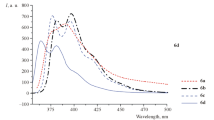
Methods are presented for the preparation of isomeric (Е)-3-, (Е)-6-, (Е)-7-[4-(dimethylamino)phenylethenyl]quinoxalin-2-ones and 2-phenylquinoxalines – compounds of “donor–π-bridge” type, which serve as precursors for new nonlinear optical chromophores with potentially high first hyperpolarizability values. The introduction of dimethylanilinoethenyl moiety at position 3 of the quinoxaline system was achieved in good yields by fusion of 3-methyl derivatives with 4-(dimethylamino)benzaldehyde at 220°С in the presence of catalytic amounts of acetic anhydride and pyridine during the synthesis of 3-(dimethylaminophenylethenyl)quinoxalin-2-ones or as a result of condensation of these reactants by the action of 20 М sodium hydroxide solution in the presence of Aliquat 336 during the synthesis of 3-(dimethylaminophenylethenyl)-2-phenylquinoxalines. The introduction of dimethylanilinoethenyl moiety at positions 6 and 7 was achieved by Heck reaction of p-dimethylaminovinylbenzene with 6- or 7-bromoquinoxalines in the presence of palladium acetate. The structures of isomeric 6-bromo- and 7-bromo-3-methyl derivatives of quinoxalines were confirmed by X-ray diffraction analysis.



Similar content being viewed by others
References
(a) Guenter, P. Nonlinear Optical Effects and Materials; Springer, 2002, p. 495. (b) Dalton, L. R.; Sullivan, P. A.; Bale, D. H. Chem. Rev. 2010, 110, 25. (c) Sharipova, S. M.; Kalinin, A. A. Chem. Heterocycl. Compd. 2017, 53, 36. [Khim. Geterotsikl. Soedin. 2017, 53, 36.]
(a) Cheng, Y.-J.; Luo, J.; Hau, S.; Bale, D. H.; Kim, T.-D.; Shi, Z.; Lao, D. B.; Tucker, N. M.; Tian, Y.; Dalton, L. R.; Reid, P. J.; Jen, A. K.-Y. Chem. Mater. 2007, 19, 1154. (b) Quist, F.; Vande Velde, C. M. L.; Didier, D.; Teshome, A.; Asselberghs, I.; Clays, K.; Sergeyev, S. Dyes Pigm. 2009, 81, 203. (c) Yang, Y.; Liu, F.; Wang, H.; Zhang, M.; Xu, H.; Bo, S.; Liu, J.; Qiu, L.; Zhena, Z.; Liu, X. Phys. Chem. Chem. Phys. 2014, 16, 20209. (d) Wu, J.; Peng, C.; Xiao, H.; Bo, S.; Qiu, L.; Zhen, Z.; Liu, X. Dyes Pigm. 2014, 104, 15. (e) Wang, H.; Liu, F.; Yang, Y.; Xu, H.; Peng, C.; Bo, S.; Zhen, Z.; Liu, X.; Qiu, L. Dyes Pigm. 2015, 112, 42.
(a) Hammond, S. R.; Clot, O.; Firestone, K. A.; Bale, D. H.; Lao, D.; Haller, M.; Phelan, G. D.; Carlson, B.; Jen, A. K.-Y.; Reid, P. J.; Dalton, L. R. Chem. Mater. 2008, 20, 3425. (b) Akelaitis, A. J. P.; Olbricht, B. C.; Sullivan, P. A.; Liao, Y.; Lee, S. K.; Bale, D. H.; Lao, D. B.; Kaminsky, W.; Eichinger, B. E.; Choi, D. H.; Reid, P. J.; Dalton, L. R. Opt. Mater. 2008, 30, 1504. (c) Hammond, S. R.; Sinness, J.; Dubbury, S.; Firestone, K. A.; Benedict, J. B.; Wawrzak, Z.; Clot, O.; Reida, P. J.; Dalton, L. R. J. Mater. Chem. 2012, 22, 6752.
(a) Marco, A. B.; Andreu, R.; Franco, S.; Garin, J.; Orduna, J.; Villacampa, B.; Diosdado, B. E.; Lopez Navarrete, J. T.; Casado, J. Org. Biomol. Chem. 2013, 11, 6338. (b) Zhang, A.; Xiao, H.; Cong, S.; Zhang, M.; Zhang, H.; Bo, S.; Wang, Q.; Zhen, Z.; Liu, X. J. Mater. Chem. C. 2015, 2, 370.
Cho, M. J.; Kim, J. Y.; Kim, J. H.; Lee, S. H.; Dalton, L. R.; Choi, D. H. Bull. Korean Chem. Soc. 2005, 26, 77.
(a) Ashraf, M.; Teshome, A.; Kay, A. J.; Gainsford, G. J.; Bhuiyan, M. D. H.; Asselberghs, I.; Clays, K. Dyes Pigm. 2013, 98, 82. (b) Wu, J.; Bo, S.; Liu, J.; Zhou, T.; Xiao, H.; Qiu, L.; Zhen, Z.; Liu, X. Chem. Commun. 2012, 48, 9637. (c) Levitskaya, A. I; Kalinin, A. A.; Fominykh, O. D.; Vasilyev, I. V.; Balakina, M. Yu. Comput. Theor. Chem. 2016, 1094, 17.
Goikhman, M. Ya.; Subbotina, L. I.; Martynenkov, A. A.; Smirnov, M. A.; Smyslov, R. Yu.; Popova, E. N.; Yakimanskii, A. V. Russ. Chem. Bull., Int. Ed. 2011, 60, 295. [Izv. Akad. Nauk, Ser. Khim. 2011, 288.]
Levitskaya, A. I.; Kalinin, A. A.; Fominykh, O. D.; Balakina, M. Yu. Comput. Theor. Chem. 2015, 1074, 91.
(a) Johnson, L. E.; Dalton, L. R.; Robinson, B. H. Acc. Chem. Res. 2014, 47, 3258. (b) Isborn, C. M.; Leclercq, A.; Vila, F. D.; Dalton, L. R.; Bredas, J. L.; Eichinger, B. E.; Robinson, B. H. J. Phys Chem. A 2007, 111, 1319.
(a) Negwer, M; Scharnow, H. G. Organic Chemical Drugs and their Synonyms; Wiley-VCH: Weinheim, 2001. (b) Mamedov, V. A.; Kalinin, A. A. Russ. Chem. Rev. 2014, 83, 820. [Usp. Khim. 2014, 83, 820.]
(a) Mamedov, V. A.; Kalinin, A. A. Chem. Heterocycl. Compd. 2010, 46, 641. [Khim. Geterotsikl. Soedin. 2010, 803.] (b) Mamedov, V. A.; Kalinin, A. A.; Zhukova, N. A.; Syakaev, V. V.; Rizvanov, I. Kh.; Latypov, S. K.; Sinyashin, O. G. Tetrahedron 2015, 71, 147. (c) Kalinin, A. A.; Voloshina, A. D.; Kulik, N. V.; Zobov, V. V.; Mamedov, V. A. Eur. J. Med. Chem. 2013, 66, 345.
(a) Mamedov, V. A.; Kalinin, A. A.; Gubaidullin, A. T.; Litvinov, I. A.; Levin, Y. A. Chem. Heterocycl. Compd. 2002, 38, 1504. [Khim. Geterotsikl. Soedin. 2002, 1704.] (b) Mamedov, V. A.; Kalinin, A. A.; Gubaidullin, A. T.; Isaikina, O. G.; Litvinov, I. A. Russ. J. Org. Chem. 2005, 41, 599. [Zh. Org. Khim. 2005, 41, 609.]
Mamedov, V. A.; Murtazina, A. M. Russ. Chem. Rev. 2011, 80, 397. [Usp. Khim. 2011, 80, 419.]
(a) Mamedov, V. A.; Kalinin, A. A. In Advances in Heterocyclic Chemistry; Katritzky, A. R., Ed.; Academic Press, 2014, Vol. 112, p. 51. (b) Kalinin, A. A.; Mamedov, V. A. Chem. Heterocycl. Compd. 2014, 50, 195. [Khim. Geterotsikl. Soedin. 2014, 219.] (c) Mamedov, V. A.; Kalinin, A. A.; Gubaidullin, A. T.; Katsuba, S. A.; Syakaev, V. V.; Rizvanov, I. K.; Latypov, Sh. K. Tetrahedron 2013, 69, 10675. (d) Singh, N. J.; Jun, E. J.; Chellappan, K.; Thangadurai, D.; Chandran, R. P.; Hwang, I. C.; Yoon, J.; Kim, K. S. Org. Lett. 2007, 9, 485.
(а) Yang, Y.; Liu, J.; Xiao, H.; Zhen, Z.; Bo, S. Dyes Pigm. 2017, 139, 239. (b) Piao, X.; Zhang, X.; Inoue, S.; Yokoyama, S.; Aoki, I.; Miki, H.; Otomo, A.; Tazawa, H. Org. Electron. 2011, 12, 1093.
(а) Krishnan, V. S. H.; Chowdary, K. S.; Dubey, P. K.; Vijaya, S. Indian J. Chem. 2001, 40B, 565. (b) Benzeid, H.; Mothes, E.; Essassi, El Mokhtar; Faller, P.; Pratviel, G. C. R. Chimie. 2012, 15, 79. (c) Sain, D. Kr.; Thadhaney, B.; Joshi, A.; Hussain, N.; Talesara, G.L. Indian J. Chem. B. 2010, 49, 818. (d) Cazaux, L.; Faher, M.; Picard, C.; Tisnès, P. Can. J. Chem. 1993, 71, 1236. (е) Mamedov, V. A.; Kadyrova, S. F.; Zhukova, N. A.; Galimullina, V. R.; Polyancev, F. M.; Latypov, Sh. K. Tetrahedron 2014, 70, 5934.
Achelle, S.; Barsella, A.; Baudequin, C.; Caro, B.; Robin-le Guen, F. J. Org. Chem. 2012, 77, 4087.
Cho, M. J.; Kim, J. Y.; Kim, J. H.; Lee, S. H.; Dalton, L. R.; Choi, D. H. Bull. Korean Chem. Soc. 2005, 26, 77.
Bruker. APEX2 Software Suite for Crystallographic Programs; Bruker AXS, Inc.: Madison, 2009.
Bruker. Area detector control and integration software. Version 6.0. In SMART and SAINT; Bruker Analytical X-ray Instruments Inc.: Madison, 2003.
Sheldrick, G. M. SADABS. Program for absorption correction; University of Göttingen, Institut für Anorganische Chemie: Göttingen, 1997.
Sheldrick, G. M. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, A64, 112.
Macrae, C. F.; Edgington, P. R.; McCabe, P.; Pidcock, E.; Shields, G. P.; Taylor, R.; Towler, M.; van de Streek, J. J. Appl. Crystallogr. 2006, 39, 453.
Chen, Y.; Li, K.; Zhao, M.; Li, Y.; Chen, B. Tetrahedron Lett. 2013, 54, 1627.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2017, 53(5), 504–510
Rights and permissions
About this article
Cite this article
Sharipova, S.M., Gilmutdinova, A.A., Krivolapov, D.B. et al. Synthesis of isomeric (E)-[4-(dimethylamino)phenyl]-vinylquinoxalines – precursors for a new class of nonlinear optical chromophores. Chem Heterocycl Comp 53, 504–510 (2017). https://doi.org/10.1007/s10593-017-2084-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-017-2084-y