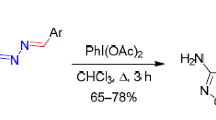
The reduction of 3,4-bis(4-R-furazan-3-yl)furoxans with hydrazine or hydrogen in the presence of palladium on carbon resulted in opening of the furoxan ring and formation of (Z,E)-dioximes of 1,2-di(4-R-furazan-3-yl)ethane-1,2-diones in high yield. The reactivity of the resultant glyoximes was studied.


Similar content being viewed by others
References
V. G. Andrianov and A. V. Eremeev, Khim. Geterotsikl. Soedin., 1155 (1984). [Chem. Heterocycl. Compd., 20, 937 (1984)].
R. M. Paton, in: A. R. Katritzky, C. W. Rees, and E. F. V. Scriven (editors), Comprehensive Heterocyclic Chemistry, Vol. 6, Pergamon Press, Oxford (1984), p. 393.
V. G. Andrianov and A. V. Eremeev, Khim. Geterotsikl. Soedin., 1443 (1990). [Chem. Heterocycl. Compd., 30, 1199 (1990)].
R. M. Paton, in: A. R. Katritzky, C. W. Rees, and E. F. V. Scriven (editors), Comprehensive Heterocyclic Chemistry II, Vol. 4, Pergamon Press, Oxford (1996), p. 229.
L. I. Khmel'nitskii, S. S. Novikov, and T. I. Godovikova, Chemistry of Furoxans: Structure and Synthesis [in Russian], Nauka, Moscow (1996).
L. I. Khmel'nitskii, S. S. Novikov, and T. I. Godovikova, Chemistry of Furoxans: Reactions and Applications [in Russian], Nauka, Moscow (1996).
A. B. Sheremetev, Rossiisk. Khim. Zh., 43 (1997). [Mendeleev Chem. J., 41, 62 (1997)].
A. B. Sheremetev, Usp. Khim., 68, 154 (1999). [Russ. Chem. Rev., 68, 137 (1999)].
A. B. Sheremetev, N. N. Makhova, and W. Friedrichsen, in: Advances in Heterocyclic Chemistry, Vol. 78, Academic Press, New York (2001), p. 65.
A. B. Sheremetev and I. L. Yudin, Usp. Khim., 72, 93 (2003). [Russ. Chem. Rev., 72, 87 (2003)].
G. Nikonov and S. Bobrov, in: A. R. Katritzky, C. A. Ramsden, E. F. V. Scriven, and R. J. K. Taylor (editors), Comprehensive Heterocyclic Chemistry III, Vol. 5, Elsevier Science, Amsterdam-London (2008), p. 315.
A. V. Sheremetev, E. A. Ivanova, N. P. Spiridonova, S. F. Melnikova, I. V. Tselinsky, K. Yu. Suponitsky, and M. Yu. Antipin, J. Heterocycl. Chem., 42, 1237 (2005).
Z. Feng-qi, C. Pei, H. Rong-zu, L. Yang, Z. Zhi-zhong, Z. Yan-shui, Y.Xu-wu, G. Yin, G. Sheng-li, and S. Qi-zhen, J. Hazard. Mater., 113, 67 (2004).
W. Jun, D. Haishan, H. Y. Gang, and L. J. Shan, in: Proceedings of the 11 th Seminar: New Trends in Research of Energetic Materials, Pardubice, Czech Republic, April 09–11, 2008, p. 182.
A. A. Astrat'ev, D. V. Dashko, and A. I. Stepanov, Vestn. Kazan. Tekhnol. Univ., No. 21, 35 (2011).
A. A. Astrat'ev, D. V. Dashko, and A. I. Stepanov, Cent. Eur. J. Chem., 10, 1087 (2012).
A. I. Stepanov, A. A. Astrat'ev, D. V. Dashko, N. P. Spiridonova, S. F. Mel'nikova, and I. V. Tselinsky, Izv. Akad. Nauk, Ser. Khim., 1019 (2012). [Russ. Chem. Bull., 61, 1024 (2012)].
T. W. Greene and P. G. M. Wuts, Protective Groups in Organic Synthesis, Wiley, Toronto (1999), p. 355.
S. R. Sandier and W. Karo, Organic Functional Group Preparations, Academic Press, San Diego (1989), p. 431.
A. I. Mikhaleva, A. B. Zaitsev, and B. A. Trofimov, Usp. Khim., 75, 884 (2006). [Russ. Chem. Rev., 75, 797 (2006)].
H. M. S. Kumar, B. V. S. Reddy, P. T. Reddy, and J. S. Yadav, Synthesis, 586 (1999).
B. Das, P. Madhusudhan, and B. Venkataiah, Synlett, 1569 (1999).
A. I. Bosch, P. de la Cruz, E. Diez-Barra, A. Loupy, and F. Langa, Synlett, 1259 (1995).
C. Ramalingan and Y.-T. Park, J. Org. Chem., 72, 4536 (2007).
Y. Furuya, K. Ishihara, and H. Yamamoto, J. Am. Chem. Soc., 127, 11240 (2005).
P. R. Dave, F. Forohar, T. Axenrod, K. K. Das, L. Qi, C. Watnick, and H. Yazdekhasti, J. Org. Chem., 61, 8897 (1996).
F. P. Ballisteri, E. Barbuzzi, G. A. Tomaselli, and R. M. Toscano, Synlett, 1093 (1996).
S. Sasatani, T. Miyazaki, K. Maruoka, and H. Yamamoto, Tetrahedron Lett., 24, 4711 (1983).
S. Negi, M. Matsukura, M. Mizuno, K. Miyake, and N. Minami, Synthesis, 991 (1996).
P. A. S. Smith and S. E. Gloyer, J. Org. Chem., 40, 2508 (1975).
K. Narasaka, Pure Appl. Chem., 75, 19 (2003).
V. M. Peshkova, V. M. Savostina, and E. K. Ivanova, Oximes [in Russian], Nauka, Moscow (1977).
B.-A. Song, X.-H. Liu, S. Yang, D.-Y. Hu, L.-H. Jin, and Y.-T. Zhang, Chin. J. Org. Chem., 25, 507 (2005).
G. W. A. Milne, CRC Handbook of Pesticides, CRC Press, Boca Raton (1995).
F. Sevgi and A. D. Bedük, World Appl. Sci. J., 19, 192 (2012).
J. Kassa, K. Kuca, J. Karasova, and K. Musilek, Mini-Rev. Med. Chem., 8, 1134 (2008).
E. Ābele and E. Lukevics, Org. Prep. Proced. Int., 32, 235 (2000).
P. V. Anokina, T. V. Romanova, S. F. Mel'nikova, and I. V. Tselinsky, Zh. Org. Khim., 47, 1575 (2011). [Russ. J. Org. Chem., 47, 1606 (2011)].
S. Gao and S. W. Ng, Acta Crystallogr., Sect. E: Struct. Rep. Online, 68, o1573 (2012).
A. A. Astrat'ev, S. F. Mel'nikova, S. A. Dushenok, A. A. Kotomin, D. V. Dashko, A. I. Stepanov, O. F. Yakovleva, A. S. Kozlov, D. A. Barannik, L. A. Loskutova, and I. M. Chernega, International Conference "Shock Waves in Condensed Matter" [in Russian], Kiev, 16–21 September, 2012, Kiev (2012), p. 380.
P. Pagoria, M. Zhang, A. DeHope, G. Lee, A. Mitchell, and P. Leonard, in: Proceedings of the 15 th Seminar on New Trends in Research of Energetic Materials (NTREM), Pardubice, Czech Republic, April 18–20, 2012, p. 54.
T. Pasinszki, B. Havasi, B. Hajgató, and N. P. C. Westwood, J. Phys. Chem. A, 113, 170 (2009).
T. K. Kim, J. H. Choe, B. W. Lee, and K.-H. Chung, Bull. Korean Chem. Soc., 33, 2765 (2012).
K. J. Dignam and A. F. Hegarty, J. Chem. Soc., Chem. Commun., 862 (1976).
A. Gunasekaran, T. Jayachandran, J. H. Boyer, and M. L. Trudell, J. Heterocycl. Chem., 32, 1405 (1995).
Yu. A. Strelenko, O. A. Rakitin, and L. I. Khmel'nitskii, Izv. Akad. Nauk SSSR, Ser. Khim., 1023 (1990). [Bull. Acad. Sci. USSR, Div. Chem. Sci., 39, 912 (1990)].
M. Tanaka, T. Shono, and K. Shinra, Anal. Chim. Acta, 46, 125 (1969).
S. Kinoshita, H. Wakita, and I. Masuda, Bull. Chem. Soc. Jpn, 59, 653 (1986).
Y. Gök and H. Kantekin, Chem. Ber., 123, 1479 (1990).
E. Buehler and G. B. Brown, J. Org. Chem., 32, 265 (1967).
E. Abele, R. Abele, J. Popelis, and E. Lukevics, Org. Prep. Proc. Int., 32, 153 (2000).
S. Yavuz, H. Ozkan, N. Colak, and Y. Yildirir, Molecules, 16, 6677 (2011).
D. Kubmarawa, J. T. Barminas, and A. O. C. Aliyu, Arch. Appl. Sci. Res., 3, 126 (2011).
N. E. Alexandrou and A. G. Varvoglis, Org. Magn. Res., 3, 293 (1971).
M. Macit, H. Bati, and B. Bati, Synth. React. Inorg. Met.-Org. Chem., 28, 833 (1998).
P. Walstra, W. P. Trompen, and J. T. Hackman, Rec. Trav. Chim. Pays-Bas, 87, 452 (1968).
L. I. Khmel'nitskii, V. A. Shlyaposhnikov, N. O. Cherskaya, O. L. Lebedev, L. V. Epishina, and G. V. Korotkova, Izv. Akad. Nauk SSSR, Ser. Khim., 174 (1984). [Bull. Acad. Sci. USSR, Div. Chem. Sci., 33, 155 (1981)].
A. Corsaro, U. Chiacchio, and V. Pistarà, Synthesis, 1903 (2001).
F. P. Invidiata, S. Aiello, G. Furnò, E. Aiello, D. Simoni, and R. Rondanin, J. Heterocycl. Chem., 37, 355 (2000).
A. Szabó and A. Kovács, J. Mol. Struct., 651–653, 615 (2003).
V. I. Kovalenko, I. V. Vigalok, and G. G. Petrova, Zh. Strukt. Khim., 54 (1992). [J. Struct. Chem., 33, 202 (1992)].
H. Cerecetto, M. González, G. Seoane, C. Stanko, O. E. Piro, and E. Castellano, J. Braz. Chem. Soc., 15, 232 (2004).
J. Charalambous, G. Soobramanien, A. D. Stylianou, G. Manini, L. Operti, and G. A. Vaglio, Org. Mass Spectrom., 18, 406 (1983).
I. V. Tselinsky, S. F. Mel'nikova, T. V. Romanova, N. P. Spiridonova, and E. A. Dundukova, Zh. Org. Khim., 1419 (2001). [Russ. J. Org. Chem., 37, 1355 (2001)].
Y.-S. Zhou, B.-Z. Wang, J.-K. Li, C. Zhou, L. Hu, Z.-Q. Chen, and Z. Z. Zhang, Acta Chim. Sin., 69, 1673 (2011).
A. B. Sheremetev, V. G. Andrianov, E. V. Mantseva, E. V. Shatunova, N. S. Aleksandrova, I. L. Yudin, D. E. Dmitriev, B. B. Averkiev, and M. Yu. Antipin, Izv. Akad. Nauk, Ser. Khim., 569 (2004). [Russ. Chem. Bull., 53, 596 (2004)].
S. Nishimura, Handbook of Heterogeneous Catalytic Hydrogenation for Organic Synthesis, John Wiley & Sons, New York (2001), p. 37.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 830–845, May, 2013.
Rights and permissions
About this article
Cite this article
Stepanov, A.I., Dashko, D.V. & Astrat’ev, A.A. 1,2-Di(4-R-furazan-3-yl)glyoximes: Synthesis by the Reduction of 3,4-Bis(4-R-furazan-3-yl)furoxans and Study of the Reactivity of these Compounds. Chem Heterocycl Comp 49, 776–790 (2013). https://doi.org/10.1007/s10593-013-1309-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-013-1309-y