Abstract
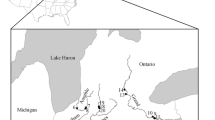
Understanding how populations are genetically and demographically connected is beneficial for species management, since gene flow and dispersal contribute to genetic diversity and population persistence. For hellbenders (Cryptobranchus alleganiensis), an aquatic salamander species experiencing dramatic declines in population size, fine-scale (i.e. within river) patterns of genetic diversity and gene flow are not well understood. Previous findings indicate that hellbenders are habitat specialists that exhibit extreme site fidelity and low vagility, suggesting that gene flow is restricted among the several, discrete habitat patches within a river. Using 15 polymorphic microsatellite loci and 497 hellbender samples from four Missouri rivers, we assessed fine-scale patterns of genetic diversity in order to infer population connectivity and aid in population management. Results indicate moderate levels of genetic variation (HO = 0.66–0.78) with little differentiation among habitat patches (avg. FST = 0.002) and no evidence of isolation by distance. Our data suggest that hellbender gene flow has been extensive even among habitat patches separated by distances greater than >100 km. These results are useful for hellbender management, especially in terms of making informed decisions regarding restorative releases of captively propagated individuals.


Similar content being viewed by others
References
Antao T, Lopes A, Lopes RJ, Beja-Pereira A, Luikart G (2008) LOSITAN: a workbench to detect molecular adaptation based on a Fst-outlier method. BMC Bioinform 9(1):323
Bodinof C (2010) Translocation and conservation of hellbenders (Cryptobranchus alleganiensis) in Missouri. Thesis, University of Missouri, Columbia
Bodinof CM, Briggler JT, Junge RE, Beringer J, Wanner MD, Schuette CD, Ettling J, Gitzen RA, Millspaugh JJ (2012a) Postrelease movements of captive-reared Ozark hellbenders (Cryptobranchus alleganiensis bishopi). Herpetologica 68(2):160–173. doi:10.1655/herpetologica-d-11-00033.1
Bodinof CM, Briggler JT, Junge RE, Beringer J, Wanner MD, Schuette CD, Ettling J, Millspaugh JJ (2012b) Habitat attributes associated with short-term settlement of Ozark hellbender (Cryptobranchus alleganiensis bishopi) salamanders following translocation to the wild. Freshw Biol 57(1):178–192. doi:10.1111/j.1365-2427.2011.02697.x
Briggler JT, Larson KA, Irwin KJ (2008) Presence of the amphibian chytrid fungus (Batrachochytrium dendrobatidis) on hellbenders (Cryptobranchus alleganiensis) in the Ozark Highlands. Herpetol Rev 39:443–444
Briggler JT, Crabill TL, Irwin KJ, Davidson C, Civiello JA, Wanner MD, Schuette CD, Armstrong SL, Grant V, Davidson T, Ettling JA (2012) Propagation, augmentation, and reintroduction plan for the Ozark Hellbender (Cryptobranchus alleganiensis bishopi). Ozark Hellbender Propagation Committee, Jefferson City
Bruce RC (1985) Larval periods, population structure and the effects of stream drift in larvae of the salamanders Desmognathus quadramaculatus and Leurognathus marmoratus in a Southern Appalachian stream. Copeia 1985(4):847–854. doi:10.2307/1445232
Bruce RC (1986) Upstream and downstream movements of Eurycea bislineata and other salamanders in a Southern Appalachian stream. Herpetologica 42(2):149–155. doi:10.2307/3892380
Burgmeier NG, Sutton TM, Williams RN (2011a) Spatial ecology of the Eastern hellbender (Cryptobranchus alleganiensis alleganiensis) in Indiana. Herpetologica 67(2):135–145. doi:10.1655/herpetologica-d-10-00027.1
Burgmeier NG, Unger SD, Sutton TM, Williams RN (2011b) Population status of the Eastern hellbender (Cryptobranchus alleganiensis alleganiensis) in Indiana. J Herpetol 45(2):195–201. doi:10.1670/10-094.1
Cano JM, Laurila A, Palo J, Merila J (2004) Population differentiation in G matrix structure due to natural selection in Rana Temporaria. Evolution 58(9):2013–2020
Coatney CE (1982) Home range and nocturnal activity of the Ozark hellbender. Thesis, Southwest Missouri State University, Springfield
Core Team R (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Cornuet JM, Luikart G (1996) Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144(4):2001–2014
Crowhurst R, Faries K, Collantes J, Briggler J, Koppelman J, Eggert L (2011) Genetic relationships of hellbenders in the Ozark highlands of Missouri and conservation implications for the Ozark subspecies (Cryptobranchus alleganiensis bishopi). Conserv Genet 12(3):637–646. doi:10.1007/s10592-010-0170-0
Di Rienzo A, Peterson AC, Garza JC, Valdes AM, Slatkin M, Freimer NB (1994) Mutational processes of simple-sequence repeat loci in human populations. Proc Natl Acad Sci USA 91(8):3166–3170
Earl DA, vonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Res 4(2):359–361
Estoup A, Rousset F, Michalakis Y, Cornuet JM, Adriamanga M, Guyomard R (1998) Comparative analysis of microsatellite and allozyme markers: a case study investigating micorgeographic differentiation in brown trout (Salmo trutta). Mol Ecol 7(3):339–353
Ettling JA, Wanner MD, Schuette CD, Armstrong SL, Pedigo AS, Briggler JT (2013) Captive reproduction and husbandry of adult Ozark hellbenders Cryptobranchus alleganiensis bishopi. Herpetol Rev 44(4):604–610
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Excoffier L, Lischer HEL (2010) Arlequin suite ver. 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol Res 10(3):564–567. doi:10.1111/j.1755-0998.2010.02847.x
Ferguson H (1998) Demography, dispersal, and colonisation of larvae of Pacific giant salamanders (Dicamptodon tenebrosus) at the northern extent of their range. Thesis, University of British Columbia, Vancouver
Fobes TM (1995) Habitat analysis of the Ozark hellbender, Cryptobranchus alleganiensis bishopi, in Missouri. Thesis, Southwest Missouri State University, Springfield
Foster RL, McMillan AM, Roblee KJ (2009) Population status of hellbender salamanders (Cryptobranchus alleganiensis) in the Allegheny River drainage of New York State. J Herpetol 43(4):579–588. doi:10.1670/08-156.1
Frankham R (1995) Conservation genetics. Annu Rev Genet 29:305–327
Fraser DJ, Bernatchez L (2001) Adaptive evolutionary conservation: towards a unified concept for defining conservation units. Mol Ecol 10(12):2741–2752. doi:10.1046/j.0962-1083.2001.01411.x
Gall BG, Mathis A (2010) Innate predator recognition and the problem of introduced trout. Ethology 116:47–58
Gates JE, Hocutt CH, Stauffer JR Jr, Taylor GJ (1985a) The distribution and status of Cryptobranchus alleganiensis in Maryland. Herpetol Rev 16:17–18
Gates JE, Stouffer RH Jr, Stauffer JR Jr, Hocutt CH (1985b) Dispersal patterns of translocated Cryptobranchus alleganiensis in a Maryland stream. J Herpetol 19(3):436–438. doi:10.2307/1564279
Gomez-Mestre I, Tejedo M (2004) Contrasting patterns of qualitative and neutral genetic variation in locally adapted populations of the natterjack toad Bufo calamita. Evolution 58(10):2343–2352
Goslee SC, Urban DL (2007) The ecodist package for dissimilarity-based analysis of ecological data. J Stat Softw 22(7):1–19
Hangartner S, Laurila A, Räsänen K (2011) Adaptive divergence in moor frog (Rana arvalis) populations along an acidification gradient: Influences from QST to FST correlations. Evolution 66:867–881
Humphries WJ (2005) Cryptobranchus alleganiensis displacement by a flood. Herpetol Rev 36(4):428
Johnson JE, Goldberg AS (1975) Movement of larval two lined salamanders (Eurycea bislineata) in the Mill River, Massachusetts. Copeia 3:588–589. doi:10.2307/1443669
Johnson J, Faries K, Rabenold J, Crowhurst R, Briggler J, Koppelman J, Eggert L (2009) Polymorphic microsatellite loci for studies of the Ozark hellbender (Cryptobranchus alleganiensis bishopi). Conserv Genet 10(6):1795–1797. doi:10.1007/s10592-009-9818-z
Jombart T (2000) Adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405
Kalinowski ST (2005) Hp-rare 1.0: a computer program for performing rarefaction on measures of allelic richness. Mol Ecol Notes 5(1):187–189. doi:10.1111/j.1471-8286.2004.00845.x
Keitzer SC (2007) Habitat preferences of the eastern hellbender in West Virginia. Thesis, Marshall University, Huntington
Kimura M, Crow JF (1964) The number of alleles that can be maintained in a finite population. Genetics 49(4):725–738
Kimura M, Ohta T (1978) Stepwise mutation model and distribution of allelic frequencies in a finite population. Proc Natl Acad Sci USA 75(6):2868–2872
Kuo C-H, Janzen FJ (2004) Genetic effects of a persistent bottleneck on a natural population of ornate box turtles (Terrapene ornata). Conserv Genet 5(4):425–437. doi:10.1023/b:coge.0000041020.54140.45
Landguth EL, Cushman SA, Schwartz MK, McKelvey KS, Murphy M, Luikart G (2010) Quantifying the lag time to detect barriers in landscape genetics. Mol Ecol 19(19):4179–4191. doi:10.1111/j.1365-294X.2010.04808.x
Latch E, Dharmarajan G, Glaubitz J, Rhodes O (2006) Relative performance of Bayesian clustering software for inferring population substructure and individual assignment at low levels of population differentiation. Conserv Genet 7(2):295–302
Leinonen T, O’Hara RB, Cano JM, Merila J (2008) Comparative studies of quantitative trait and neutral marker divergence: a meta-analysis. J Evol Biol 21(1):1–17
Lenormand T (2002) Gene flow and the limits to natural selection. Trends Ecol Evol 17(4):183–189
Lippe C, Dumont P, Bernatchez L (2006) High genetic diversity and no inbreeding in the endangered copper redhorse, Moxostoma hubbsi (Catostomidae, Pisces): the positive sides of a long generation time. Mol Ecol 15(7):1769–1780. doi:10.1111/j.1365-294X.2006.02902.x
Lowe WH, Allendorf FW (2010) What can genetics tell us about population connectivity? Mol Ecol 19(15):3038–3051. doi:10.1111/j.1365-294X.2010.04688.x
Luikart G, Cornuet JM (1998) Empirical evaluation of a test for identifying recently bottlenecked populations from allele frequency data. Conserv Biol 12(1):228–237
Lynch M (1996) A quantitative genetic perspective on conservation issues. In: Avise J, Hamrick J (eds) Conservation genetics: case histories from nature. Chapman & Hall, New York, pp 471–501
Merilä J, Crnokrak P (2001) Comparison of genetic differentiation at marker loci and quantitative traits. J Evol Biol 14(6):892–903
Nei M, Chakravarti A (1977) Drift variances of FST and GST statistics obtained from a finite number of isolated populations. Theor Popul Biol 11:307–325
Nei M, Maruyama T, Chakraborty R (1975) The bottleneck effect and genetic variability in populations. Evolution 29(1):1–10. doi:10.2307/2407137
Nickerson MA, Mays CE (1973) A study of the Ozark hellbender Cryptobranchus alleganiensis bishopi. Ecology 54(5):1164–1165. doi:10.2307/1935586
Paetkau D, Calvert W, Stirling I, Strobeck C (1995) Microsatellite analysis of population structure in Canadian polar bears. Mol Ecol 4(3):347–354. doi:10.1111/j.1365-294X.1995.tb00227.x
Palo JU, O’Hara RB, Laugen T, Laurila A, Primmers CR, Merila J (2003) Latitudinal divergence of common frog (Rana temporaria) life history traits by natural selection: evidence from a comparison of molecular and quantitative genetic data. Mol Ecol 12(7):1963–1978
Peakall ROD, Smouse PE (2006) Genalex 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6(1):288–295. doi:10.1111/j.1471-8286.2005.01155.x
Peterson CL (1987) Movement and catchability of the hellbender, Cryptobranchus alleganiensis. J Herpetol 21(3):197–204
Peterson CL, Wilkinson RF (1996) Home range size of the hellbender (Cryptobranchus alleganiensis) in Missouri. Herpetol Rev 27(3):126–127
Peterson CL, Wilkinson RF Jr, Milton ST, Metter DE (1983) Age and growth of the Ozark hellbender (Cryptobranchus alleganiensis bishopi). Copeia 1:225–231
Petranka JW (1998) Salamanders of the United States and Canada. Smithsonian Institution Press, Washington, D.C.
Petranka JW, Sih A (1986) Environmental instability, competition, and density-dependent growth and survivorship of a stream-dwelling salamander. Ecology 67(3):729–736. doi:10.2307/1937696
Petranka JW, Sih A, Kats LB, Holomuzki JR (1987) Stream drift, size-specific predation, and the evolution of ovum size in an amphibian. Oecologia 71(4):624–630. doi:10.2307/4218213
Pfingsten RA (1989) The status and distribution of the hellbender, Cryptobranchus alleganiensis, Ohio. Ohio J Sci 89(2):3
Piry S, Luikart G, Cornuet J-M (1999) BOTTLENECK: a computer program for detecting recent reduction in the effective population size using allele frequency data. J Hered 90:502–503
Pittman S, King T, Faurby S, Dorcas M (2011) Demographic and genetic status of an isolated population of bog turtles (Glyptemys muhlenbergii): implications for managing small populations of long-lived animals. Conserv Genet 12(6):1589–1601. doi:10.1007/s10592-011-0257-2
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Quinn SA, Gibbs JP, Hall MH, Petokas PJ (2013) Multiscale factors influencing distribution of the eastern hellbender salamander (Cryptobranchus alleganiensis alleganiensis) in the northern segment of its range. J Herpetol 47(1):78–84. doi:10.1670/11-127
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17(1):230–237. doi:10.1046/j.1523-1739.2003.01236.x
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43(1):223–225
Richter-Boix A, Quintela M, Kierczak M, Franch M, Laurila A (2013) Fine-grained adaptive divergence in an amphiban: genetic basis of phenotypic divergence and the role of nonrandom gene flow in restricting effective migration among wetlands. Mol Ecol 22:1322–1340
Ross KG, Shoemaker DD, Krieger MJB, DeHeer CJ, Keller L (1999) Assessing genetic structure with multiple classes of molecular markers: a case study involving the introduced fire ant Solenopsis invicta. Mol Biol Evol 16(4):525–543
Rousset F (2008) Genepop’007: a complete re-implementation of the genepop software for Windows and Linux. Mol Ecol Res 8(1):103–106. doi:10.1111/j.1471-8286.2007.01931.x
Routman E (1993) Mitochondrial DNA variation in Cryptobranchus alleganiensis, a salamander with extremely low allozyme diversity. Copeia 2:407–416
Sabatino S, Routman E (2009) Phylogeography and conservation genetics of the hellbender salamander (Cryptobranchus alleganiensis). Conserv Genet 10(5):1235–1246. doi:10.1007/s10592-008-9655-5
Semlitsch RD (2008) Differentiating migration and dispersal processes for pond-breeding amphibians. J Wildl Manage 72(1):260–267. doi:10.2193/2007-082
Slatkin M (1985) Gene flow in natural populations. Annu Rev Ecol Syst 16:393–430
Slatkin M (1987) Gene flow and the geographic structure of natural populations. Science 236(4803):787–792. doi:10.1126/science.3576198
Smith BG (1907) The life history and habits of Cryptobranchus alleganiensis. Biol Bull 13:5–39
Stoneburner DL (1978) Salamander drift. Freshw Biol 8(3):291–293. doi:10.1111/j.1365-2427.1978.tb01450.x
Storfer A (1999) Gene flow and endangered species translocations: a topic revisited. Biol Conserv 87:173–180
Taber CA, Wilkinson RF Jr, Milton ST (1975) Age and growth of hellbenders in the Niangua River, Missouri. Copeia 4:633–639
Templeton AR, Shaw K, Routman E, Davis SK (1990) The genetic consequences of habitat fragmentation. Ann Missouri Bot Gard 77(1):13–27
Thiesmeier B, Schuhmacher H (1990) Causes of larval drift of the fire salamander, Salamandra salamandra terrestris, and its effects on population dynamics. Oecologia 82(2):259–263. doi:10.2307/4219231
Topping MS, Peterson CL (1985) Movement in the hellbender, Cryptobranchus alleganiensis. Trans Missouri Acad Sci 19:121
Trauth SE, Wilhide JD, Daniel P (1992) Status of the Ozark hellbender, Cryptobranchus bishopi (Urodela: Cryptobranchidae), in the Spring River, Fulton County, Arkansas. Arkansas Acad Sci 46:83–85
Unger S, Fike J, Sutton T, Rhodes O, Williams R (2010) Isolation and development of 12 polymorphic tetranucleotide microsatellite markers for the eastern hellbender (Cryptobranchus alleganiensis alleganiensis). Conserv Genet Res 2(1):89–91. doi:10.1007/s12686-009-9170-0
Unger SD, Rhodes OE, Sutton TM, Williams RN (2013) Population genetics of the eastern hellbender (Cryptobranchus alleganiensis alleganiensis) across multiple spatial scales. PLoSONE 8(10):e74180. doi:10.1371/journal.pone.0074180
USFWS (U.S. Fish and Wildlife Service) (2011) Endangered and threatened wildlife and plants; endangered status for the Ozark hellbender salamander. Fed Reg 76(194):61956–61978
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) Micro-checker: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4(3):535–538. doi:10.1111/j.1471-8286.2004.00684.x
Vineyard JD, Feder GL (1974) Springs of Missouri. Missouri Geological Survey and Water Resources, Rolla
Waits LP, Luikart G, Taberlet P (2001) Estimating the probability of identity among genotypes in natural populations: Cautions and guidelines. Mol Ecol 10(1):249–256. doi:10.1046/j.1365-294X.2001.01185.x
Wang J (2007) Triadic IBD coefficients and applications to estimating pairwise relatedness. Genet Res 89(03):135–153. doi:10.1017/S0016672307008798
Wang J (2011) Coancestry: a program for simulating, estimating and analysing relatedness and inbreeding coefficients. Mol Ecol Res 11(1):141–145. doi:10.1111/j.1755-0998.2010.02885.x
Wheeler BA, Prosen E, Mathis A, Wilkinson RF (2003) Population declines of a long-lived salamander: A 20+-year study of hellbenders, Cryptobranchus alleganiensis. Biol Conserv 109(1):151–156. doi:10.1016/s0006-3207(02)00136-2
Wright S (1943) Isolation by distance. Genetics 28(2):114–138
Acknowledgments
Funding for this study was provided by the Missouri Department of Conservation. Samples were collected by Jeffery T. Briggler of the Missouri Department of Conservation under the authority of the Wildlife Code of Missouri. Aaron Schuh provided lab assistance for which we are grateful. Many thanks to William E. Peterman, Mari Jose Ruiz-Lopez, and Raymond D. Semlitsch for their valuable advice and suggestions for revision.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Feist, S.M., Briggler, J.T., Koppelman, J.B. et al. Within-river gene flow in the hellbender (Cryptobranchus alleganiensis) and implications for restorative release. Conserv Genet 15, 953–966 (2014). https://doi.org/10.1007/s10592-014-0591-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-014-0591-2