Abstract
Purpose
Dofetilide is class III antiarrhythmic agent which prolongs cardiac action potential duration because of selective inhibition of I Kr, the rapid component of the delayed rectifier K+ current. Although clinical studies reported on proarrhythmic risk associated with dofetilide treatment, the contributing electrophysiological mechanisms remain poorly understood. This study was designed to determine if dofetilide-induced proarrhythmia may be attributed to abnormalities in ventricular repolarization and refractoriness.
Methods
The monophasic action potential duration and effective refractory periods (ERP) were assessed at distinct epicardial and endocardial sites along with volume-conducted ECG recordings in isolated, perfused guinea-pig heart preparations.
Results
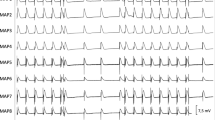
Dofetilide was found to produce the reverse rate-dependent prolongation of ventricular repolarization, increased the steepness of action potential duration rate adaptation, and amplified transepicardial variability in electrical restitution kinetics. Dofetilide also prolonged the T peak-to-end interval on ECG, and elicited a greater prolongation of endocardial than epicardial ERP, thereby increasing transmural dispersion of refractoriness. At epicardium, dofetilide prolonged action potential duration to a greater extent than ERP, thus extending the critical interval for ventricular re-excitation. This change was associated with triangulation of epicardial action potential because of greater dofetilide-induced prolonging effect at 90 % than 30 % repolarization. Premature ectopic beats and spontaneous short-lasting episodes of monomorphic ventricular tachycardia were observed in 44 % of dofetilide-treated heart preparations.
Conclusions
Proarrhythmic potential of dofetilide in guinea-pig heart is attributed to steepened electrical restitution, increased transepicardial variability in electrical restitution kinetics, amplified transmural dispersion of refractoriness, increased critical interval for ventricular re-excitation, and triangulation of epicardial action potential.







Similar content being viewed by others
References
Bashir Y, Thomsen PEB, Kingma JH, et al. Electrophysiologic profile and efficacy of intravenous dofetilide (UK-68,798) a new class III antiarrhythmic drug, in patients with sustained monomorphic ventricular tachycardia. Am J Cardiol. 1995;76:1040–4.
Bazett HC. An analysis of the time-relations of electrocardiogram. Heart. 1920;7:353–70.
Bianconi L, Castro A, Dinelli M, et al. Comparison of intravenously administered dofetilide versus amiodarone in the acute termination of atrial fibrillation and flutter. Eur Heart J. 2000;21:1265–73.
Boyett MR, Jewell BR. Analysis of the effects of changes in rate and rhythm upon electrical activity in the heart. Prog Biophys Mol Biol. 1980;36:1–52.
Brahmajothi MV, Morales MJ, Reimer KA, Strauss HC. Regional localization of ERG, the channel protein responsible for the rapid component of the delayed rectifier, K+ current in the ferret heart. Circ Res. 1997;81:128–35.
Bryant SM, Wan X, Shipsey SJ, Hart G. Regional differences in the delayed rectifier current (I Kr and I Ks) contribute to the differences in action potential duration in basal left ventricular myocytes in guinea-pig. Cardiovasc Res. 1998;40:322–31.
Carmeliet E. Voltage—and time-dependent block of the delayed K+ current in cardiac myocytes by dofetilide. J Pharmacol Exp Ther. 1992;262:809–17.
Cheng J, Kamiya K, Liu W, Tsuji Y, Toyama J, Kodama I. Heterogeneous distribution of the two components of delayed rectifier K + current: a potential mechanism of the proarrhythmic effects of methanesulfonanilide class III agents. Cardiovasc Res. 1999;43:135–47.
Choy AMJ, Darbar D, Dell’Orto S, Roden DM. Exaggerated QT prolongation after cardioversion of atrial fibrillation. J Am Col Cardiol. 1999;34:396–401.
Darbar D, Hardin B, Harris P, Roden DM. A rate-independent method of assessing QT-RR slope following conversion of atrial fibrillation. J Cardiovasc Electrophysiol. 2007;18:636–41.
Diaz A, Bourassa MG, Guertin MC, Tardif JC. Long-term prognostic value of resting heart rate in patients with suspected or proven coronary artery disease. Eur Heart J. 2005;26:967–74.
Du CY, El Harchi A, Zhang YH, Orchard CH, Hancox JC. Pharmacological inhibition of the hERG potassium channel is modulated by extracellular but not intracellular acidosis. J Cardiovasc Electrophysiol. 2011;22:1163–70.
Duff HJ, Feng ZP, Fiset C, Wang L, Lees-Miller J, Sheldon RS. [3H]Dofetilide binding to cardiac myocytes: modulation by extracellular potassium. J Mol Cell Cardiol. 1997;29:183–91.
Falk RH, Pollak A, Singh SN, Friedrich T. Intravenous dofetilide, a class III antiarrhythmic agent, for the termination of sustained atrial fibrillation or flutter. J Am Col Cardiol. 1997;29:385–90.
Garfinkel A, Kim YH, Voroshilovsky O, et al. Preventing ventricular fibrillation by flattening cardiac restitution. Proc Natl Acad Sci USA. 2000;97:6061–6.
Gibbs M, Veliotes DG, Anamourlis C, et al. Chronic beta-adrenoreceptor activation increases cardiac cavity size through chamber remodelling and not via modifications in myocardial material properties. Am J Physiol Heart Circ Physiol. 2004;287:H2762–7.
Gjini V, Korth M, Schreieck J, Weyerbrock S, Schomig A, Schmitt C. Differential class III antiarrhythmic effects of ambasilide and dofetilide at different extracellular potassium and pacing frequencies. J Cardiovasc Pharmacol. 1996;28:314–20.
Grassi G, Seravalle G, Quarti-Trevano F, et al. Sympathetic and baroreflex cardiovascular control in hypertension-related left ventricular dysfunction. Hypertension. 2009;53:205–9.
Hondeghem L, Carlsson L, Duker G. Instability and triangulation of the action potential predict serious proarrhythmia, but APD prolongation is antiarrhythmic. Circulation. 2001;103:2004–13.
Jurkiewicz NK, Sanguinetti MC. Rate-dependent prolongation of cardiac action potentials by a methanesulfonanilide class III antiarrhythmic agent. Circ Res. 1993;72:75–83.
Koller ML, Riccio ML, Gilmour RF. Dynamic restitution of action potential duration during electrical alternans and ventricular fibrillation. Am J Physiol Heart Circ Physiol. 1998;275:H1635–42.
Laurita KR, Girouard SD, Rosenbaum DS. Modulation of ventricular repolarization by a premature stimulus. Role of epicardial dispersion of repolarization kinetics demonstrated by optical mapping of the intact guinea-pig heart. Circ Res. 1996;79:493–503.
Marschang H, Beyer T, Karolyi L, Kubler W, Brachmann J. Differential rate and potassium-dependent effects of the class III agents d-sotalol and dofetilide on guinea-pig papillary muscle. Cardiovasc Drugs Ther. 1998;12:573–83.
Nabauer M, Kaab S. Potassium channel down-regulation in heart failure. Cardiovasc Res. 1998;37:324–34.
Nash MP, Bradley CP, Sutton PM, et al. Whole heart action potential duration restitution properties in cardiac patients: a combined clinical and modelling study. Exp Physiol. 2006;91:339–54.
Osadchii O, Norton G, Deftereos D, Woodiwiss A. Rat strain-related differences in myocardial adrenergic tone and the impact on cardiac fibrosis, adrenergic responsiveness and myocardial structure and function. Pharmacol Res. 2007;55:287–94.
Osadchii O, Woodiwiss A, Alves N, Norton G. Mechanisms of preserved baseline cardiac systolic function in rats with adrenergic inotropic downregulation. Life Sci. 2005;78:366–75.
Osadchii OE. Mechanisms of hypokalemia-induced ventricular arrhythmogenicity. Fund Clin Pharmacol. 2010;24:547–59.
Osadchii OE, Larsen AP, Olesen SP. Predictive value of electrical restitution in hypokalemia-induced ventricular arrhythmogenicity. Am J Physiol Heart Circ Physiol. 2010;298:H210–20.
Osadchii OE, Olesen SP. Electrophysiological determinants of hypokalemia-induced arrhythmogenicity in the guinea-pig heart. Acta Physiol (Oxf). 2009;197:273–87.
Osadchii OE. Effects of ventricular pacing protocol on electrical restitution assessments in guinea-pig heart. Exp Physiol. 2012;97:807–21.
Pak HN, Hong SJ, Hwang GS, et al. Spatial dispersion of action potential duration restitution kinetics is associated with induction of ventricular tachycardia/fibrillation in humans. J Cardiovasc Electrophysiol. 2004;15:1357–63.
Riccio ML, Koller ML, Gilmour RF. Electrical restitution and spatiotemporal organization during ventricular fibrillation. Circ Res. 1999;84:955–63.
Sabir IN, Fraser JA, Killeen MJ, Grace AA, Huang CL. The contribution of refractoriness to arrhythmic substrate in hypokalemic Langendorff-perfused murine hearts. Pflugers Arch. 2007;454:209–22.
Sanguinetti MC, Jurkiewicz NK. Role of external Ca2+ and K+ in gating of cardiac delayed rectifier K+ currents. Pflugers Arch. 1992;420:180–6.
Scamps F, Carmeliet E. Delayed K+ current and external K+ in single cardiac Purkinje cells. Am J Physiol Cell Physiol. 1989;257:C1086–92.
Sedgwick ML, Rasmussen HS, Cobbe SM. Effects of the class III antiarrhythmic drug dofetilide on ventricular monophasic action potential duration and QT interval dispersion in stable angina pectoris. Am J Cardiol. 1992;70:1432–7.
Sedgwick ML, Rasmussen HS, Cobbe SM. Clinical and electrophysiologic effects of intravenous dofetilide (UK-68,798), a new class III antiarrhythmic drug, in patients with angina pectoris. Am J Cardiol. 1992;69:513–7.
Shah RR, Hondeghem LM. Refining detection of drug-induced proarrhythmia: QT interval and TRIaD. Hear Rhythm. 2005;2:758–72.
Singh S, Zoble RG, Yellen L, et al. Efficacy and safety of oral dofetilide in converting to and maintaining sinus rhythm in patients with chronic atrial fibrillation or atrial flutter. Circulation. 2000;102:2385–90.
Soltysinska E, Olesen SP, Osadchii OE. Myocardial structural, contractile and electrophysiological changes in the guinea-pig heart failure model induced by chronic sympathetic activation. Exp Physiol. 2011;96:647–63.
Tomaselli GF, Marban E. Electrophysiological remodelling in hypertrophy and heart failure. Cardiovasc Res. 1999;42:270–83.
Torp-Pedersen C, Moller M, Bloch-Thomsen PE, et al. Dofetilide in patients with congestive heart failure and left ventricular dysfunction. New Engl J Med. 1999;341:857–65.
Volders PGA, Vos MA, Szabo B, et al. Progress in the understanding of cardiac early afterdepolarizations and torsade de pointes: time to revise current concepts. Cardiovasc Res. 2000;46:376–92.
Weiss JN, Garfinkel A, Karaguezian HS, Qu Z, Chen PS. Chaos and the transition to ventricular fibrillation. A new approach to antiarrhythmic drug evaluation. Circulation. 1999;99:2819–26.
Yan GX, Antzelevitch C. Cellular basis for the normal T wave and the electrocardiographic manifestations of the long QT syndrome. Circulation. 1998;98:1928–36.
Yang T, Tande PM, Lathrop DA, Refsum H. Effects of altered extracellular potassium and pacing cycle length on the class III antiarrhythmic actions of dofetilide (UK-68,798) in guinea-pig papillary muscle. Cardiovasc Drugs Ther. 1992;6:429–36.
Yang T, Roden DM. Extracellular potassium modulation of drug block of I Kr. Circulation. 1996;93:407–11.
Zeng J, Laurita KR, Rosenbaum DS, Rudy Y. Two components of the delayed rectifier K+ current in ventricular myocytes of the guinea-pig type. Theoretical formulation and their role in repolarization. Circ Res. 1995;77:140–52.
Acknowledgments
This study was supported by the Novo Nordisk Foundation.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Osadchii, O.E. Dofetilide Promotes Repolarization Abnormalities in Perfused Guinea-pig Heart. Cardiovasc Drugs Ther 26, 489–500 (2012). https://doi.org/10.1007/s10557-012-6405-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-012-6405-1