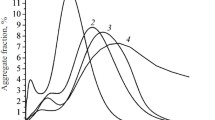
The mechanochemical synthesis of calcium ferrite from iron oxalate FeC2O4 ∙ 2H2O and calcium hydroxide Ca(OH)2 is studied by means of x-ray phase, x-ray diffraction, and simultaneous thermal analysis and scanning electron microscopy. The optimal conditions for obtaining calcium ferrite are determined. It is shown that the use of mechanochemical synthesis can significantly reduce the heat treatment temperature and duration.





Similar content being viewed by others
References
Yu. Sitidze and H. Sato, Ferrites [Russian translation], Mir, Moscow (1964).
N. Ikenaga, Y. Ohgaito, and T. Suzuki, “H2S absorption behavior of calcium ferrite prepared in the presence of coal,” Energy & Fuels, 19(1), 170 – 179 (2005).
R. A. Candeia, M. I. B. Bernardi, E. Longo, et al., “Synthesis and characterization of spinel pigment CaFe2O4 obtained by the polymeric precursor method,” Mater. Lett., 58(5), 569 – 572 (2004).
R. N. Rumyantsev, S. P. Kochetkov, M. A. Lapshin, et al., “Effect of copper and calcium ferrites on the activity and selectivity of the catalysts of medium temperature CO conversion,” Izv. Vyssh. Uchebn. Zaved., Khim. Khimich. Tekhnol., 59(3), 43 – 48 (2016).
R. N. Rumyantsev, A. A. Ilyin, I. V. Babichev, and A. P. Ilyin, “Decomposition of nitric acid (I) on the ferrite with different crystal structures,” SITA-Journal, 16(3), 1 – 5 (2014).
D. Hirabayashi and T. Yoshikawa, “Formation of brownmillerite type calcium ferrite (Ca2Fe2O5) and catalytic properties in propylene combustion,” Catalysis Lett., 110(3), 26 – 274 (2006).
L. A. Isupova, S. V. Tsibulya, G. N. Kryukov, et al., “Mechanochemical synthesis and catalytic properties of calcium ferrite Ca2Fe2O5,” Kinetika Kataliz, 43(1), 132 – 139 (2002).
B. F. Amorim and M. A. Morales, “Synthesis of stoichiometric Ca2Fe2O5 nanoparticles by high-energy ball milling and thermal annealing,” Physica B: Condensed Matter., 488, 43 – 48 (2016).
A. A. Ilyin, A. P. Ilyin, V. Yu. Kurochkin, and N. N. Smirnov, “Mechanochemical synthesis of the catalyst for medium-temperature conversion of carbon monoxide with water vapor,” Usp. Khim. Khim. Tekhnol., 21[9(77)], 79 – 83 (2007).
R. N. Rumyantsev, A. P. Ilyin, A. A. Ilyin AA, and N. N. Smirnov, “Mechanochemical synthesis of iron oxides, ferrites, and catalysts based on them,” in: V. I. Svetsov (ed.), R&D in Nanotechnologies – 2013 [in Russian], Ivanovo State Chemical Technological University, Ivanovo (2013), pp. 114 – 134.
E. G. Avvakumov, N. V. Kosovo, and M. V. Chaikin, “Regularities and prospects of ‘soft’ mechano-chemical synthesis,” in: Reports at the International Scientific Seminar on Mechanochemistry and Mechanical Activation [in Russian], St. Petersburg (1995), pp. 82 – 83.
V. V. Boldyrev, E. V. Avvakumov, E. V. Boldyrev, et al., Fundamentals of Mechanical Activation, Mechano-Synthesis, and Mechanochemical Technologies [in Russian], SO RAN, Novosibirsk (2009).
E. G. Avvakumov, “Mechanochemical synthesis as a method of obtaining nano-dispersed particles of oxide materials,” Khim. v Interesakh Ustoich. Razvitiya, 12(3), 287 – 292 (2004).
H. Heegn, “Change in the properties of solids under mechanical activation and solid comminution,” Izv. Sib. Otd. Akad. Nauk SSSR, Ser. Khim. Nauk, 1(2), 3 – 9 (1984).
E. G. Avvakumov and M. Senna, Soft Mechanochemical Synthesis: a Basis for New Chemical Technologies, Springer Science & Business Media, N.Y. (2001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Steklo i Keramika, No. 10, pp. 24 – 28, October, 2016.
Rights and permissions
About this article
Cite this article
Rumyantsev, R.N., Il’in, A.A., Denisova, K.O. et al. Calcium Ferrite Structure Formation During Mechanochemical Interaction in the System FeC2O4 ∙ 2H2O–Ca(OH)2 . Glass Ceram 73, 374–377 (2017). https://doi.org/10.1007/s10717-017-9892-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10717-017-9892-5