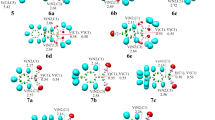
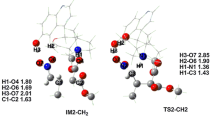
In this paper, we report theoretical studies of the addition–cyclization–isomerization reaction of propargyl cyanamides with thiol and methanol by density functional theory (DFT) calculation. The results reveal that this reaction takes place via five steps: 1) nucleophilic attack of S or O atom to C atom in the cyanogen group of propargyl cyanamide to form a cisoid-intermediate; 2) the conversion of the latter to its trans-conformer; 3) nucleophilic attack by N atom at the alkyne group to produce a five-membered thermodynamically unstable zwitterionic 4-ethylidene-4,5-dihydroimidazole intermediate; 4) proton transfer from N to C(4) atom to produce a more stable intermediate; 5) proton transfer from C(5) to ethylidene group to form the final 4-ethyl-1,5-dimethyl-2-methylsulfanyl- or 4-ethyl-2-methoxy-1,5-dimethylimidazole. We find that the autocatalysis by thiol or methanol is able to largely decrease the energy barrier of intramolecular proton transfer in the isomerization step and the proton transfer in the addition step.








Similar content being viewed by others
References
M. A. Jarosinski and W. K. Anderson, J. Org. Chem., 56, 4058 (1991).
M.-E. Theoclitou, N. G. J. Delaet, and L. A. Robinson, J. Comb. Chem., 4, 315 (2002).
R. L. Giles, R. A. Nkansah, and R. E. Looper, J. Org. Chem., 75, 261 (2010).
R. L. Giles, J. D. Sullivan, A. M. Steiner, and R. E. Looper, Angew. Chem., Int. Ed., 48, 3116 (2009).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, Jr., T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. J. Gonzalez, and A. Pople, Gaussian 03, revision C.02, Gaussian Inc., Wallingford CT (2004).
A. Bottoni, V. Frenna, C. Z. Lanza, G. Macaluso, and D. Spinelli, J. Phys. Chem. A, 108, 1731 (2004).
A. Salvà, J. Donoso, J. Frau, and F. Muñoz, J. Phys. Chem. A, 108, 11709 (2004).
A. Salvà, J. Donoso, J. Frau, and F. Muñoz, J. Phys. Chem. A, 107, 9409 (2003).
W. Yang and D. G. Drueckhammer, Org Lett., 2, 4133 (2000).
The support of the Natural Science Foundation of China (10947171) is gratefully acknowledged. H. Wang also acknowledges the finance support by Nanchang University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 284–296, February, 2013.
Rights and permissions
About this article
Cite this article
Ren, X., Yuan, Y., Ju, Y. et al. DFT Study of the Addition–Cyclization–Isomerization Reaction between Propargyl Cyanamides and Thiol or Alcohol: the Role of Catalyst. Chem Heterocycl Comp 49, 260–272 (2013). https://doi.org/10.1007/s10593-013-1243-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-013-1243-z