Abstract
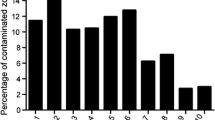
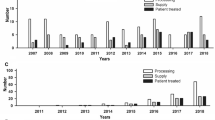
Radiation sterilization eliminates microbiological infections but causes the degradation of the cell factor. The negative result of microbiological examination for tissue transplants is one of the conditions for approval for distribution in patients. The study attempts to verify impact of the presence of microbes onto material for transplant loss. In the 2011–2015 period, we analyzed 293 donors of skin and amnion. Microbiological sampling was performed. The total of 21 strains of bacteria, molds and fungi was identified in collected tissue. The widest spectrum of strains was found in skin (17), followed by amnia (8). The total number of positive findings was 147 and was again highest in skin (129), while the number of positive findings in amnia was 18 only. The general percentage of fungal infections was very low. The presence of fungal strains was only observed in allogeneic skin (2%). Large number of microorganisms isolated from the skin before sterilization was observed, so it seems impossible to use allogeneic intravital skin. However, the intravital application of allogeneic amnion obtained from cesarean section remains to be considered.





Similar content being viewed by others
References
Aghayan HR, Goodarzi P, Baradaran-Rafii A, Larijani B, Moradabadi L, Rahim F, Arjmand B (2013) Bacterial contamination of amniotic membrane in a tissue bank from Iran. Cell Tissue Bank 14(3):401–406
Campbell DG, Li P (1999) Sterilization of HIV with irradiation: relevance to infected bone allografts. Aust N Z J Surg 69(7):517–521
Church D, Elsayed S, Reid O, Winston B, Lindsay R (2006) Burn wound infections. Clin Microbiol Rev 19(2):403–434. doi:10.1128/CMR.19.2.403-434.2006
Deijkers RL, Bloem RM, Petit PL, Brand R, Vehmeyer SB, Veen MR (1997) Contamination of bone allografts: analysis of incidence and predisposing factors. J Bone Joint Surg Br 79(1):161–166
Delloye C (1994) Tissue allografts and health risks. Acta Orthop Belg 60(Suppl. 1):62–67
Djefal A, Tahtat D, Khodja AN, Bouzid SS, Remane N (2007) Validation and substantiation of 25 kGy as sterilization dose for lyophilized human amnion membrane. Cell Tissue Bank 8(1):9–12
Dziedzic-Goclawska A, Kaminski A, Uhrynowska-Tyszkiewicz I, Stachowicz W (2005) Irradiation as a safety procedure in tissue banking. Cell Tissue Bank 6(3):201–219
Eastlund T (1995) Infectious disease transmission through cell, tissue, and organ transplantation: reducing the risk through donor selection. Cell Transpl 4(5):455–477
Eastlund T (2006) Bacterial infection transmitted by human tissue allograft transplantation. Cell Tissue Bank 7(3):147–166
Gierek M, Kawecki M, Mikuś K, Glik J, Klama-Baryła A, Hoff-Lenczewska D, Nowak M (2012a) Use of human amniotic membrane in treatment of Burns. J Orthop Trauma Surg Relat Res 3(29):70–75
Gierek M, Mikuś K, Kawecki M, Glik J, Hoff-Lenczewska D, Nowak M (2012b) The treatment of burns wounds with human amniotic membrane dressings in the Centre for the Treatment of Burns in Siemianowice Śląskie—own experience. J Orthop Trauma Surg Relat Res 3(29):76–83
Gocke DJ (2005) Tissue donor selection and safety. Clin Orthop Relat Res 435:17–21
Ibrahim T, Stafford H, Esler CN, Power RA (2004) Cadaveric allograft microbiology. Int Orthop 28(5):315–318
Ireland L, Spelman D (2005) Bacterial contamination of tissue allografts—experiences of the donor tissue bank of Victoria. Cell Tissue Bank 6(3):181–189
Journeaux SF, Johnson N, Bryce SL, Friedman SJ, Sommerville SM, Morgan DA (1999) Bacterial contamination rates during bone allograft retrieval. J Arthroplasty 14(6):677–681
Kawecki M, Hoff-Lenczewska D, Klama-Baryła A, Glik J, Łabuś W, Nowak M (2012) [Burns]. Adam Dziki, Warsaw 2012, 20th ed., pp 185-196
Klama-Baryła A, Kraut M, Łabuś W, Maj M, Kawecki M, Nowak M, Glik J, Cichowski A, Szydło A, Lesiak M, Anioł J, Sieroń AL (2011) Zastosowanie zagęszczonej masy bogato-leukocytarno-płytkowej w przeszczepach hodowanych in vitro autologicznych keratynocytów. J Orthop Trauma Surg Relat Res 2:77–86
Marsit N, Dwejen S, Saad I, Abdalla S, Shaab A, Salem S, Khanfas E, Hasan A, Mansur M, Abdul Sammad M (2014) Substantiation of 25 kGy radiation sterilization dose for banked air dried amniotic membrane and evaluation of personnel skill in influencing finished product bioburden. Cell Tissue Bank 15(4):603–611
Nguyen H, Morgan DA, Forwood MR (2007a) Sterilization of allograft bone: is 25 kGy the gold standard for gamma irradiation? Cell Tissue Bank 8(2):81–91
Nguyen H, Morgan DA, Forwood MR (2007b) Sterilization of allograft bone: effects of gamma irradiation on allograft biology and biomechanics. Cell Tissue Bank 8(2):93–105
Nguyen H, Morgan DA, Forwood MR (2007c) Sterilization of allograft bone: is 25 kGy the gold standard for gamma irradiation? Cell Tissue Bank 8(2):81–91
Saad ZM, Khoo TL, Dorai AA, Halim AS (2009) The versatility of a glycerol-preserved skin allograft as an adjunctive treatment to free flap reconstruction. Indian J Plast Surg 42:94–99
Segur JM, Almela M, Farinas O, Lazaro A, Navarro A, Trias E, Domingo A, Marco F (2005) Bone contamination and blood culture in tissue donors. Ann Transpl 10(2):11–13
Singh R, Purohit S, Chacharkar MP, Bhandari PS, Bath AS (2007) Microbiological safety and clinical efficacy of radiation sterilized amniotic membranes for treatment of second-degree burns. Burns 33(4):505–510
Stacey GN (2011) Cell culture contamination. Methods Mol Biol 731:79–91
Vehmeyer SB, Bloem RM, Deijkers RL, Veen MR, Petit PL (1999) A comparative study of blood and bone marrow cultures in cadaveric bone donation. J Hosp Infect 43(4):305–308
Vehmeyer SB, Bloem RM, Petit PL (2001) Microbiological screening of post-mortem bone donors—two case reports. J Hosp Infect 47(3):193–197
Vehmeyer S, Wolkenfelt J, Deijkers R, Petit P, Brand R, Bloem R (2002) Bacterial contamination in postmortem bone donors. Acta Orthop Scand 73(6):678–683
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kitala, D., Klama-Baryła, A., Kawecki, M. et al. Infections in the tissue material and their impact on the loss of transplants in the Laboratory of in vitro Cell and Tissue Culture with Tissue Bank in the years 2011–2015. Cell Tissue Bank 18, 61–68 (2017). https://doi.org/10.1007/s10561-016-9597-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-016-9597-y