Abstract
Purpose
Inhibition of the renin-angiotensin system (RAS) is beneficial in patient management after myocardial infarction (MI). However, whether RAS inhibition also provides cardiac protection in the acute phase of MI is unclear.
Methods
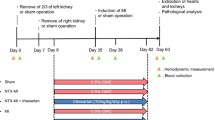
Male 129sv mice underwent coronary artery occlusion to induce MI, followed by treatment with losartan (L, 20 and 60 mg/kg), perindopril (P, 2 and 6 mg/kg), amlodipine (20 mg/kg as a BP-lowering agent) or vehicle as control. Drug effects on hemodynamics were examined. Effects of treatments on incidence of cardiac rupture, haematological profile, monocyte and neutrophil population in the spleen and the heart, cardiac leukocyte density, expression of inflammatory genes and activity of MMPs were studied after MI.
Results
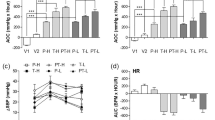
Incidence of cardiac rupture within 2 weeks was significantly and similarly reduced by both losartan (L) and perindopril (P) in a dose-dependent manner [75% (27/36) in vehicle, 40–45% in low-dose (L 10/22, P 8/20) and 16–20% (L 5/32, P 4/20) in high-dose groups, all P < 0.05]. This action was independent of their BP-lowering action, as amlodipine reduced BP to a similar degree without effect on rupture (70%, 21/30). Compared to the control group, high dose losartan and perindopril decreased counts of white blood cells, neutrophils and lymphocytes (all P < 0.05), and inhibited splenic monocyte and neutrophil release into the circulation. Consequently, monocyte, neutrophil and leukocyte infiltration, inflammatory gene expressions (IL-1β, IL-6, MMP9, MCP-1, TNF-α and TGFβ1) and activity of MMP2 and MMP9 in the infarct tissue were attenuated by losartan and/or perindopril treatment (all P < 0.05).
Conclusions
RAS inhibition by losartan or perindopril prevented cardiac rupture at the acute phase of MI through blockade of splenic release of monocytes and neutrophils and consequently attenuation of systemic and regional inflammatory responses.






Similar content being viewed by others
References
Creemers EE, Cleutjens JP, Smits JF, Daemen MJ. Matrix metalloproteinase inhibition after myocardial infarction: a new approach to prevent heart failure? Circ Res. 2001;89(3):201–10.
Jugdutt BI. Ventricular remodeling after infarction and the extracellular collagen matrix: when is enough enough? Circulation. 2003;108(11):1395–403.
Cohn JN, Ferrari R, Sharpe N. Cardiac remodeling--concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J Am Col Cardio. 2000;35(3):569–82.
Gonzalez GE, Seropian IM, Krieger ML, et al. Effect of early versus late AT(1) receptor blockade with losartan on post myocardial infarction ventricular remodeling in rabbits. Am J Physio l Heart Circ Physio l. 2009;297(1):H375–86.
Oyamada S, Bianchi C, Takai S, et al. Impact of acute myocardial ischemia reperfusion on the tissue and blood-borne renin-angiotensin system. Basic Res Cardio. 2010;105(4):513–22.
Kumar R, Singh VP, Baker KM. The intracellular renin-angiotensin system in the heart. Curr Hypertens Rep. 2009;11(2):104–10.
ISIS-4: a randomised factorial trial assessing early oral captopril, oral mononitrate, and intravenous magnesium sulphate in 58,050 patients with suspected acute myocardial infarction. ISIS-4 (Fourth International Study of Infarct Survival) Collaborative Group. Lancet. 1995;345(8951):669–85.
GISSI-3: effects of lisinopril and transdermal glyceryl trinitrate singly and together on 6-week mortality and ventricular function after acute myocardial infarction. Gruppo Italiano per lo Studio della Sopravvivenza nell’infarto Miocardico. Lancet. 1994;343(8906):1115–22.
Dickstein K, Kjekshus J. Group OSCotOS. Effects of losartan and captopril on mortality and morbidity in high-risk patients after acute myocardial infarction: the OPTIMAAL randomised trial. Optimal Trial in Myocardial Infarction with Angiotensin II Antagonist Losartan. Lancet. 2002;360(9335):752–60.
Dai W, Kloner RA. Potential role of renin-angiotensin system blockade for preventing myocardial ischemia/reperfusion injury and remodeling after myocardial infarction. Postgrad Med. 2011;123(2):49–55.
De Mello WC, Frohlich ED. Clinical perspectives and fundamental aspects of local cardiovascular and renal Renin-Angiotensin systems. Front Endocrinol (Lausanne). 2014;5:16.
Leuschner F, Panizzi P, Chico-Calero I, et al. Angiotensin-converting enzyme inhibition prevents the release of monocytes from their splenic reservoir in mice with myocardial infarction. Circ Res. 2010;107(11):1364–73.
Sciarretta S, Paneni F, Palano F, et al. Role of the renin-angiotensin-aldosterone system and inflammatory processes in the development and progression of diastolic dysfunction. Clin Sci (Lond). 2009;116(6):467–77.
Swirski FK, Nahrendorf M, Etzrodt M, et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science. 2009;325(5940):612–6.
Gao XM, White DA, Dart AM, Du XJ. Post-infarct cardiac rupture: recent insights on pathogenesis and therapeutic interventions. Pharmacol Ther. 2012;134(2):156–79.
Gao XM, Xu Q, Kiriazis H, Dart AM, Du XJ. Mouse model of post-infarct ventricular rupture: time course, strain- and gender-dependency, tensile strength, and histopathology. Cardiovasc Res. 2005;65(2):469–77.
Flynn ER, Marbury DC, Sawyer RT, et al. Amlodipine reduces inflammation despite promoting albuminuria in the streptozotocin-induced diabetic Rat. Nephron Extra. 2012;2(1):205–18.
Jenkins TA, Wong JY, Howells DW, Mendelsohn FA, Chai SY. Effect of chronic angiotensin-converting enzyme inhibition on Striatal dopamine content in the MPTP-treated mouse. J Neurochem. 1999;73(1):214–9.
Liang C, Wu ZG, Ding J, et al. Losartan inhibited expression of matrix metalloproteinases in rat atherosclerotic lesions and angiotensin II-stimulated macrophages. Acta Pharmacol Sin. 2004;25(11):1426–32.
McLennan SV, Kelly DJ, Cox AJ, et al. Decreased matrix degradation in diabetic nephropathy: effects of ACE inhibition on the expression and activities of matrix metalloproteinases. Diabetologia. 2002;45(2):268–75.
Gao XM, Liu Y, White D, et al. Deletion of macrophage migration inhibitory factor protects the heart from severe ischemia-reperfusion injury: a predominant role of anti-inflammation. J Mol Cell Cardio. 2011;50(6):991–9.
White DA, Su Y, Kanellakis P, et al. Differential roles of cardiac and leukocyte derived macrophage migration inhibitory factor in inflammatory responses and cardiac remodelling post myocardial infarction. J Mol Cell Cardio. 2014;69:32–42.
Fang L, Gao XM, Moore XL, et al. Differences in inflammation, MMP activation and collagen damage account for gender difference in murine cardiac rupture following myocardial infarction. J Mol Cell Cardio. 2007;43(5):535–44.
Bates ER. Reperfusion therapy reduces the risk of myocardial rupture complicating ST-elevation myocardial infarction. J Am Heart Assoc. 2014;3(5):e001368.
Ong HT, Ong LM, Ho JJ. Angiotensin-Converting Enzyme Inhibitors (ACEIs) and Angiotensin-Receptor Blockers (ARBs) in Patients at high risk of cardiovascular events: a meta-analysis of 10 randomised placebo-controlled trials. ISRN Cardio. 2013;2013:478597.
Christensen DJ, Ford M, Reading J, Castle CH. Effect of hypertension on myocardial rupture after acute myocardial infarction. Chest. 1977;72(5):618–22.
Figueras J, Alcalde O, Barrabes JA, et al. Changes in hospital mortality rates in 425 patients with acute ST-elevation myocardial infarction and cardiac rupture over a 30-year period. Circulation. 2008;118(25):2783–9.
Shamshad F, Kenchaiah S, Finn PV, et al. Fatal myocardial rupture after acute myocardial infarction complicated by heart failure, left ventricular dysfunction, or both: the VALsartan In Acute myocardial iNfarcTion Trial (VALIANT). Am Heart J. 2010;160(1):145–51.
Unger T. Blood pressure lowering and renin-angiotensin system blockade. J Hypertens Suppl. 2003;21(6):S3–7.
Anzai T. Post-infarction inflammation and left ventricular remodeling: a double-edged sword. Circ J. 2013;77(3):580–7.
van den Borne SW, Cleutjens JP, Hanemaaijer R, et al. Increased matrix metalloproteinase-8 and −9 activity in patients with infarct rupture after myocardial infarction. Cardiovasc Pathol. 2009;18(1):37–43.
Barron HV, Harr SD, Radford MJ, Wang Y, Krumholz HM. The association between white blood cell count and acute myocardial infarction mortality in patients > or =65 years of age: findings from the cooperative cardiovascular project. J Am Col Cardio. 2001;38(6):1654–61.
Maekawa Y, Anzai T, Yoshikawa T, et al. Prognostic significance of peripheral monocytosis after reperfused acute myocardial infarction:a possible role for left ventricular remodeling. J Am Col Cardio. 2002;39(2):241–6.
Liu Y, Gao XM, Fang L, et al. Novel role of platelets in mediating inflammatory responses and ventricular rupture or remodeling following myocardial infarction. Arterioscler Thromb Vasc Biol. 2011;31(4):834–41.
Ma Y, Yabluchanskiy A, Lindsey ML. Neutrophil roles in left ventricular remodeling following myocardial infarction. Fibrogenesis Tissue Repair. 2013;6(1):11.
Nahrendorf M, Swirski FK, Aikawa E, et al. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J Exp Med. 2007;204(12):3037–47.
Hofmann U, Beyersdorf N, Weirather J, et al. Activation of CD4+ T lymphocytes improves wound healing and survival after experimental myocardial infarction in mice. Circulation. 2012;125(13):1652–63.
Nahrendorf M, Swirski FK. Monocyte and macrophage heterogeneity in the heart. Circ Res. 2013;112(12):1624–33.
Frantz S, Nahrendorf M. Cardiac macrophages and their role in ischaemic heart disease. Cardiovasc Res. 2014;102(2):240–8.
Puga I, Cols M, Barra CM, et al. B cell-helper neutrophils stimulate the diversification and production of immunoglobulin in the marginal zone of the spleen. Nat Immunol. 2011;13(2):170–80.
Tian Y, French BA, Kron IL, Yang Z. Splenic leukocytes mediate the hyperglycemic exacerbation of myocardial infarct size in mice. Basic Res Cardio. 2015;110(4):39.
Kain V, Ingle KA, Colas RA, et al. Resolvin D1 activates the inflammation resolving response at splenic and ventricular site following myocardial infarction leading to improved ventricular function. J Mol Cell Cardio. 2015;84:24–35.
Bolger AP, Sharma R, von Haehling S, et al. Effect of interleukin-10 on the production of tumor necrosis factor-alpha by peripheral blood mononuclear cells from patients with chronic heart failure. Am J Cardio. 2002;90(4):384–9.
Dhingra S, Sharma AK, Arora RC, Slezak J, Singal PK. IL-10 attenuates TNF-alpha-induced NF kappaB pathway activation and cardiomyocyte apoptosis. Cardiovasc Res. 2009;82(1):59–66.
Kaur K, Sharma AK, Dhingra S, Singal PK. Interplay of TNF-alpha and IL-10 in regulating oxidative stress in isolated adult cardiac myocytes. J Mol Cell Cardio. 2006;41(6):1023–30.
Danielsen CC, Wiggers H, Andersen HR. Increased amounts of collagenase and gelatinase in porcine myocardium following ischemia and reperfusion. J Mol Cell Cardio. 1998;30(7):1431–42.
White DA, Fang L, Chan W, et al. Pro-inflammatory action of MIF in acute myocardial infarction via activation of peripheral blood mononuclear cells. PLoS One. 2013;8(10):e76206.
Jin Y, Han HC, Lindsey ML. ACE inhibitors to block MMP-9 activity: new functions for old inhibitors. J Mol Cell Cardio. 2007;43(6):664–6.
Lods N, Ferrari P, Frey FJ, et al. Angiotensin-converting enzyme inhibition but not angiotensin II receptor blockade regulates matrix metalloproteinase activity in patients with glomerulonephritis. J Am Soc Nephrol. 2003;14(11):2861–72.
Yamamoto D, Takai S, Miyazaki M. Inhibitory profiles of captopril on matrix metalloproteinase-9 activity. Eur J Pharmacol. 2008;588(2–3):277–9.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study was funded by grants (ID 1081710, 1004235) from the National Health and Medical Research Council (NHMRC) of Australia and the Victorian Government’s Operational Infrastructure Support Program. A.J.M. is supported by a career development fellowship from the NHMRC (ID1085752), a future leader fellowship from the National Heart Foundation (ID100440). X.J.D. and A.M.D are NHMRC research fellow (ID1043026 and ID586656).
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
The mouse was used in this study. All experimental protocols were approved by a local Animal Ethics Committee and complied with the Australian Code for the Care and Use of Animals for Scientific Purposes (8th edition, 2013).
Rights and permissions
About this article
Cite this article
Gao, XM., Tsai, A., Al-Sharea, A. et al. Inhibition of the Renin-Angiotensin System Post Myocardial Infarction Prevents Inflammation-Associated Acute Cardiac Rupture. Cardiovasc Drugs Ther 31, 145–156 (2017). https://doi.org/10.1007/s10557-017-6717-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-017-6717-2