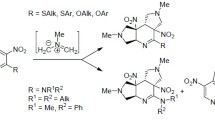
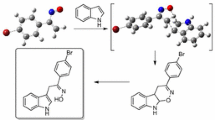
The reactions of 4-chloro-5,7-dinitro-4-benzofurazan with indole and pyrrole derivatives, occurring by SNAr–SEAr mechanism, led to the formation of dihetaryls with intramolecular charge transfer. A method was developed for the annelation of pyrrole and dihydropyrrole ring to nitrobenzofurazan fragment by adding unstabilized azomethine ylide to the С=С bond of dinitrobenzofurazan. The structure of nitrobenzofurazan derivatives was studied by X-ray structural analysis, NMR spectroscopy, and quantum-chemical calculations using ab initio and DFT methods.




Similar content being viewed by others
References
L. I. Hmelnitskii, S. S. Novikov, and T. I. Godovikova, Chemistry of Furoxans. Reactions and Applications [in Russian], Nauka, Moscow (1996), p. 430.
F. Terrier, in: Organic Reactivity: Physical and Biological Aspects, Royal Society of Chemistry, Cambridge (1995), p. 399.
F. Terrier, A. P. Chatrousse, Y. Soudais, and M. Hlaibi, J. Org. Chem., 49, 4176 (1984).
F. Terrier, M. J. Pouet, J. C. Halle, E. Kizilian, and E. Buncel, J. Phys. Org. Chem., 11, 707 (1998).
R. Goumont, E. Kizilian, E. Buncel, and F. Terrier, Org. Biomol. Chem., 1, 1741 (2003).
S. V. Kurbatov, Z. N. Budarina, G. S. Vaslyaeva, N. I. Borisenko, A. P. Knyazev, V. I. Minkin, Yu. A. Zhdanov, and L. P. Olekhnovich, Russ. Chem. Bull., Int. Ed., 46, 1445 (1997). [Izv. Akad. Nauk, Ser. Khim., 1509 (1997).]
V. A. Voronina, A. E. Frumkin, S. V. Kurbatov, A. M. Churakov, O. Yu. Smirnov, and L. P. Olekhnovich, Russ. Chem. Bull., Int. Ed., 51, 668 (2002). [Izv. Akad. Nauk, Ser. Khim., 617 (2002).]
P. G. Morozov, S. V. Kurbatov, F. M. Dolgushin, M. Yu. Antipin, and L. P. Olekhnovich, Russ. Chem. Bull., Int. Ed., 53, 2075 (2004). [Izv. Akad. Nauk, Ser. Khim., 1990 (2004).]
A. V. Tkachuk, S. V. Kurbatov, O. N. Burov, M. E. Kletskii, Yu. P. Tavunova, P. G. Morozov, V. A. Voronina, and V. I. Minkin, Russ. J. Org. Chem., 49, 1373 (2013). [Zh. Org. Khim., 49, 1388 (2013).]
V. I. Minkin, A. V. Tkachuk, M. E. Kletskii, D. V. Steglenko, V. A. Voronina, and S. V. Kurbatov, Russ. Chem. Bull., Int. Ed., 62, 464 (2013). [Izv. Akad. Nauk, Ser. Khim., 464 (2013).]
S. Kurbatov, P. Rodriguez-Dafonte, R. Goumont, and F. Terrier, Chem. Commun., 2150 (2003).
S. Kurbatov, A. Tatarov, V. Minkin, R. Goumont, and F. Terrier, Chem. Commun., 4279 (2006).
A. Tatarov, S. Kurbatov, G. Borodkin, R. Goumont, and F. Terrier, Tetrahedron, 66, 995 (2010).
F. Terrier, Modern Nucleophilic Aromatic Substitution, Wiley-VCH (2013), 488 p.
E. Buncel and F. Terrier, Org. Biomol. Chem., 8, 2285 (2010).
F. Terrier, J. M. Dust, and E. Buncel, Tetrahedron, 68, 1829 (2012).
S. Kurbatov, S. Lakhdar, R. Goumont, and F. Terrier, Org. Prep. Proced. Int., 44, 289 (2012).
S. A. Shevelev and A. M. Starosotnikov, Chem. Heterocycl. Compd., 49, 92 (2013). [Khim. Geterotsikl. Soedin., 102 (2013).]
C. Boga, E. Del Vecchio, L. Forlani, A. Mazzanti, and P. E. Todesco, Angew. Chem., Int. Ed., 44, 3285 (2005).
E. Pretsch, F. Bühlmann, and K. Affolter, Structure Determination of Organic Compounds [Russian translation], Mir, Moscow (2006), p. 438.
B. P. Nikolskii (editor), Chemist's Handbook [in Russian], Vol. 1, Khimiya, Moscow–Leningrad (1982), p. 352.
C. K. Prout, O. J. R. Hodder, and D. Viterbo, Acta Crystallogr., Sect. B.: Struct. Crystallogr. Cryst. Chem., B28, 1523 (1972).
H. Cerecetto and W. Porcal, Mini Rev. Med. Chem., 5, 57 (2005).
G. P. Sharnin, F. S. Levinson, S. A. Akimova, and R. Kh. Khasanov, USSR Inventor's Certificate 627129; Byul. Izobret., No. 37 (1978).
G. M. Sheldrick, SHELXTL, v. 5.10, Structure Determination Software Suite, Bruker AXS, Madison (1998).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, Jr., T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, and J. A. Pople, Gaussian 03, Revision D.01, Gaussian, Inc., Wallingford (2004).
D. V. Steglenko, M. E. Kletsky, S. V. Kurbatov, A. V. Tatarov, V. I. Minkin, R. Goumont, and F. Terrier, J. Phys. Org. Chem., 22, 298 (2009).
The quantum-chemical calculations and resonance spectroscopy studies of the structure and properties of nitrobenzofurazan derivatives were performed with support from the Russian Science Foundation (project No. 14-13-00103).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1881-1891, December, 2014.
Rights and permissions
About this article
Cite this article
Semenyuk, Y.P., Kochubei, A.S., Morozov, P.G. et al. [3+2] Cycloaddition Reactions to Indolyl- and Pyrrolyl Derivatives of Dinitrobenzofurazan. Chem Heterocycl Comp 50, 1731–1740 (2015). https://doi.org/10.1007/s10593-015-1645-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-015-1645-1