. Annu Rev Pathol 9:315–347. doi:10.1146/annurev-pathol-012513-104708
Maria OM, Kim JM, Gerstenhaber JA, Baum BJ, Tran SD (2008) Distribution of tight junction proteins in adult human salivary glands. J Histochem Cytochem 56:1093–1098. doi:10.1369/jhc.2008.951780
Martinez CA, de Campos FG, de Carvalho VR, de Castro Ferreira C, Rodrigues MR, Sato DT, Pereira JA (2015) Claudin-3 and occludin tissue content in the glands of colonic mucosa with and without a fecal stream. J Mol Histol 46:183–194. doi:10.1007/s10735-015-9610-y
Martin-Padura I, Lostaglio S, Schneemann M, Williams L, Romano M, Fruscella P, Panzeri C, Stoppacciaro A, Ruco L, Villa A, Simmons D, Dejana E (1998) Junctional adhesion molecule, a novel member of the immunoglobulin superfamily that distributes at intercellular junctions and modulates monocyte transmigration. J Cell Biol 142:117–127
Masaki Y, Sugai S, Umehara H (2010) IgG4-related diseases including Mikulicz’s disease and sclerosing pancreatitis: diagnostic insights. J Rheum 37:1380–1385. doi:10.3899/jrheum.091153
Melvin JE, Yule D, Shuttleworth T, Begenisich T (2005) Regulation of fluid and electrolyte secretion in salivary gland acinar cells. Ann Rev Physiol 33:445–469. doi:10.1146/annurev.physiol.67.041703.084745
Meredith LW, Wilson GK, Fletcher NF, McKeating JA (2012) Hepatitis C virus entry: beyond receptors. Rev Med Virol 22:182–193. doi:10.1002/rmv.723
Mineta K, Yamamoto Y, Yamazaki Y, Tanaka H, Tada Y, Saito K, Tamura A, Igarashi M, Endo T, Takeuchi K, Tsukita S (2011) Predicted expansion of the claudin multigene family. FEBS Lett 585:606–612. doi:10.1016/j.febslet.2011.01.028
Monteiro AC, Parkos CA (2012) Intracellular mediators of JAM-A-dependent epithelial barrier function. Ann NY Acad Sci 1257:115–124. doi:10.1111/j.1749-6632.2012.06521.x
Naydenov NG, Baranwal S, Khan S, Feygin A, Gupta P, Ivanov AI (2013) Novel mechanism of cytokine-induced disruption of epithelial barriers: Janus kinase and protein kinase D-dependent downregulation of junction protein expression. Tissue Barriers 1:e25231. doi:10.4161/tisb.25231
Saitou M, Fujimoto K, Doi Y, Itoh M, Fujimoto T, Furuse M, Takano H, Noda T, Tsukita S (1998) Occludin-deficient embryonic stem cells can differentiate into polarized epithelial cells bearing tight junctions. J Cell Biol 141:397–408. doi:10.1083/jcb.141.2.397
Sawada N (2013) Tight junction-related human diseases. Pathol Int 63:1–12. doi:10.1111/pin.12021
Sawada N, Murata M, Kikuchi K, Osanai M, Tobioka H, Kojima T, Chiba H (2003) Tight junctions and human diseases. Med Electron Microsc 36:147–156. doi:10.1111/pin.12021
Schneeberger EE, Lynch RD (1992) Structure, function, and regulation of cellular tight junctions. Am J Physiol Lung Cell Mol Physio 262:L647–L661
Schneeberger EE, Lynch RD (2004) The tight junction: a multifunctional complex. Am J Physiol Cell Physiol 286:1213–1228. doi:10.1152/ajpcell.00558.2003
Schulzke JD, Gitter AH, Mankertz J, Spiegel S, Seidler U, Amasheh S, Saitou M, Tsukita S, Fromm M (2005) Epithelial transport and barrier function in occludin-deficient mice. Biochim Biophys Acta 1669:34–42. doi:10.1016/j.bbamem.2005.01.008
Someya M, Kojima T, Ogawa M, Ninomiya T, Nomura K, Takasawa A, Murata M, Tanaka S, Saito T, Sawada N (2013) Regulation of tight junctions by sex hormones in normal human endometrial epithelial cells and uterus cancer cell line Sawano. Cell Tissue Res 354:481–494. doi:10.1007/s00441-013-1676-9
Stone JH, Zen Y, Deshpande V (2012) IgG4-related disease. N Engl J Med 366:539–551. doi:10.1056/NEJMra1104650
Takano K, Kojima T, Go M, Murata M, Ichimiya S, Himi T, Sawada N (2005) HLA-DR- and CD11c-positive dendritic cells penetrate beyond well-developed epithelial tight junctions in human nasal mucosa of allergic rhinitis. J Histochem Cytochem 53:611–619. doi:10.1369/jhc.4A6539.2005
Tsukita S, Furuse M, Itoh M (2001) Multifunctional strands in tight junctions. Nat Rev Mol Cell Biol 2:285–293. doi:10.1038/35067088
Umehara H, Okazaki K, Masaki Y et al (2011) Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol 22:21–30. doi:10.1007/s10165-011-0571-z
van Meer G, Simons K (1986) The function of tight junctions in maintaining differences in lipid composition between the apical and the basolateral cell surface domains of MDCK cells. EMBO J 5:1455–1464
Visser J, Rozing J, Sapone A, Lammers K, Fasano A (2009) Tight junctions, intestinal permeability, and autoimmunity: celiac disease and type 1 diabetes paradigms. Ann N Y Acad Sci 1165:195–205. doi:10.1111/j.1749-6632.2009.04037.x
Yamaguchi H, Kojima T, Ito T, Kimura Y, Imamura M, Son S, Koizumi J, Murata M, Nagayama M, Nobuoka T, Tanaka S, Hirata K, Sawada N (2010) Transcriptional control of tight junction proteins via a protein kinase C signal pathway in human telomerase reverse transcriptase-transfected human pancreatic duct epithelial cells. Am J Pathol 177:698–712. doi:10.2353/ajpath.2010.091226
Yamamoto M, Takahashi H, Shinomura Y (2014) Mechanisms and assessment of IgG4-related disease: lessons for the rheumatologist. Nat Rev Rheumatol 10:148–159. doi:10.1038/nrrheum.2013.183
Acknowledgments
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan (Grant no.: 26861401).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Rights and permissions
About this article
Cite this article
Abe, A., Takano, K., Kojima, T. et al. Interferon-gamma increased epithelial barrier function via upregulating claudin-7 expression in human submandibular gland duct epithelium. J Mol Hist 47, 353–363 (2016). https://doi.org/10.1007/s10735-016-9667-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-016-9667-2