Abstract
Background
Associating liver partition and portal vein ligation (ALPPS) is a promising two-step hepatectomy that is beneficial for accumulative regeneration of the future liver remnant (FLR) and avoids postoperative liver failure.
Aims
Our study aimed to evaluate whether nonalcoholic fatty liver disease affected the liver regeneration induced by ALPPS.
Methods
Sprague-Dawley rats fed a high-fat diet were used to construct the NAFLD model. ALPPS were performed, and blood and future liver remnant samples were collected at postoperative days 1 (POD1), POD3, and POD7.
Results
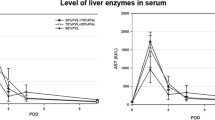
The hepatic regeneration rate (HRR) of ALPPS was higher than that of portal vein ligation (PVL) at POD3 and POD7 (p < 0.05), and the number of Ki-67-positive hepatocytes (POD3) and CD68-positive Kupffer cells (POD7) per visual field was higher in the ALPPS group than in the PVL group (p < 0.05). The serum TNF-α, hepatocyte growth factor protein, and the serum IL-6 level were higher in the ALPPS group than in the PVL group at POD3 and POD7. Compared with those of the standard laboratory diet (SLD)-fed rats, the rats with NAFLD exhibited a decrease in the HRR, Ki-67-positive hepatocytes, and CD68-positive Kupffer cells in the FLR. The number of CD68-positive Kupffer cells was lower in rats with NAFLD than that in SLD-fed rats; noteworthily, the serum level of IL-6 and TNF-α changed dramatically after surgeries.
Conclusions
NAFLD induction delayed liver regeneration induced by the ALPPS procedure, which might be associated with hepatocyte proliferation and the number of Kupffer cells.









Similar content being viewed by others
Abbreviations
- FLR:
-
Portal vein ligation
- SLD:
-
Standard laboratory diet
- HGF:
-
Hepatocyte growth factor
- LML:
-
Left median lobe
- LLL:
-
Left lateral lobe
- PLF:
-
Postoperative liver failure
- PBE:
-
Portal branch embolization
- TCHO:
-
Total cholesterol
- ALPPS:
-
Associating liver partition and portal vein ligation
- NAFLD:
-
Nonalcoholic fatty liver disease
References
Clavien PA, Oberkofler CE, Raptis DA, Lehmann K, Rickenbacher A, El-Badry AM. What is critical for liver surgery and partial liver transplantation: size or quality? Hepatology. 2010;52:715–729.
Rahbari NN, Garden OJ, Padbury R, et al. Posthepatectomy liver failure: a definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery. 2011;149:713–724.
Schnitzbauer AA, Lang SA, Goessmann H, et al. Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Ann Surg. 2012;255:405–414.
Makuuchi M, Takayasu K, Takuma T, et al. Preoperative transcatheter embolization of the portal venous branch for patients receiving extended lobectomy due to the bile duct carcinoma. J Jpn Pract Surg Soc. 1984;45:1558–1564.
Budai A, Fulop A, Hahn O, et al. Animal models for associating liver partition and portal vein ligation for staged hepatectomy (ALPPS): achievements and future perspectives. Eur Surg Res. 2017;58:140–157.
Bellentani S. The epidemiology of non-alcoholic fatty liver disease. Liver Int. 2017;37:81–84.
Baffy G, Brunt EM, Caldwell SH. Hepatocellular carcinoma in non-alcoholic fatty liver disease: an emerging menace. J Hepatol. 2012;56:1384–1391.
White DL, Kanwal F, El-Serag HB. Association between nonalcoholic fatty liver disease and risk for hepatocellular cancer, based on systematic review. Clin Gastroenterol Hepatol. 2012;10:1342–1359.
Picard C, Lambotte L, Starkel P, et al. Steatosis is not sufficient to cause an impaired regenerative response after partial hepatectomy in rats. J Hepatol. 2002;36:645–652.
Rao MS, Papreddy K, Abecassis M, Hashimoto T. Regeneration of liver with marked fatty change following partial hepatectomy in rats. Dig Dis Sci. 2001;46:1821–1826. https://doi.org/10.1023/A:1010654908938.
Garnol T, Kucera O, Stankova P, Lotkova H, Cervinkova Z. Does simple steatosis affect liver regeneration after partial hepatectomy in rats? Acta Med (Hradec Kralove). 2016;59:35–42.
Yamauchi H, Uetsuka K, Okada T, Nakayama H, Doi K. Impaired liver regeneration after partial hepatectomy in db/db mice. Exp Toxicol Pathol. 2003;54:281–286.
DeAngelis RA, Markiewski MM, Taub R, Lambris JD. A high-fat diet impairs liver regeneration in C57BL/6 mice through overexpression of the NF-kappaB inhibitor, IkappaBalpha. Hepatology. 2005;42:1148–1157.
Murata H, Yagi T, Iwagaki H, et al. Mechanism of impaired regeneration of fatty liver in mouse partial hepatectomy model. J Gastroenterol Hepatol. 2007;22:2173–2180.
Veteläinen RVVAV. Severe steatosis increases hepatocellular injury and impairs liver regeneration in a rat model of partial hepatectomy. Ann Surg. 2007;1:44–50.
Hamano M, Ezaki H, Kiso S, et al. Lipid overloading during liver regeneration causes delayed hepatocyte DNA replication by increasing ER stress in mice with simple hepatic steatosis. J Gastroenterol. 2014;49:305–316.
Neuschwander-Tetri BA, Caldwell SH. Nonalcoholic steatohepatitis: summary of an AASLD Single Topic Conference. Hepatology. 2003;37:1202–1219.
Schlegel A, Lesurtel M, Melloul E, et al. ALPPS: from human to mice highlighting accelerated and novel mechanisms of liver regeneration. Ann Surg. 2014;260:839–847.
Yao L, Li C, Ge X, et al. Establishment of a rat model of portal vein ligation combined with in situ splitting. Plos One. 2014;9:e105511.
Shi H, Yang G, Zheng T, et al. A preliminary study of ALPPS procedure in a rat model. Sci Rep. 2015;5:17567.
Wei W, Zhang T, Zafarnia S, et al. Establishment of a rat model: associating liver partition with portal vein ligation for staged hepatectomy. Surgery. 2016;159:1299–1307.
García-Pérez R, Revilla-Nuin B, Martínez CM, et al. Associated liver partition and portal vein ligation (ALPPS) versus selective portal vein ligation (PVL) for staged hepatectomy in a rat model Similar regenerative response? PLoS One. 2015;10:e144096.
Almau TH, Moulin LE, Padin JM, Stringa P, Gondolesi GE, Barros SP. Development of an experimental model of portal vein ligation associated with parenchymal transection (ALPPS) in rats. Cir Esp. 2014;92:676–681.
Dhar DK, Mohammad GH, Vyas S, et al. A novel rat model of liver regeneration: possible role of cytokine induced neutrophil chemoattractant-1 in augmented liver regeneration. Ann Surg Innov Res. 2015;9:11.
Michalopoulos GK. Liver regeneration after partial hepatectomy: critical analysis of mechanistic dilemmas. Am J Pathol. 2010;176:2–13.
Moris D, Vernadakis S, Papalampros A, et al. Mechanistic insights of rapid liver regeneration after associating liver partition and portal vein ligation for stage hepatectomy. World J Gastroenterol. 2016;22:7613–7624.
Nishiyama K, Nakashima H, Ikarashi M, et al. Mouse CD11b+ Kupffer cells recruited from bone marrow accelerate liver regeneration after partial hepatectomy. Plos One. 2015;10:e136774.
Shiratori Y, Hongo S, Hikiba Y, et al. Role of macrophages in regeneration of liver. Dig Dis Sci. 1996;41:1939–1946. https://doi.org/10.1007/BF02093593.
Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology. 2006;43:S45–S53.
Cressman DE, Greenbaum LE, DeAngelis RA, et al. Liver failure and defective hepatocyte regeneration in interleukin-6-deficient mice. Science. 1996;274:1379–1383.
Aldeguer X, Debonera F, Shaked A, et al. Interleukin-6 from intrahepatic cells of bone marrow origin is required for normal murine liver regeneration. Hepatology. 2002;35:40–48.
Wang J, Kubes P. A reservoir of mature cavity macrophages that can rapidly invade visceral organs to affect tissue repair. Cell. 2016;165:668–678.
Hsiao IT, Lin KJ, Chang SI, Yen TC, Chen TC, Yeh TS. Impaired liver regeneration of steatotic rats after portal vein ligation: a particular emphasis on (99 m)Tc-DISIDA scintigraphy and adiponectin signaling. J Hepatol. 2010;52:540–549.
Acknowledgments
This work was supported by the Fund for Science and Technology Development of Jilin Province (20160101060JC, 20150101108JC); the National Key R&D Program of China (2017YFD0502200, 2016YFD0501302); the Project of the Education Department of Jilin Province (No. 2016444).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Zhao, J., Xu, H., Li, Y. et al. NAFLD Induction Delays Postoperative Liver Regeneration of ALPPS in Rats. Dig Dis Sci 64, 456–468 (2019). https://doi.org/10.1007/s10620-018-5346-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-5346-3