Abstract
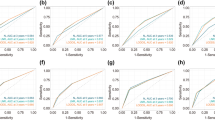
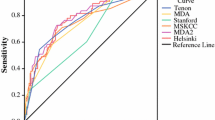
To present a systemic review and meta-analysis to evaluate the nomograms developed to predict non-sentinel lymph node (NSLN) metastasis in breast cancer patients. We focused on the six nomograms (Cambridge, MSKCC, Mayo, MDA, Tenon, and Stanford) that are the most widely validated. The AUCs were converted to odds ratios for the meta-analysis. In total, the Cambridge, Mayo, MDA, MSKCC, Stanford, and Tenon models were validated in 2,156, 2,431, 843, 8,143, 3,700, and 3,648 patients, respectively. The pooled AUCs for the Cambridge, MDA, MSKCC, Mayo, Tenon, and Stanford models were 0.721, 0.706, 0.715, 0.728, 0.720, and 0.688, respectively. Subgroup analysis revealed that in populations with a higher micrometastasis rate in the SLNs, the Tenon and Stanford models had a significantly higher predictive accuracy. A meta-regression analysis revealed that the SLN micrometastasis rate, but not the NSLN-positivity rate, was associated with improved predictive accuracy in the Tenon and Stanford models. The performance of the MSKCC and Cambridge models was not influenced by these two factors. All of these prediction models perform better than random chance. The Stanford model seems to be relatively inferior to the other models. The accuracy of the Tenon and Stanford models is influenced by the tumor burden in the SLNs.







Similar content being viewed by others
References
Krag DN et al (2010) Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: overall survival findings from the NSABP B-32 randomised phase 3 trial. Lancet Oncol 11(10):927–933
Giuliano AE et al (2011) Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. J Am Med Assoc 305(6):569–575
Chen K et al (2012) Validation and comparison of models to predict non-sentinel lymph node metastasis in breast cancer patients. Cancer Sci 103(2):274–281
Pal A et al (2008) A model for predicting non-sentinel lymph node metastatic disease when the sentinel lymph node is positive. Br J Surg 95(3):302–309
Chagpar AB et al (2006) Prediction of sentinel lymph node-only disease in women with invasive breast cancer. Am J Surg 192(6):882–887
Zee KJ et al (2003) A nomogram for predicting the likelihood of additional nodal metastases in breast cancer patients with a positive sentinel node biopsy. Ann Surg Oncol 10(10):1140–1151
Saidi RF et al (2004) Nonsentinel lymph node status after positive sentinel lymph node biopsy in early breast cancer. Am Surg 70(2):101–105 discussion 105
Degnim AC et al (2005) Nonsentinel node metastasis in breast cancer patients: assessment of an existing and a new predictive nomogram. Am J Surg 190(4):543–550
Cho J et al (2008) A scoring system to predict nonsentinel lymph node status in breast cancer patients with metastatic sentinel lymph nodes: a comparison with other scoring systems. Ann Surg Oncol 15(8):2278–2286
Hwang RF et al (2003) Clinicopathologic factors predicting involvement of nonsentinel axillary nodes in women with breast cancer. Ann Surg Oncol 10(3):248–254
Kohrt HE et al (2008) New models and online calculator for predicting non-sentinel lymph node status in sentinel lymph node positive breast cancer patients. BMC Cancer 8:66
Barranger E et al (2005) An axilla scoring system to predict non-sentinel lymph node status in breast cancer patients with sentinel lymph node involvement. Breast Cancer Res Treat 91(2):113–119
Perhavec A et al (2009) Ljubljana nomograms for predicting the likelihood of non-sentinel lymph node metastases in breast cancer patients with a positive sentinel lymph node. Breast Cancer Res Treat 119(2):357–366
Coufal O et al (2009) Predicting non-sentinel lymph node status after positive sentinel biopsy in breast cancer: what model performs the best in a czech population? Pathol Oncol Res 15(4):733–740
Walter SD, Sinuff T (2007) Studies reporting ROC curves of diagnostic and prediction data can be incorporated into meta-analyses using corresponding odds ratios. J Clin Epidemiol 60(5):530–534
Berrang TS et al (2012) Which prediction models best identify additional axillary disease after a positive sentinel node biopsy for breast cancer? Breast Cancer Res Treat 133:695–702
Cripe MH et al (2006) The likelihood of additional nodal disease following a positive sentinel lymph node biopsy in breast cancer patients: validation of a nomogram. Am J Surg 192(4):484–487
Dauphine CE et al (2007) Evaluation of three scoring systems predicting non sentinel node metastasis in breast cancer patients with a positive sentinel node biopsy. Ann Surg Oncol 14(3):1014–1019
Ponzone R et al (2007) Comparison of two models for the prediction of nonsentinel node metastases in breast cancer. Am J Surg 193(6):686–692
Zgajnar J et al (2007) Low performance of the MSKCC nomogram in preoperatively ultrasonically negative axillary lymph node in breast cancer patients. J Surg Oncol 96(7):547–553
Klar M et al (2008) The MSKCC nomogram for prediction the likelihood of non-sentinel node involvement in a German breast cancer population. Breast Cancer Res Treat 112(3):523–531
Poirier E et al (2008) Analysis of clinical applicability of the breast cancer nomogram for positive sentinel lymph node: the Canadian experience. Ann Surg Oncol 15(9):2562–2567
Coutant C et al (2009) Comparison of models to predict nonsentinel lymph node status in breast cancer patients with metastatic sentinel lymph nodes: a prospective multicenter study. J Clin Oncol 27(17):2800–2808
Coutant C et al (2009) Validation of the Tenon breast cancer score for predicting non-sentinel lymph node status in breast cancer patients with sentinel lymph node metastasis: a prospective multicenter study. Breast Cancer Res Treat 113(3):537–543
Gur AS et al (2009) Predictive probability of four different breast cancer nomograms for nonsentinel axillary lymph node metastasis in positive sentinel node biopsy. J Am Coll Surg 208(2):229–235
Scow JS et al (2009) Assessment of the performance of the Stanford online calculator for the prediction of nonsentinel lymph node metastasis in sentinel lymph node-positive breast cancer patients. Cancer 115(18):4064–4070
van la Parra RF et al (2009) Validation of a nomogram to predict the risk of nonsentinel lymph node metastases in breast cancer patients with a positive sentinel node biopsy: validation of the MSKCC breast nomogram. Ann Surg Oncol 16(5):1128–1135
D’Eredita G et al (2010) Sentinel lymph node micrometastasis and risk of non-sentinel lymph node metastasis: validation of two breast cancer nomograms. Clin Breast Cancer 10(6):445–451
Moghaddam Y et al (2010) Comparison of three mathematical models for predicting the risk of additional axillary nodal metastases after positive sentinel lymph node biopsy in early breast cancer. Br J Surg 97(11):1646–1652
Sanjuan A et al (2010) Predicting non-sentinel lymph node status in breast cancer patients with sentinel lymph node involvement: evaluation of two scoring systems. Breast J 16(2):134–140
van den Hoven I et al (2010) Value of Memorial Sloan–Kettering Cancer Center nomogram in clinical decision making for sentinel lymph node-positive breast cancer. Br J Surg 97(11):1653–1658
D’Eredita G et al (2011) Comparison of two models for predicting non-sentinel lymph node metastases in sentinel lymph node-positive breast cancer patients. Updates Surg 63(3):163–170
Hessman CJ et al (2011) Comparative validation of online nomograms for predicting nonsentinel lymph node status in sentinel lymph node-positive breast cancer. Arch Surg 146(9):1035–1040
Hidar S et al (2011) Validation of nomograms to predict the risk of non-sentinels lymph node metastases in North African Tunisian breast cancer patients with sentinel node involvement. Breast 20(1):26–30
Lombardi A et al (2011) Non-sentinel lymph node metastases in breast cancer patients with a positive sentinel lymph node: validation of five nomograms and development of a new predictive model. Tumori 97(6):749–755
Orsoni M et al (2011) Axillary lymph node dissection in the case of sentinel lymph node micrometastatic invasion: evaluation of three predictive models. Eur J Obstet Gynecol Reprod Biol 158(2):334–337
Tan EY et al (2011) Predictors of nonsentinel nodal involvement to aid intraoperative decision making in breast cancer patients with positive sentinel lymph nodes. ISRN Oncol 2011:539503
Andersson Y et al (2012) Prediction of non-sentinel lymph node status in breast cancer patients with sentinel lymph node metastases: evaluation of the tenon score. Breast Cancer (Auckl) 6:31–38
Sasada T et al (2012) Memorial Sloan–Kettering Cancer Center Nomogram to predict the risk of non-sentinel lymph node metastasis in Japanese breast cancer patients. Surg Today 42(3):245–249
Mittendorf EA et al (2012) Incorporation of sentinel lymph node metastasis size into a nomogram predicting nonsentinel lymph node involvement in breast cancer patients with a positive sentinel lymph node. Ann Surg 255(1):109–115
Fisher B et al (2002) Twenty-five-year follow-up of a randomized trial comparing radical mastectomy, total mastectomy, and total mastectomy followed by irradiation. N Engl J Med 347(8):567–575
Galimberti V, Cole BF, Zurrida S, Update of International Breast Cancer Study Group Trial 23–01 et al (2011) To compare axillary dissection versus no axillary dissection in patients with clinically node negative breast cancer and micrometastases in the sentinel node. Cancer Res 71(24 Suppl):102s
Cody HS 3rd, Houssami N (2012) Axillary management in breast cancer: what’s new for 2012? Breast 21(3):411–415
Kim T, Giuliano AE, Lyman GH (2006) Lymphatic mapping and sentinel lymph node biopsy in early-stage breast carcinoma: a metaanalysis. Cancer 106(1):4–16
Guth U et al (2012) The post ACOSOG Z0011 era: does our new understanding of breast cancer really change clinical practice? Eur J Surg Oncol 38(8):645–650
Meretoja TJ et al (2012) International multicenter tool to predict the risk of nonsentinel node metastases in breast cancer. J Natl Cancer Inst
Lee HS et al (2012) Predicting nonsentinel lymph node metastasis using lymphoscintigraphy in patients with breast cancer. J Nucl Med 53(11):1693–1700
Glasgow PD, Satchidanand N, Kowdley GC (2012) The predictive value of micrometastasis in nonsentinel lymph nodes. Am Surg 78(6):669–674
Ozbas S et al (2012) Predicting the likelihood of nonsentinel lymph node metastases in triple negative breast cancer patients with a positive sentinel lymph node: Turkish Federation of Breast Disease Associations protocol MF09-01. Clin Breast Cancer 12(1):63–67
Meretoja TJ et al (2012) A simple nomogram to evaluate the risk of nonsentinel node metastases in breast cancer patients with minimal sentinel node involvement. Ann Surg Oncol 19(2):567–576
Haffty BG et al (2011) Positive sentinel nodes without axillary dissection: implications for the radiation oncologist. J Clin Oncol 29(34):4479–4481
Shahar KH et al (2004) Factors predictive of having four or more positive axillary lymph nodes in patients with positive sentinel lymph nodes: implications for selection of radiation fields. Int J Radiat Oncol Biol Phys 59(4):1074–1079
Katz A et al (2008) Nomogram for the prediction of having four or more involved nodes for sentinel lymph node-positive breast cancer. J Clin Oncol 26(13):2093–2098
Kapur U et al (2007) Prediction of nonsentinel lymph node metastasis in sentinel node-positive breast carcinoma. Ann Diagn Pathol 11(1):10–12
Acknowledgments
We thank Dr. Zhaopeng Cai for extensive discussion of this research. This study was supported by the National Natural Science Foundation of China (Grant 81172524/H1622).
Conflict of interest
The authors have no conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Liling Zhu, Liang Jin, and Shunrong Li contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhu, L., Jin, L., Li, S. et al. Which nomogram is best for predicting non-sentinel lymph node metastasis in breast cancer patients? A meta-analysis. Breast Cancer Res Treat 137, 783–795 (2013). https://doi.org/10.1007/s10549-012-2360-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-2360-6