Abstract
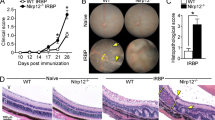
Previously we demonstrated that intraperitoneal (IP) inoculation of Rev-2-T-6 mouse lymphoma into syngeneic Balb/c hosts resulted in brain metastasis, migration along the optic nerve sheath, and ocular infiltration. In a second model: intravitreal inoculation of Rev-2-T-6 cells, the developing lymphoma was largely confined within the eye, seldom breaching the retinal pigment epithelium to reside in the choroid and sclera. There was no retrograde infiltration into the brain. Here, we describe a third, complementary model, whereby intravitreal inoculation of Rev-2-T-6 cells into Balb/c mice, followed by repeated IP inoculations of anti-LFA-1/CD11a monoclonal antibodies, results in extensive infiltration of the choroid, sclera, conjunctiva, eyelids and orbit. Furthermore, the lymphoma cells metastasize along the optic nerve sheath into the brain, and through the contralateral optic nerve tract into the contralateral eye. There is no systemic involvement of the lymphoma. Furthermore, anti-LFA-1 treatment results in elevated levels of serum anti-Rev-2-T-6 antibodies. Inoculation of Rev-2-T-6 cells into the vitreous of severe combined immune deficient mice demonstrates a course of clinical signs and histopathological findings similar to those in immune-competent mice treated with anti-LFA-1 antibodies, including invasion of the contralateral eye. Taken together, these findings suggest that confinement of Rev-2-T-6 lymphoma cells to the eye depends on active immune surveillance using a population of effector cells expressing the cell surface integrin LFA-1. Impairing this protection enhances tumor aggressiveness within the eye, and the likelihood of early retrograde lymphoma metastasis into the brain and the contralateral eye.





Similar content being viewed by others
Abbreviations
- ICAM-1:
-
Intracellular adhesion molecule-1
- LFA-1:
-
Lymphocyte function-associated antigen-1
- SCID:
-
Severe combined immune deficient
- IP:
-
Intraperitoneal
- APCs:
-
Antigen presenting cells
References
Gonzalez-Amaro R, Sanchez-Madrid F (1999) Cell adhesion molecules: selectins and integrins. Crit Rev Immunol 19:389–429
Hogg N, Laschinger M, Giles K, McDowall A (2003) T-cell integrins: more than just sticking points. J Cell Sci 116:4695–4705
Hynes RO (2002) Integrins: bidirectional, allosteric signaling machines. Cell 110:673–687
Ostermann G, Weber KS, Zernecke A, Schroder A, Weber C (2002) JAM-1 is a ligand of the beta(2) integrin LFA-1 involved in transendothelial migration of leukocytes. Nat Immunol 3:151–158
Shulman Z, Shinder V, Klein E et al (2009) Lymphocyte crawling and transendothelial migration require chemokine triggering of high-affinity LFA-1 integrin. Immunity 30:384–396
Kavanaugh AF, Lightfoot E, Lipsky PE, Oppenheimer-Marks N (1991) Role of CD11/CD18 in adhesion and transendothelial migration of T cells. Analysis utilizing CD18-deficient T cell clones. J Immunol 146:4149–4156
Warnock RA, Askari S, Butcher EC, von Andrian UH (1998) Molecular mechanisms of lymphocyte homing to peripheral lymph nodes. J Exp Med 187:205–216
Evans R, Patzak I, Svensson L et al (2009) Integrins in immunity. J Cell Sci 122:215–225
Reichardt P, Dornbach B, Gunzer M (2007) The molecular makeup and function of regulatory and effector synapses. Immunol Rev 218:165–177
O’Rourke AM, Ybarrondo B, Mescher MF (1993) CD8 and antigen-specific T cell adhesion cascades. Semin Immunol 5:263–270
Nicolls MR, Gill RG (2006) LFA-1 (CD11a) as a therapeutic target. Am J Transplant 6:27–36
Suzuki J, Isobe M, Izawa A et al (1999) Differential Th1 and Th2 cell regulation of murine cardiac allograft acceptance by blocking cell adhesion of ICAM-1/LFA-1 and VCAM-1/VLA-4. Transpl Immunol 7:65–72
Smits HH, de Jong EC, Schuitemaker JH et al (2002) Intercellular adhesion molecule-1/LFA-1 ligation favors human Th1 development. J Immunol 168:1710–1716
Koopman G, Keehnen RM, Lindhout E et al (1994) Adhesion through the LFA-1 (CD11a/CD18)-ICAM-1 (CD54) and the VLA-4 (CD49d)-VCAM-1 (CD106) pathways prevents apoptosis of germinal center B cells. J Immunol 152:3760–3767
Smith A, Stanley P, Jones K, Svensson L, McDowall A, Hogg N (2007) The role of the integrin LFA-1 in T-lymphocyte migration. Immunol Rev 218:135–146
Poston RS, Robbins RC, Chan B et al (2000) Effects of humanized monoclonal antibody to rhesus CD11a in rhesus monkey cardiac allograft recipients. Transplantation 69:2005–2013
Harrison PC, Madwed JB (1999) Anti-LFA-1 alpha reduces the dose of cyclosporin A needed to produce immunosuppression in heterotopic cardiac transplanted rats. J Heart Lung Transplant 18:279–284
Hasegawa Y, Yokono K, Taki T et al (1994) Prevention of autoimmune insulin-dependent diabetes in non-obese diabetic mice by anti-LFA-1 and anti-ICAM-1 mAb. Int Immunol 6:831–838
Fischer A, Friedrich W, Fasth A et al (1991) Reduction of graft failure by a monoclonal antibody (anti-LFA-1 CD11a) after HLA nonidentical bone marrow transplantation in children with immunodeficiencies, osteopetrosis, and Fanconi’s anemia: a European Group for Immunodeficiency/European Group for Bone Marrow Transplantation report. Blood 77:249–256
Whitcup SM, Hikita N, Shirao M et al (1995) Monoclonal antibodies against CD54 (ICAM-1) and CD11a (LFA-1) prevent and inhibit endotoxin-induced uveitis. Exp Eye Res 60:597–601
Whitcup SM, DeBarge LR, Caspi RR, Harning R, Nussenblatt RB, Chan CC (1993) Monoclonal antibodies against ICAM-1 (CD54) and LFA-1 (CD11a/CD18) inhibit experimental autoimmune uveitis. Clin Immunol Immunopathol 67:143–150
Simmons DL (2005) Anti-adhesion therapies. Curr Opin Pharmacol 5:398–404
Gonzalez-Amaro R, Mittelbrunn M, Sanchez-Madrid F (2005) Therapeutic anti-integrin (alpha4 and alphaL) monoclonal antibodies: two-edged swords? Immunology 116:289–296
Giblin PA, Lemieux RM (2006) LFA-1 as a key regulator of immune function: approaches toward the development of LFA-1-based therapeutics. Curr Pharm Des 12:2771–2795
Marecki S, Kirkpatrick P (2004) Efalizumab. Nat Rev Drug Discov 3:473–474
Vincenti F, Mendez R, Pescovitz M et al (2007) A phase I/II randomized open-label multicenter trial of efalizumab, a humanized anti-CD11a, anti-LFA-1 in renal transplantation. Am J Transplant 7:1770–1777
Cavazzana-Calvo M, Bordigoni P, Michel G et al (1996) A phase II trial of partially incompatible bone marrow transplantation for high-risk acute lymphoblastic leukaemia in children: prevention of graft rejection with anti-LFA-1 and anti-CD2 antibodies. Societe Francaise de Greffe de Moelle Osseuse. Br J Haematol 93:131–138
Gerstner E, Batchelor T (2007) Primary CNS lymphoma. Expert Rev Anticancer Ther 7:689–700
Bayraktar S, Bayraktar UD, Ramos JC, Stefanovic A, Lossos IS (2011) Primary CNS lymphoma in HIV positive and negative patients: comparison of clinical characteristics, outcome and prognostic factors. J Neurooncol 101:257–265
Sacktor N (2002) The epidemiology of human immunodeficiency virus-associated neurological disease in the era of highly active antiretroviral therapy. J Neurovirol 8(Suppl 2):115–121
Assaf N, Hasson T, Hoch-Marchaim H et al (1997) An experimental model for infiltration of malignant lymphoma to the eye and brain. Virchows Arch 431:459–467
Hochman J, Assaf N, Deckert-Schluter M, Wiestler OD, Pe’er J (2001) Entry routes of malignant lymphoma into the brain and eyes in a mouse model. Cancer Res 61:5242–5247
Chan CC, Fischette M, Shen D, Mahesh SP, Nussenblatt RB, Hochman J (2005) Murine model of primary intraocular lymphoma. Invest Ophthalmol Vis Sci 46:415–419
Hochman J, Bar-Sinai A, Engelhardt B (2001) Involvement of LFA-1/ICAM-1 in metastasis of lymphoma to the Brain in a mouse model. Proc Am Assoc Cancer Res 42:914
Wang S, Coleman EJ, Pop LM, Brooks KJ, Vitetta ES, Niederkorn JY (2006) Effect of an anti-CD54 (ICAM-1) monoclonal antibody (UV3) on the growth of human uveal melanoma cells transplanted heterotopically and orthotopically in SCID mice. Int J Cancer 118:932–941
Tamatani T, Kotani M, Miyasaka M (1991) Characterization of the rat leukocyte integrin, CD11/CD18, by the use of LFA-1 subunit-specific monoclonal antibodies. Eur J Immunol 21:627–633
Whitcup SM, DeBarge LR, Rosen H, Nussenblatt RB, Chan CC (1993) Monoclonal antibody against CD11b/CD18 inhibits endotoxin-induced uveitis. Invest Ophthalmol Vis Sci 34:673–681
Hochman N, Houri-Haddad Y, Koblinski J et al (2008) Cranberry juice constituents impair lymphoma growth and augment the generation of antilymphoma antibodies in syngeneic mice. Nutr Cancer 60:511–517
Zahalka MA, Okon E, Gosslar U, Holzmann B, Naor D (1995) Lymph node (but not spleen) invasion by murine lymphoma is both CD44- and hyaluronate-dependent. J Immunol 154:5345–5355
Yun Z, Menter DG, Nicolson GL (1996) Involvement of integrin alphavbeta3 in cell adhesion, motility, and liver metastasis of murine RAW117 large cell lymphoma. Cancer Res 56:3103–3111
Zahalka MA, Okon E, Naor D (1993) Blocking lymphoma invasiveness with a monoclonal antibody directed against the beta-chain of the leukocyte adhesion molecule (CD18). J Immunol 150:4466–4477
Camp RL, Scheynius A, Johansson C, Pure E (1993) CD44 is necessary for optimal contact allergic responses but is not required for normal leukocyte extravasation. J Exp Med 178:497–507
Scheynius A, Camp RL, Pure E (1993) Reduced contact sensitivity reactions in mice treated with monoclonal antibodies to leukocyte function-associated molecule-1 and intercellular adhesion molecule-1. J Immunol 150:655–663
Cohen S, Haimovich J, Hollander N (2003) Anti-idiotype × anti-LFA-1 bispecific antibodies inhibit metastasis of B cell lymphoma. J Immunol 170:2695–2701
Heusinkveld M, de Vos van Steenwijk PJ, Goedemans R et al (2011) M2 macrophages induced by prostaglandin E2 and IL-6 from cervical carcinoma are switched to activated M1 macrophages by CD4+ Th1 cells. J Immunol 187:1157–1165
Bronkhorst IH, Jager MJ (2012) Uveal melanoma: the inflammatory microenvironment. J Innate Immun (in press)
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23:549–555
Pollard JW (2004) Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer 4:71–78
Pessoa VF, Ikeda H (1984) Increase in axonal transport in demyelinating optic nerve fibres in the mouse infected with Semliki Forest virus. Brain 107(Pt 2):433–446
Friedl P, Weigelin B (2008) Interstitial leukocyte migration and immune function. Nat Immunol 9:960–969
Mempel TR, Henrickson SE, Von Andrian UH (2004) T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427:154–159
Shimonaka M, Katagiri K, Nakayama T et al (2003) Rap1 translates chemokine signals to integrin activation, cell polarization, and motility across vascular endothelium under flow. J Cell Biol 161:417–427
Smith A, Bracke M, Leitinger B, Porter JC, Hogg N (2003) LFA-1-induced T cell migration on ICAM-1 involves regulation of MLCK-mediated attachment and ROCK-dependent detachment. J Cell Sci 116:3123–3133
Barenfanger J, MacDonald AB (1974) The role of immunoglobulin in the neutralization of trachoma infectivity. J Immunol 113:1607–1617
Nishibori M, Takahashi HK, Mori S (2003) The regulation of ICAM-1 and LFA-1 interaction by autacoids and statins: a novel strategy for controlling inflammation and immune responses. J Pharmacol Sci 92:7–12
Rutishauser J (2006) The role of statins in clinical medicine–LDL–cholesterol lowering and beyond. Swiss Med Wkly 136:41–49
Demierre MF, Higgins PD, Gruber SB, Hawk E, Lippman SM (2005) Statins and cancer prevention. Nat Rev Cancer 5:930–942
Acknowledgments
This work was supported by the A.M.N foundation, Jerusalem (JH) and by the Intramural Research Programs of the National Institutes of Health, National Eye Institute (DS, CCC) and National Cancer Institute (JH, MMG).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hochman, J., Shen, D., Gottesman, M.M. et al. Anti-LFA-1 antibodies enhance metastasis of ocular lymphoma to the brain and contralateral eye. Clin Exp Metastasis 30, 91–102 (2013). https://doi.org/10.1007/s10585-012-9512-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-012-9512-2