Abstract
Background
Recent data have demonstrated that long-lived memory T cells are present in the human lung and can play significant roles in the pathogenesis of specific allergic and autoimmune diseases. However, most evidence has been obtained from mouse studies, and the potential roles of memory T cells in human allergic diseases, such as asthma, remain largely unknown.
Methods
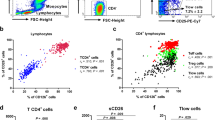
Thirty-three asthmatics, 26 chronic obstructive pulmonary disease (COPD) patients, and 22 healthy volunteers were enrolled in this study. Peripheral blood mononuclear cells (PBMCs) were isolated from the peripheral blood, and cell surface staining (CD4, CD45RO, CRTH2, CD62L, and CCR7) was performed for the detection of memory CD4+ T cells in blood. After stimulation with interleukin-27 (IL-27) or IL-4 for 15 min, the STAT1/STAT6 phosphorylation of memory CD4+ T cells was measured separately by flow cytometric techniques. The cytokine-releasing profiles after 6 days of culture under neutralization, TH2, TH2 + lipopolysaccharide (LPS), and TH2 + house dust mite (HDM) conditions were detected by intracellular protein (IL-5, IL-17, and interferon (IFN)-γ) staining. Correlation analyses between the profile of memory CD4+ T cells and clinical characteristics of asthma were performed.
Results
The number of circulating memory CD4+ T (CD4+ Tm) cells in asthmatics was increased compared with that in the healthy subjects (48 ± 5.7 % vs. 32 ± 4.1 %, p < 0.05). Compared with COPD and healthy subjects, the phosphorylation of signal transducer and activator of transcription 1 (STAT1-py) was impaired in asthmatics, whereas the phosphorylation of signal transducer and activator of transcription 6 (STAT6-py) was slightly enhanced. This imbalance of STAT1-py/STAT6-py was attributed to TH2 memory cells but not non-TH2 memory cells in blood. The cytokine-releasing profiles of asthmatics was unique, specifically IL-5high, IL-17high, and IFN-rlow, compared with those of COPD patients and healthy subjects. The IL-17 production levels in CD4+ Tm cells are associated with disease severity and positively correlated with medication consumption in asthma.
Conclusions
The long-lived, antigen-specific memory CD4+ T cells, rather than PBMCs or peripheral lymphocytes, might be the ideal T cell subset candidates for analyzing the endotype of asthma. Memory CD4+ T cells exhibiting a shift in STAT phosphorylation and specific cytokine-releasing profiles have the potential to facilitate the understanding of disease heterogeneity and severity, allowing the more personalized treatment of patients.






Similar content being viewed by others
Abbreviations
- STATs:
-
Signal transducer and activator of transcription
- CD4+Tm:
-
Memory CD4+ T Cells
- COPD:
-
Chronic obstructive pulmonary disease
- PBMCs:
-
Peripheral blood mononuclear cells
- STAT1-py:
-
STAT1 phosphorylation
- STAT6-py:
-
STAT6 phosphorylation
- LPS:
-
Lipopolysaccharide
- HDM:
-
house dust mite
- ILs:
-
interleukins
- Tem:
-
effector memory T cells
- Tcm:
-
central memory T cells
- Trm:
-
resident memory T cells
- Treg:
-
T regulatory cell
- MFIs:
-
Mean fluorescence intensities
- GINA:
-
Global Initiative for Asthma
References
Abdulamir AS, Hafidh RR, Abubakar F, Abbas KA. Changing survival, memory cell compartment, and T-helper balance of lymphocytes between severe and mild asthma. BMC Immunol. 2008;9:–73.
Chapoval SP, Dasgupta P, Smith EP, DeTolla LJ, Lipsky MM, Kelly-Welch AE, et al. STAT6 expression in multiple cell types mediates the cooperative development of allergic airway disease. J Immunol. 2011;186:2571–83.
Chen Z, Wang X, Gao L, Bai L, Zhu R, Bai C. Regulation of MUC5AC mucin secretion by depletion of AQP5 in SPC-A1 cells. Biochem Biophys Res Commun. 2006;342:775–81.
Chen Z, Wang S, Erekosima N, Li Y, Hong J, Qi X, et al. IL-4 confers resistance to IL-27-mediated suppression on CD4+ T cells by impairing signal transducer and activator of transcription 1 signaling. J Allergy Clin Immunol. 2013;132:912–21.
Cosmi L, Maggi L, Santarlasci V, Capone M, Cardilicchia E, Frosali F, et al. Identification of a novel subset of human circulating memory CD4(+) T cells that produce both IL-17A and IL-4. J Allergy Clin Immunol. 2010;125:222–30.
Farber DL, Yudanin NA, Restifo NP. Human memory T cells: generation, compartmentalization and homeostasis. Nat Rev Immunol. 2014;14:24–35.
Fukushima A, Yamaguchi T, Ishida W, Fukata K, Udaka K, Ueno H. Mice lacking the IFN-gamma receptor or fyn develop severe experimental autoimmune uveoretinitis characterized by different immune responses. Immunogenetics. 2005;57:337–43.
Hida S, Tadachi M, Saito T, Taki S. Negative control of basophil expansion by IRF-2 critical for the regulation of Th1/Th2 balance. Blood. 2005;106:2011–7.
Kalliolias GD, Ivashkiv LBIL. 27 activates human monocytes via STAT1 and suppresses IL-10 production but the inflammatory functions of IL-27 are abrogated by TLRs and p38. J Immunol. 2008;180:6325–33.
Kim BS, Kim IK, Park YJ, Kim YS, Kim YJ, Chang WS, et al. Conversion of Th2 memory cells into Foxp3+ regulatory T cells suppressing Th2-mediated allergic asthma. Proc Natl Acad Sci U S A. 2010;107:8742–7.
Leung DY, Gao PS, Grigoryev DN, Rafaels NM, Streib JE, Howell MD, et al. Human atopic dermatitis complicated by eczema herpeticum is associated with abnormalities in IFN-gamma response. J Allergy Clin Immunol. 2011;127:965–73.
Lumsden JM, Schwenk RJ, Rein LE, Moris P, Janssens M, Ofori-Anyinam O, et al. Protective immunity induced with the RTS,S/AS vaccine is associated with IL-2 and TNF-alpha producing effector and central memory CD4 T cells. PLoS One. 2011;6:e20775.
MacLeod MK, Clambey ET, Kappler JW, Marrack P. CD4 memory T cells: what are they and what can they do? Semin Immunol. 2009;21:53–61.
MacLeod MK, Kappler JW, Marrack P. Memory CD4 T cells: generation, reactivation and re-assignment. Immunol. 2010;130:10–5.
Miklossy G, Hilliard TS, Turkson J. Therapeutic modulators of STAT signalling for human diseases. Nat Rev Drug Discov. 2013;12:611–29.
Mojtabavi N, Dekan G, Stingl G, Epstein MM. Long-lived Th2 memory in experimental allergic asthma. J Immunol. 2002a;169:4788–96.
Mojtabavi N, Dekan G, Stingl G, Epstein MM. Long-lived Th2 memory in experimental allergic asthma. J Immunol. 2002b;169:4788–96.
Nakagome K, Dohi M, Okunishi K. To Y, Sato A, Komagata Y, et al. Antigen-sensitized CD4+CD62Llow memory/effector T helper 2 cells can induce airway hyperresponsiveness in an antigen free setting. Respir Res. 2005;6:46–52.
Ouaked N, Mantel PY, Bassin C, Burgler S, Siegmund K, Akdis CA. Regulation of the foxp3 gene by the Th1 cytokines: the role of IL-27-induced STAT1. J Immunol. 2009;182:1041–9.
Rudin A, Macaubas C, Wee C, Holt BJ, Slya PD, Holt PG. Bystander" amplification of PBMC cytokine responses to seasonal allergen in polysensitized atopic children. Allergy. 2001;56:1042–8.
Sathaliyawala T, Kubota M, Yudanin N, Turner D, Camp P, Thome JJ, et al. Distribution and compartmentalization of human circulating and tissue-resident memory T cell subsets. Immunity. 2013;38:187–97.
Seder RA, Darrah PA, Roederer M. T-cell quality in memory and protection: implications for vaccine design. Nat Rev Immunol. 2008;8:247–58.
Tang X, Chen XK. Z1. Memory T cells and asthma. Zhonghua Jie He He Hu Xi Za Zhi. 2015;38:69–71.
Turner DL, Farber DL. Mucosal resident memory CD4 T cells in protection and immunopathology. Front Immunol. 2014;5:331.
Wang YH, Voo KS, Liu B, Chen CY, Uygungil B, Spoede W, et al. A novel subset of CD4(+) T(H)2 memory/effector cells that produce inflammatory IL-17 cytokine and promote the exacerbation of chronic allergic asthma. J Exp Med. 2010;207:2479–91.
Wilkinson TM, Li CK, Chui CS, Huang AK, Perkins M, Liebner JC, et al. Preexisting influenza-specific CD4+ T cells correlate with disease protection against influenza challenge in humans. Nat Med. 2012;18:274–80.
Xiaoqiong S, Jue P, Jianjun J, et al. Intranasal administration of interleukin-27 alleviate the airway allergic inflammation of ovalbumin-induced mouse asthma model via the STAT1 signal pathway. Zhongguo Hu; Xi He Wei Zhong Jian Hu. 2015;14(5):425–31.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81270078 and 81470211 to ZHC) and the “Zhengyi” Scholar Program of Fudan University (JYH6273202/002/023/014 to GS and SXC).
Author contributions
ZHC, XDW, LZ, and JP conceived and designed the study. ZHC, DDL, ZHM, XQS, HLY, GS, and SXC performed the biological experiments. YJ was responsible for the statistical analysis. ZHC and JP wrote the paper. All the authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Additional information
Zhihong Chen and Jue Pan contributed equally to this work.
Electronic supplementary material
Suppl Fig. 1
No liner correlations between IL-17+CD4+Tm cell number and several clinical characteristics (Eos, IgE and FEV1) (PDF 169 kb)
Rights and permissions
About this article
Cite this article
Chen, Z., Pan, J., Jia, Y. et al. Effect of memory CD4+ T cells’ signal transducer and activator of transcription (STATs) functional shift on cytokine-releasing properties in asthma. Cell Biol Toxicol 33, 27–39 (2017). https://doi.org/10.1007/s10565-016-9357-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-016-9357-6