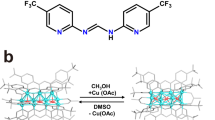
Abstract
A hydride-centered dithiophosphate cluster [Cu8(μ4-H){S2P(OEt)2}6](PF6) (1)] previously developed by us was applied as a new catalyst to the 1,3-dipolar cycloaddition of organic azides and alkynes for preparing substituted trizoles. With the required catalyst loading as low as 0.4 mol%, the reactions of terminal alkynes with BnN3 all proceeded smoothly at ambient temperature in CH3CN to exclusively produce 1,4-triazoles in good yields. For these reactions, it is assumed that the formation of the requisite copper acetylide intermediate is facilitated by the abstraction of the terminal hydrogen of alkynes by the hydride released from the central of the cluster. With only few examples being documented in literatures, the reactions of a range of internal alkynes have also been realized under the catalysis of 1 (0.8 mol%) in DMF at elevated temperature, to yield 1,4,5-trisubstituted triazoles in moderate to high yields. Our study has provided a preliminary insight into the effect of sulfur-based ligands on the activity of copper ion.
Graphical Abstract







Similar content being viewed by others
Notes
The hydride-donating ability of 1 has recently been realized by its ability to reduce 2-cyclohexenone into cyclohexanone. Treatment of 2-cyclohexenone (10 mg, 0.104 mmol) with 1 (624 mg) in THF (25 mL) at ambient temperature for 12 h resulted in 30 % conversion into cyclohexanone as monitored by gas chromatography
References
Tornøe CW, Christensen C, Meldal M (2002) J Org Chem 67:3057
Tornøe CW, Meldal M (2001) Peptidotriazoles: Copper(I)-catalyzed 1,3-dipolar cycloaddition on solid-phase, peptide 2001. In: Proceedings american peptide symposium. American Peptides Society and Kluwer, San Diego, pp 263–264
Rostovtsev VV, Green LG, Fokin VV, Sharpless KB (2002) Angew Chem Int Ed 41:2596
Lahann J (ed) (2009) Click chemistry for biotechnology and material science. Wiley, Chichester
Whiting M, Tripp JC, Lin YC, Lindstrom W, Olson AJ, Elder JH, Sharpless KB, Fokin VV (2006) J Med Chem 49:7697
Aragão-Leoneti V, Campo VL, Gomes AS, Field RA, Carvalho I (2010) Tetrahedron 66:9475
Bock VD, Perciaccante R, Jansen TP, Hiemstra H, van Maarseveen JH (2006) Org Lett 8:919
Angell YL, Burgess K (2007) Chem Soc Rev 36:1674
Kalesh KA, Shi H, Ge J, Yao SQ (2010) Org Biomol Chem 8:1749
Amblard F, Cho JH, Schinazi RF (2009) Chem Rev 109:4207
Lutz JF (2007) Angew Chem Int Ed 46:1018
Johnson JA (2002) Biomacromolecules 37
Qin A, Lam JWY, Tang BZ (2010) Chem Soc Rev 39:2522
Iha RK, Wooley KL, Nyström AM, Burke DJ, Kade MJ, Hawker CJ (2009) Chem Rev 109:5620
Evans RA (2007) Aust J Chem 60:384
Huisgen R, Szeimies G, Moebius L (1967) Chem Ber 100:2494
Cuevas F, Oliva AI, Pericas M A (2010) Synlett 1873
Fu X, Albermann C, Zhang CC, Thorson JS (2005) Org Lett 7:1513
Himo F, Lovell T, Hilgraf R, Rostovtsev VV, Noodleman L, Sharpless KB, Fokin VV (2005) J Am Chem Soc 127:210
van Dijk M, Rijkers DTS, Liskamp RM, van Nostrum CF, Hennink WE (2009) Bioconjugate Chem 20:2001
Jin T, Yan M, Yamamoto Y (2012) ChemCatChem 4:1217
Meldal M, Tornøe CW (2008) Chem Rev 108:2952
Díez-González S, Correa A, Cavallo L, Nolan SP (2006) Chem Eur J 12:7558
Li PH, Wang L, Zhang Y (2008) Tetrahedron 64:10825
Lazreg F, Slawin AMZ, Cazin CSJ (2012) Organometallics 31:7969
Liao PK, Sarkar B, Chang HW, Wang JC, Liu CW (2009) Inorg Chem 48:4089
Wang F, Fu H, Jiang Y, Zhao YF (2008) Green Chem 10:452
Hein JE, Tripp JC, Krasnova LB, Sharplesss KB, Fokin VV (2009) Angew Chem Int Ed 48:8018
Alonso F, Moglie Y, Radivoy G, Yus M (2010) Eur J Org Chem 2010:1875
Kuang GC, Guha PM, Brotheron WS, Simmons JT, Stankee LA, Nguyen BT, Clark RJ, Zhu L (2011) J Am Chem Soc 133:13984
Boren BC, Narayan S, Rasmussen LK, Zhang L, Zhao H, Lin Z, Jia G, Fokin VV (2008) J Am Chem Soc 130:8923
Meng X, Xu X, Gao T, Chen B (2010) Eur J Org Chem 2010:5409
Acknowledgments
We are grateful to the National Science Council of Republic of China (Taiwan) for financial support.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, BH., Wu, CC., Fang, X. et al. [Cu8(μ4-H){S2P(OEt)2}6](PF6): A Novel Catalytic Hydride-Centered Copper Cluster for Azide-Alkyne Cycloaddtion. Catal Lett 143, 572–577 (2013). https://doi.org/10.1007/s10562-013-0993-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-013-0993-7