Abstract
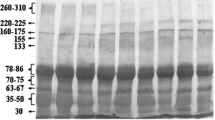
Immunoglobulin G (IgG) is a major glycoprotein in ruminant colostrum. First day buffalo colostrum protein was purified on Sephadex G-100 and its mass was determined by MALDI-TOF as 147.848 KDa. The PMF data of protein subunits revealed its homology to IgG, which was supported by the identification of peptide sequences LLIYGATSR and VYNEYLPAPIVR corresponding to light and heavy chains of IgG by CID MS/MS analysis. The N-glycan microheterogeneity was established based on chemoselective glycoblotting technique with the identification of high mannose, neutral complex/hybrid and sialylated complex/hybrid glycans. A complete structural assignment of 54 N-linked oligosaccharides were identified and the ratio of sialyl oligosaccharides was found to be higher compared to neutral saccharides. The fucosylation observed in more than 20 oligosaccharides, high mannose and trisialyl oligosaccharides were present in diminutive amount. The high non-fucosyl and sialyl oligosaccharides in buffalo colostrum IgG provide ample scope for its utilization in targeted therapies to elicit effective ADCC and anti-inflammatory responses.






Similar content being viewed by others
Abbreviations
- MALDI-TOF:
-
Matrix-assisted laser desorption/ionization-time of flight
- LC-MS/MS:
-
Liquid chromatography–mass spectrometry
- PNGase F:
-
Peptide -N-Glycosidase F
- PHM:
-
1-propanesulfonic acid 2-hydroxyl-3-myristamido
- DTT:
-
Dithiothreitol
- IAA:
-
Iodoacetic acid CID, Collision induced dissociation
- PMF:
-
Peptide mass fingerprinting
- ADCC:
-
Antibody-dependent cellular cytotoxicity
- CDC:
-
Complement-dependent cytotoxicity
References
Pramod, G., Gill, H.S.: Oligosaccharides and glycoconjugates in bovine milk and colostrum. Br. J. Nutr. 84, 69–74 (2000)
Aurelia, P., Cristian, C., Camelia, R., Vioara, M., Gheorghe, M.: The study of the main parameters quality of buffalo milk. Afr. J. Microbiol. Res. 10, 201–206 (2009)
Antonius, M.: In vivo antimicrobial and an-tiviral activity of components in bovine milk and colostrum involved in non-specific defence. Br. J. Nutr. 84, 127–134 (2000)
Uruakpa, O., Ismond, M.A.H., Akobundu, E.N.T.: Colostrum and its benefits: a review. Nutr. Res. 22, 755–767 (2002)
Martinez-Ferez, A., Rudloff, S., Guadix, A., Cordula, A.H., Pohlentz, G., Boza, J.B., Guadix, E.M., Kunz, C.: Goat’s milk as a natural source of lactose-derived oligosaccharides: isolation by membrane technology. Int. Dairy J. 16, 173–181 (2006)
Colarow, L., Turini, M., Tencherg, S., Berger, A.: Characterization and biological activity of gangliosides in buffalo milk. Biochim. Biophys. Acta 1631, 94–106 (2003)
Bulter, J.E.: Bovine immunoglobulins: an augmented review. Vet. Immunol. Immunopath. 4, 43–152 (1983)
Dang, A.K., Kapila, S., Purohit, M., Singh, C.: Changes in colostrum of Murrah buffaloes after calving. Trop. Anim. Health. Pro. 41, 1213–1217 (2008)
Kulkarni, B.A.: Immunoglobulins of the Indian buffalo. V. Changes in colostrum milk and neonatal calf serum immunoglobulins in early lactation. Indian J. Biochem. Biophys. 18, 28–31 (1981)
Goodman, W.: Immunoglobulin structure and function. In: Stites, D.P., et al. (eds.) Basic and clinical Immunology, p. 27. Lang. Med. Pub., Los Altos, CA (1987)
Arnold, N., Wormald, M.R., Sim, R.B., Rudd, P.M., Dwek, R.A.: The impact of glycosylation on the biological function and structure of human immunoglobulins. Ann. Rev. Immunol. 25, 21–50 (2007)
Zeitlin, L., Pettitt, J.J., Scully, C.C., Bohorova, N.N., Do, D.K., Pauly, M.M., Hiatt, A.A., Ngo, L.L., Steinkellner, H.H., Whaley, K.J.K., Olingerb, G.G.: Enhanced potency of a fucose-free monoclonal antibody being developed as an Ebola virus immunoprotectant. Proc. Natl. Acad. Sci. 108, 20690–20694 (2011)
Kai-Ting, S., Robert, M.A.: Antibody glycosylation and Inflammation. Antibodies 2, 392–414 (2013)
Nimmerjahn, F., Ravetch, J.V.: Divergent Immunoglobulin G subclass activity through selective Fc receptor binding. Sci. 310, 1510–1512 (2005)
Junttila, T.T., Parsons, K., Olsson, C., Lu, Y., Xin, Y., Theriault, J., Crocker, L., Pabonan, O., Baginski, T., Meng, G., Totpal, K., Kelley, R.F., Sliwkowski, M.X.: Superior in vivo efficacy of afucosylated trastuzumab in the treatment of HER2-amplified. Breast Canc. Canc. Res. 70, 4481–4489 (2010)
Kaneko, Y., Nimmerjahn, F., Ravetch, J.V.: Anti-inflammatory activity of Immunoglobulin G resulting from Fc sialylation. Sci. 313, 670–673 (2006)
Aparna, H.S., Salimath, P.V.: Purification of an antigenic glycopeptide from buffalo colostrum. J. Food Sci. Technol. 38, 450–452 (2001)
Kanade, S.R., Rao, D.H., Hegde, R.N., Gowda, L.R.: The unique enzymatic function of field bean (Dolichos lablab) D-galactose specific lectin: a polyphenol oxidase. Glycoconj. J. 26, 535–545 (2009)
Rosenfeld, J., Capdevielle, Guillemot, C., Ferrara, P.: In-gel digestion for internal sequence analysis after one-or two-dimensional gel electrophoresis. Anal. Biochem. 203, 173–179 (1992)
Suckau, D., Resemann, A., Schuerenberg, M., Hufnagel, P., Franzen, J., Holle, A.: A novel MALDI LIFT-TOF/TOF mass spectrometer for proteomics. Anal. Bioanal. Chem. 376, 952–965 (2003)
Kita, Y., Muira, Y., Furukawa, J., Nakano, M., Shinohara, Y., Ohno, M., Takimoto, A., Nishimura, S.-I.: Quantitative glycomics of human whole serum glycoproteins based on the standardized protocol for liberating N-glycans. Mol. Cell. Proteomics 6, 1437–1445 (2007)
Furukawa, J., Shinohara, Y., Kuramoto, H., Miura, Y., Shimaoka, H., Kurogochi, M., Nakano, M., Nishimura, S.-I.: Comperhensive approach to structural and functional glycomics based on chemoselective glycoblotting and sequential tag conversion. Anal. Chem. 80, 1094–1101 (2008)
Rohit, A.C., Aparna, H.S.: Characterization of β-lactoglobulin from buffalo colostrum and its possible interaction erythrocyte lipocalin-interacting-membrane-receptor (LIMR). J. Biochem. 150, 279–288 (2011)
McKinney, M., Parkinson, A.: A simple, non-chromatographic procedure to purify immunoglobulins from serum and ascites fluid. J. Immunol. Methods 96, 271–278 (1987)
Premysl, K., Rodney, J.B., William, H.S.: Chromatographic purification of immunoglobulin G from bovine milk whey. J. Chromatograph. A 673, 45–53 (1994)
Didier, L., Alain, O.: Bovine immunoglobulin G, β-lactoglobulin, α-lactalbumin and serum albumin in colostrum and milk during the early postpartum period. J. Dairy Res. 66, 421–430 (1999)
Korhonen, H., Marnila, P., Gill, H.S.: Milk immunoglobulins and complement factors. Br. J. Nutr. 84, 75–80 (2000)
Gapper, L.W., Copestake, D.E., Otter, D.E., Indyk, H.E.: Analysis of bovine immunoglobulin G in milk, colostrum and dietary supplements a review. Anal. Bioanal. Chem. 389, 93–109 (2007)
Elke, S., Leitner, B., Ryan, M.M., Elizabeth, H.D., Farhat, K., John, W., Evelina, A.: Evaluation of immunoglobulin purification methods and their impact on qualitative and yield of antigen-specific antibodies. Malarial J. 7, 129–138 (2008)
Menegatti, S., Hussain, M., Naik, A.D., Carbonell, R.G., Rao, B.M.: mRNA display selection and solid-phase synthesis of Fc-binding cyclic peptide affinity ligands. Biotechnol. Bioeng. 110, 857–870 (2013)
Tong, F., Lin, D.Q., Yuan, X.M., Yao, S.J.: Enhancing IgG purification from serum albumin containing feedstock with hydrophobic charge-induction chromatography. J. Chromatogr. A 1244, 116–122 (2012)
Ezan, E., Bitsch, F.: Critical comparison of MS and immunoassays for the bioanalysis of therapeutic antibodies. Bioanalysis 1, 1375–1388 (2009)
Wu, H., Chen, S., Shortreed, M.R., Kreitinger, G.M., Yuan, Y., Frey, B.L., Zhang, Y., Mirza, S., Cirillo, L.A., Olivier, M., Smith, L.M.: Sequence-specific capture of protein-DNA complexes for mass spectrometric protein identification. PLoS One. 6(10):e26217 (2011)
Mesmin, C., Cholet, S., Blanchard, A., Chambon, Y., Azizi, M., Ezan, E.: Mass spectrometric quantification of AcSDKP-NH2 in human plasma and urine and comparison with an immunoassay. Rapid Commun. Mass Spectrom. 26, 163–172 (2012)
Nwosu, C.C., Aldredge, D.L., Lee, H., Lerno, L.A., Zivkovic, A.M., German, J.B., Lebrilla, C.B.: Comparison of the human and bovine milk N-glycome via high-performance microfluidic chip liquid chromatography and tandem mass spectrometry. J. Proteome Res. 11, 2912–2924 (2012)
Nongluk, S., Sachiko, K., Hirokazu, Y., Hiroaki, H., Shin-ichi, N., Keita, Y., Ito, H., Hisashi, N.J., Koichi, K., Yasuo, S.: Bovine milk whey for preparation of natural N-glycans: structural and quantitative analysis. Open Glysosci. 5, 41–50 (2012)
Takimori, S., Shimaoka, H., Furukawa, J., Yamashita, T., Amano, M., Fujitani, N., Takegawa, Y., Hammarström, L.I., Kacskovics, Shinohara, Y., Nishimura, S.: Alteration of the N-glycome of bovine milk glycoproteins during early lactation. FEBS J. 278, 3769–3781 (2011)
Robert, A., Fredrik, W., Jeffrey, V.R.: Novel roles for the Fc glycan. Ann. N.Y. Acad. Sci. 1253, 170–180 (2012)
Hodoniczky, J., Zheng, Y.Z., James, D.C.: Control of recombinant monoclonal antibody effector functions by Fc N-glycan remodeling in vitro. Biotechnol. Prog. 21, 1644–1652 (2005)
Menon, S., Rosenberg, K., Graham, S.A., Ward, E.M., Taylor, M.E., Drickamer, K.: Binding-site geometry and flexibility in DC-SIGN demonstrated with surface force measurements. Proc. Natl. Acad. Sci. U. S. A. 106, 11524–11529 (2009)
Sato, T., Takio, K., Kobata, A., Greenwalt, D.E., Furukawa, K.: Site-specific glycosylation of bovine butyrophilin. J. Biochem. 117, 147–157 (1995)
Wei, Z., Nishimura, T., Yoshida, S.: Presence of a glycan at a potential N-glycosylation site, Asn-281, of Bovine Lactoferrin. J. Dairy Sci. 83, 683–689 (2000)
Urashima, T., Taufik, E., Fukuda, K., Asakuma, S.: Recent advances in studies on milk oligosaccharides of cows and other domestic farm animals. Biosci. Biotechnol. Biochem. 77, 455–466 (2013)
Acknowledgments
HSA thanks INSA-JSPS for the award of overseas associateship and Mamatha Bhanu thanks University Post-graduate cell for the award of fellowship. Authors also thank protemics facility, IISc, Bangalore.
Conflict of interest
The authors declare no commercial or financial conflict of interest.
Funding
This work was supported by the Indian National Science Academy (INSA) and the Japan Society for the Promotion of Science (JSPS) fund.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 658 kb)
Rights and permissions
About this article
Cite this article
Bhanu, L.S.M., Amano, M., Nishimura, SI. et al. Glycome characterization of immunoglobulin G from buffalo (Bubalus bubalis) colostrum. Glycoconj J 32, 625–634 (2015). https://doi.org/10.1007/s10719-015-9608-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-015-9608-4