Abstract
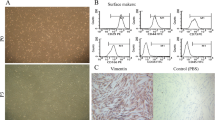
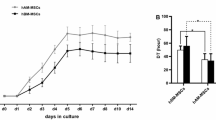
Human amnion mesenchymal stem cells (HAMSCs) can be obtained from human amniotic membrane, a highly abundant and readily available tissue. HAMSC sources present fewer ethical issues, have low immunogenicity, anti-inflammatory properties, considerable advantageous characteristics, and are considered an attractive potential treatment material in the field of regenerative medicine. We used a co-culture system to determine whether HAMSCs could promote osteogenesis in human bone marrow mesenchymal stem cells (HBMSCs). We isolated HAMSCs from discarded amnion samples and collected them using pancreatin/collagenase digestion. We cultured HAMSCs and HBMSCSs in basal medium. Activity of alkaline phosphatase (ALP), an early osteogenesis marker, was increased in the co-culture system compared to the control single cultures, which we also confirmed by ALP staining. We used immunofluorescence testing to investigate the effects of co-culturing with HAMSCs on HBMSC proliferation, which revealed that the co-culturing enhanced EdU expression in HBMSCs. Western blotting and quantitative real-time PCR indicated that co-culturing promoted osteogenesis in HBMSCs. Furthermore, Alizarin red S staining revealed that extracellular matrix calcium levels in mineralized nodule formation produced by the co-cultures were higher than that in the controls. Using the same co-culture system, we further observed the effects of HAMSCs on osteogenic differentiation in primary osteoblasts by Western blotting, which better addressed the mechanism for HAMSCs in bone regeneration. The results showed HAMSCs are osteogenic and not only play a role in promoting HBMSC proliferation and osteogenic differentiation but also in osteoblasts, laying the foundation for new regenerative medicine methods.





Similar content being viewed by others
References
Aglyamov SR et al (2007) Model-based reconstructive elasticity imaging using ultrasound. Int J Biomed Imaging 2007:35830. doi:10.1155/2007/35830
Alviano F et al (2007) Term amniotic membrane is a high throughput source for multipotent mesenchymal stem cells with the ability to differentiate into endothelial cells in vitro. BMC Dev Biol. doi:10.1186/1471-213x-7-11
Anderson HC (1995) Molecular biology of matrix vesicles. Clin Orthop Relat Res: 266–280
Bahlous A, Kalai E, Hadj Salah M, Bouzid K, Zerelli L (2006) Biochemical markers of bone remodeling: recent data of their applications in managing postmenopausal osteoporosis. Tunis Med 84:751–757
Balcerzak M, Hamade E, Zhang L, Pikula S, Azzar G, Radisson J, Bandorowicz-Pikula J, Buchet R (2003) The roles of annexins and alkaline phosphatase in mineralization process. Acta Biochim Pol 50(4):1019–1038
Baxter RC (2000) Insulin-like growth factor (IGF)-binding proteins: interactions with IGFs and intrinsic bioactivities. Am J Physiol Endocrinol Metab 278:E967–E976
Chen MQ, Wang X, Ye ZY, Zhang Y, Zhou Y, Tan WS (2011) A modular approach to the engineering of a centimeter-sized bone tissue construct with human amniotic mesenchymal stem cells-laden microcarriers. Biomaterials 32:7532–7542. doi:10.1016/j.biomaterials.2011.06.054
Cho YE et al (2010) Red yeast rice stimulates osteoblast proliferation and increases alkaline phosphatase activity in MC3T3-E1 cells. Nutr Res 30:501–510. doi:10.1016/j.nutres.2010.06.011
Ducy P et al (1996) Increased bone formation in osteocalcin-deficient mice. Nature 382:448–452. doi:10.1038/382448a0
Farley JR, Hall SL, Tanner MA, Wergedal JE (1994) Specific activity of skeletal alkaline phosphatase in human osteoblast-line cells regulated by phosphate, phosphate esters, and phosphate analogs and release of alkaline phosphatase activity inversely regulated by calcium. J Bone Miner Res 9:497–508. doi:10.1002/jbmr.5650090409
Franceschi RT, Ge C, Xiao G, Roca H, Jiang D (2009) Transcriptional regulation of osteoblasts. Cells Tissues Organs 189:144–152. doi:10.1159/000151747
Hildebrandt H, Hermann C, Knittel R, Richter-Reichhelm M, Siegel A, Witzenrath W (2010) Gesundes Kinzigtal Integrated Care: improving population health by a shared health gain approach and a shared savings contract. Int J Integr Care 10:e046
Kim SH et al (2009) Alveolar bone regeneration by transplantation of periodontal ligament stem cells and bone marrow stem cells in a canine peri-implant defect model: a pilot study. J Periodontol 80:1815–1823. doi:10.1902/jop.2009.090249
Leyva-Leyva M et al (2013) Characterization of mesenchymal stem cell subpopulations from human amniotic membrane with dissimilar osteoblastic potential. Stem Cells Dev 22:1275–1287. doi:10.1089/scd.2012.0359
Liu X et al (2014) The Rho kinase inhibitor Y-27632 facilitates the differentiation of bone marrow mesenchymal stem cells. J Mol Histol. doi:10.1007/s10735-014-9594-z
Marcus AJ, Coyne TM, Rauch J, Woodbury D, Black IB (2008) Isolation, characterization, and differentiation of stem cells derived from the rat amniotic membrane. Differentiation 76:130–144. doi:10.1111/j.1432-0436.2007.00194.x
Musial K, Fornalczyk K, Zwolinska D (2008) Osteopontin (OPN), PDGF-BB (platelet-derived growth factor) and BMP-7 (bone morphogenetic protein) as markers of atherogenesis in children with chronic kidney disease (CKD) treated conservatively-preliminary results Polski merkuriusz lekarski: organ Polskiego Towarzystwa Lekarskiego 24 (4):25–27
Ozeki K, Aoki H, Fukui Y (2008) The effect of adsorbed vitamin D and K to hydroxyapatite on ALP activity of MC3T3-E1 cell. J Mater Sci Mater Med 19:1753–1757. doi:10.1007/s10856-007-3288-y
Parolini O et al (2008) Concise review: Isolation and characterization of cells from human term placenta: outcome of the first international workshop on placenta derived stem cells. Stem Cells 26:300–311. doi:10.1634/stemcells.2007-0594
Phimphilai M, Zhao Z, Boules H, Roca H, Franceschi RT (2006) BMP signaling is required for RUNX2-dependent induction of the osteoblast phenotype. J Bone Miner Res 21:637–646. doi:10.1359/jbmr.060109
Reinholz GG, Getz B, Pederson L, Sanders ES, Subramaniam M, Ingle JN, Spelsberg TC (2000) Bisphosphonates directly regulate cell proliferation, differentiation, and gene expression in human osteoblasts. Cancer Res 60:6001–6007
Schneider GB, Whitson SW, Cooper LF (1999) Restricted and coordinated expression of β3-integrin and bone sialoprotein during cultured osteoblast differentiation. Bone 24:321–327
Thangakumaran S, Sudarsan S, Arun KV, Talwar A, James JR (2009) Osteoblast response (initial adhesion and alkaline phosphatase activity) following exposure to a barrier membrane/enamel matrix derivative combination. Indian J Dent Res 20:7–12
Tsuji H et al (2010) Xenografted human amniotic membrane-derived mesenchymal stem cells are immunologically tolerated and transdifferentiated into cardiomyocytes. Circ Res 106:1613–1623. doi:10.1161/CIRCRESAHA.109.205260
Wang SY et al (2009) Vertical alveolar ridge augmentation with beta-tricalcium phosphate and autologous osteoblasts in canine mandible. Biomaterials 30:2489–2498. doi:10.1016/j.biomaterials.2008.12.067
Wu W, Ye ZY, Zhou Y, Tan WS (2011) AICAR, a small chemical molecule, primes osteogenic differentiation of adult mesenchymal stem cells. Int J Artif Organs 34:1128–1136. doi:10.5301/Ijao.5000007
Wu B et al (2013) Lentiviral delivery of biglycan promotes proliferation and increases osteogenic potential of bone marrow-derived mesenchymal stem cells in vitro. J Mol Histol 44:423–431. doi:10.1007/s10735-013-9497-4
Wu C, Liu HL, Chen JH (2007) Modeling dynamic cerebral blood volume changes during brain activation on the basis of the blood-nulled functional MRI signal. NMR Biomed 20:643–651. doi:10.1002/nbm.1116
Yakar S et al (2002) Circulating levels of IGF-1 directly regulate bone growth and density. J Clin Investig 110:771–781. doi:10.1172/JCI15463
Zhang D, Jiang M, Miao D (2011) Transplanted human amniotic membrane-derived mesenchymal stem cells ameliorate carbon tetrachloride-induced liver cirrhosis in mouse. PLoS One 6:e16789. doi:10.1371/journal.pone.0016789
Zhao J et al (2009) Apatite-coated silk fibroin scaffolds to healing mandibular border defects in canines. Bone 45:517–527. doi:10.1016/j.bone.2009.05.026
Acknowledgments
This study was supported by the National Natural Science Foundation of China (81271109) and project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yuli Wang and Ying Yin have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Y., Yin, Y., Jiang, F. et al. Human amnion mesenchymal stem cells promote proliferation and osteogenic differentiation in human bone marrow mesenchymal stem cells. J Mol Hist 46, 13–20 (2015). https://doi.org/10.1007/s10735-014-9600-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-014-9600-5