Abstract
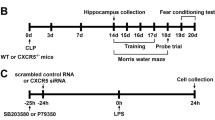
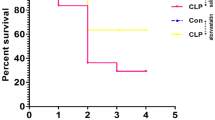
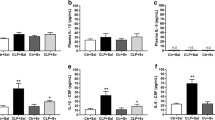
The purpose of this study is to test the hypothesis that the mammalian target of rapamycin (mTOR) signaling pathway might mediate neuroprotection in a mouse model of septic encephalopathy and also to identify the role of autophagy. Mice were subjected to cecal ligation and puncture (CLP) or a sham operation, and all 50 mice were randomly assigned to five groups: sham, CLP+ saline, CLP+ rapamycin (1, 5, 10 mg/kg) groups. Two weeks after the operation, Morris water maze was conducted for behavioral test; Nissl staining was used for observing glia infiltration; immunohistochemical staining and biochemical measures in hippocampi were performed to detect mTOR targets and autophagy indicators. Immunochemistry revealed significant loss of neurons and increased glia infiltration in hippocampus after CLP operation. Inhibition of mTOR by rapamycin rescued cognitive deficits caused by sepsis (p < 0.05). Rapamycin did not affect total mTOR targets, while phosphorylated mTOR targets (p-mTOR-Ser2448, p-p70S6k-Thr389, p-AKT-S473) decreased (p < 0.05) and autophagy indicators (LC3-II, Atg5, Atg7) were increased, and P62 was decreased in rapamycin-treated CLP mice compared with the untreated (p < 0.05) in hippocampus. Rapamycin improves learning after sepsis through enhancing autophagy and may be a potentially effective therapeutic agent for the treatment of sepsis-induced cognitive impairment.







Similar content being viewed by others
Abbreviations
- mTOR:
-
Mammalian target of rapamycin
- CLP:
-
Cecal ligation and puncture
- KM mice:
-
Kunming mice
References
Assaraf MI et al (2007) Brain erythropoietin receptor expression in Alzheimer disease and mild cognitive impairment. J Neuropathol Exp Neurol 66:389–398. doi:10.1097/nen.0b013e3180517b28
Caccamo A, Majumder S, Richardson A, Strong R, Oddo S (2010) Molecular interplay between mammalian target of rapamycin (mTOR), amyloid-β, and tau: effects on cognitive impairments. J Biol Chem 285:13107–13120. doi:10.1074/jbc.M110.100420
Casadio A et al (1999) A transient, neuron-wide form of CREB-mediated long-term facilitation can be stabilized at specific synapses by local protein synthesis. Cell 99:221–237
Cuschieri J, Bulmus V, Gourlay D, Garcia I, Hoffman A, Stayton P, Maier RV (2004) Modulation of macrophage responsiveness to lipopolysaccharide by IRAK-1 manipulation. Shock 21:182–188. doi:10.1097/01.shk.0000111828.07309.26
Diaz-Troya S, Perez-Perez ME, Florencio FJ, Crespo JL (2008) The role of TOR in autophagy regulation from yeast to plants and mammals. Autophagy 4:851–865
Ehninger D et al (2008) Reversal of learning deficits in a Tsc2± mouse model of tuberous sclerosis. Nat Med 14:843–848. doi:10.1038/nm1788
Gofton TE, Young GB (2012) Sepsis-associated encephalopathy. Nat Rev Neurol 8:557–566. doi:10.1038/nrneurol.2012.183
Granja C, Lopes A, Moreira S, Dias C, Costa-Pereira A, Carneiro A (2005) Patients’ recollections of experiences in the intensive care unit may affect their quality of life. Crit Care 9:R96–R109. doi:10.1186/cc3026
Halloran J et al (2012) Chronic inhibition of mammalian target of rapamycin by rapamycin modulates cognitive and non-cognitive components of behavior throughout lifespan in mice. Neuroscience 223:102–113. doi:10.1016/j.neuroscience.2012.06.054
Hsieh CH, Pai PY, Hsueh HW, Yuan SS, Hsieh YC (2011) Complete induction of autophagy is essential for cardioprotection in sepsis. Ann Surg 253:1190–1200. doi:10.1097/SLA.0b013e318214b67e
Inoki K, Corradetti MN, Guan KL (2005) Dysregulation of the TSC-mTOR pathway in human disease. Nat Genet 37:19–24. doi:10.1038/ng1494
Iwashyna TJ, Ely EW, Smith DM, Langa KM (2010) Long-term cognitive impairment and functional disability among survivors of severe sepsis. JAMA 304:1787–1794. doi:10.1001/jama.2010.1553
Iwashyna TJ, Cooke CR, Wunsch H, Kahn JM (2012) The population burden of long-term survivorship after severe sepsis among older Americans. J Am Geriatr Soc 60:1070–1077. doi:10.1111/j.1532-5415.2012.03989.x
Kanaan NM, Collier TJ, Marchionini DM, McGuire SO, Fleming MF, Sortwell CE (2006) Exogenous erythropoietin provides neuroprotection of grafted dopamine neurons in a rodent model of Parkinson’s disease. Brain Res 1068:221–229. doi:10.1016/j.brainres.2005.10.078
Klionsky DJ et al. (2016) Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition) Autophagy 12:1–222 doi:10.1080/15548627.2015.1100356
Lamming DW et al (2012) Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity. Science (N Y) 335:1638–1643. doi:10.1126/science.1215135
Marik PE (2011) Surviving sepsis: going beyond the guidelines. Ann Intensiv Care 1:17. doi:10.1186/2110-5820-1-17
Puighermanal E, Marsicano G, Busquets-Garcia A, Lutz B, Maldonado R, Ozaita A (2009) Cannabinoid modulation of hippocampal long-term memory is mediated by mTOR signaling. Nat Neurosci 12:1152–1158. doi:10.1038/nn.2369
Saitoh T et al (2008) Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1beta production. Nature 456:264–268. doi:10.1038/nature07383
Spilman P et al (2010) Inhibition of mTOR by rapamycin abolishes cognitive deficits and reduces amyloid-β levels in a mouse model of Alzheimer’s disease. PLoS ONE 5:e9979. doi:10.1371/journal.pone.0009979
Tischmeyer W, Schicknick H, Kraus M, Seidenbecher CI, Staak S, Scheich H, Gundelfinger ED (2003) Rapamycin-sensitive signalling in long-term consolidation of auditory cortex-dependent memory. Eur J Neurosci 18:942–950
Tsang CK, Qi H, Liu LF, Zheng XF (2007) Targeting mammalian target of rapamycin (mTOR) for health and diseases. Drug Discov Today 12:112–124. doi:10.1016/j.drudis.2006.12.008
Wang L et al (2011) Inactivation of mammalian target of rapamycin (mTOR) by rapamycin in a murine model of lipopolysaccharide-induced acute lung injury. Chin Med J 124:3112–3117
Wang GB, Ni YL, Zhou XP, Zhang WF (2014) The AKT/mTOR pathway mediates neuronal protective effects of erythropoietin in sepsis. Mol Cell Biochem 385:125–132. doi:10.1007/s11010-013-1821-5
Wullschleger S, Loewith R, Hall MN (2006) TOR signaling in growth and metabolism. Cell 124:471–484. doi:10.1016/j.cell.2006.01.016
Yen YT, Yang HR, Lo HC, Hsieh YC, Tsai SC, Hong CW, Hsieh CH (2013) Enhancing autophagy with activated protein C and rapamycin protects against sepsis-induced acute lung injury. Surgery 153:689–698. doi:10.1016/j.surg.2012.11.021
Acknowledgements
The authors thank Ms. Lihong Wan for administrative assistance. We are also grateful to Mr. Shikun Miao for their help with excellent animal care. This work was supported by National Natural Science Foundation of China (Nos. 81301659 and 81401077).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest.
Rights and permissions
About this article
Cite this article
Liu, W., Guo, J., Mu, J. et al. Rapamycin Protects Sepsis-Induced Cognitive Impairment in Mouse Hippocampus by Enhancing Autophagy. Cell Mol Neurobiol 37, 1195–1205 (2017). https://doi.org/10.1007/s10571-016-0449-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-016-0449-x