Abstract
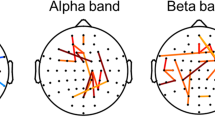
Evaluation of consciousness needs to be supported by the evidence of brain activation during external stimulation in patients with unresponsive wakefulness syndrome (UWS). Assessment of patients should include techniques that do not depend on overt motor responses and allow an objective investigation of the spontaneous patterns of brain activity. In particular, electroencephalography (EEG) coherence allows to easily measure functional relationships between pairs of neocortical regions and seems to be closely correlated with cognitive or behavioral measures. Here, we show the contribution of higher order associative cortices of patients with disorder of consciousness (N = 26) in response to simple sensory stimuli, such as visual, auditory and noxious stimulation. In all stimulus modalities an increase of short-range parietal and long-range fronto-parietal coherences in gamma frequencies were seen in the controls and minimally conscious patients. By contrast, UWS patients showed no significant modifications in the EEG patterns after stimulation. Our results suggest that UWS patients can not activate associative cortical networks, suggesting a lack of information integration. In fact, fronto-parietal circuits result to be connectively disrupted, conversely to patients that exhibit some form of consciousness. In the light of this, EEG coherence can be considered a powerful tool to quantify the involvement of cognitive processing giving information about the integrity of fronto-parietal network. This measure can represent a new neurophysiological marker of unconsciousness and help in determining an accurate diagnosis and rehabilitative intervention in each patient.


Similar content being viewed by others
References
AlA Fingelkurts, AnA Fingelkurts, Bagnato S, Boccagni C, Galardi G (2012) EEG oscillatory states as neuro-phenomenology of consciousness as revealed from patients in vegetative and minimally conscious states. Conscious Cogn 21:149–169
AnA Fingelkurts, AlA Fingelkurts, Bagnato S, Boccagni C, Galardi G (2013a) Prognostic value of resting-state electroencephalography structure in disentangling vegetative and minimally conscious states: a preliminary study. Neurorehabil Neural Repair 27:345–354
AnA Fingelkurts, AlA Fingelkurts, Bagnato S, Boccagni C, Galardi G (2013b) Dissociation of vegetative and minimally conscious patients based on brain operational architectonics: factor of etiology. Clin EEG Neurosci 44:209–220
Ansell BJ, Keenan JE (1989) The western neuro sensory stimulation profile: a tool for assessing slow-to-recover head-injured patients. Arch Phys Med Rehabil 70:104–108
Beaumont JG, Kenealy PM (2005) Incidence and prevalence of the vegetative and minimally conscious states. Neuropsychol Rehab 15:184–189
Boly M, Massimini M, Tononi G (2009) Theoretical approaches to the diagnosis of altered states of consciousness. Prog Brain Res 177:383–398
Brunet N, Bosman CA, Roberts M, Oostenveld R, Womelsdorf T, De Weerd P, Fries P (2013) visual cortical gamma-band activity during free viewing of natural images.Cereb Cortex Oct 9 (in press)
Cavinato M, Volpato C, Silvoni S, Sacchetto M, Merico A, Piccione F (2011) Event-related brain potential modulation in patients with severe brain damage. Clin Neurophysiol 122:719–724
Davey MP, Victor JD, Schiff ND (2000) Power spectra and coherence in the EEG of a vegetative patient with severe asymmetric brain damage. Clin Neurophysiol 111:1949–1954
Fingelkurts AA, Fingelkurts AA, Bagnato S, Boccagni C, Galardi G (2012) DMN operational synchrony relates to self-consciousness: evidence from patients in vegetative and minimally conscious states. Open Neuroimaging J 6:55–68
Fischer C, Luauté J, Némoz C, Morlet D, Kirkorian G, Mauguière F (2006) Improved prediction of awakening or non awakening from severe anoxic coma using tree-based classification analysis. Crit Care Med 34:1520–1524
Fries P (2009) Neuronal gamma-band synchronization as a fundamental process in cortical computation. Annu Rev Neurosci 32:209–224
Fries P, Reynolds JH, Rorie AE, Desimone R (2001) Modulation of oscillatory neuronal synchronization by selective visual attention. Science 291:1560–1563
Giacino JT, Kalmar K, Whyte J (2004) The JFK coma recovery scale—revised: measurement characteristics and diagnostic utility. Arch Phys Med Rehab 85:2020–2029
Gruber T, Muller MM, Keil A (2002) Modulation of induced gamma band responses in a perceptual learning task in the human EEG. J Cognit Neurosci 14:732–744
Guérit JM (2007) Electroencephalography: the worst traditionally recommended tool for brain death confirmation. Intensive Care Med 33:9–10
Hallett M (1999) EEG coherence: an introduction. J Clin Neurophysiol 16:499–500
Harrison AH, Connolly JF (2013) Finding a way in: a review and practical evaluation of fMRI and EEG for detection and assessment in disorders of consciousness. Neurosci Biobehav Rev 37:1403–1419
Jiang W, Chapman CE, Lamarre Y (1991) Modulation of the cutaneous responsiveness of neurones in the primary somatosensory cortex during conditioned arm movements in the monkey. Exp Brain Res 84:342–354
Kochhar A, Hildebrand MS, Smith RJ (2007) Clinical aspects of hereditary hearing loss. Genet Med 9:393–408
Kotchoubey B (2005) Event-related potential measures of consciousness: two equations with three unknowns. Prog Brain Res 150:427–444
Kwan CL, Crawley AP, Mikulis DJ, Davis KD (2000) An fMRI study of the anterior cingulate cortex and surrounding medial wall activations evoked by noxious cutaneous heat and cold stimuli. Pain 85:359–374
Laureys S, Faymonville ME, Degueldre C et al (2000) Auditory processing in the vegetative state. Brain 123:1589–1601
Laureys S, Faymonville ME, Peigneux P, Damas P, Lambermont B, Del Fiore G et al (2002) Cortical processing of noxious somatosensory stimuli in the persistent vegetative state. Neuroimage 17:732–741
Laureys S, Boly M, Maquet P (2006) Tracking the recovery of consciousness from coma. J Clin Invest 116:1823–1825
Laureys S, Celesia GG, Cohadon F, Lavrijsen J, León-Carrión J, Sannita WG, Sazbon L, Schmutzhard E, von Wild KR, Zeman A, Dolce G (2010) European task force on disorders of consciousness. Unresponsive wakefulness syndrome: a new name for the vegetative state or apallic syndrome. BMC Med 8:68
Lehembre R, Bruno MA, Vanhaudenhuyse A, Chatelle C, Cologan V, Leclercq Y, Soddu A, Macq B, Laureys S, Noirhomme Q (2012a) Resting state EEG study of comatose patients: a connectivity and frequency analysis to find differences between vegetative and minimally conscious states. Funct Neurol 27:41–47
Lehembre R, Gosseries O, Lugo Z, Jedidi Z, Chatelle C, Sadzot B, Laureys S, Noirhomme Q (2012b) Electrophysiological investigations of brain function in coma, vegetative and minimally conscious patients. Arch Ital Biol 150:122–139
Leon-Carrion J, Martin-Rodriguez JF, Damas-Lopez J, Barroso y Martin JM, Dominguez-Morales MR (2008) Brain function in the minimally conscious state: a quantitative neurophysiological study. Clin Neurophysiol 119:1506–1514
Lombardi F, Taricco M, De Tanti A, Telaro E, Liberati A. (2002) Sensory stimulation for brain injured individuals in coma or vegetative state. Cochrane Database Syst Rev (2):CD001427
Manganotti P, Formaggio E, Storti SF, Fiaschi A, Battistin L, Tonin P, Piccione F, Cavinato M (2013) Effect of high-frequency repetitive transcranial magnetic stimulation on brain excitability in severely brain-injured patients in minimally conscious or vegetative state. Brain Stimul 6:913–921
Mesulam MM (1990) Large-scale neurocognitive networks and distributed processing for attention, language, and memory. Ann Neurol 28:597–613
Mueller MM, Gruber T, Keil A (2000) Modulation of induced gamma band activity in the human EEG by attention and visual information processing. Int J Psychophysiol 38:283–299
Oh H, Seo W (2003) Sensory stimulation programme to improve recovery incomatose patients. J Clin Nurs 12:394–404
Owen AM, Coleman MR, Boly M, Davis MH, Laureys S, Pickard JD (2006) Detecting awareness in the vegetative state. Science 313(5792):1402
Paus T, Castro-Alamancos MA, Petrides M (2001) Cortico-cortical connectivity of the human mid-dorsolateral frontal cortex and its modulation by repetitive transcranial magnetic stimulation. Eur J Neurosci 14:1405–1411
Pereda E, Quiroga RQ, Bhattacharya J (2005) Nonlinear multivariate analysis of neurophysiological signals. Progr. Neurobiol 77:1–37
Piccione F, Cavinato M, Manganotti P, Formaggio E, Storti S, Battistin L, Cagnin A, Tonin P, Dam M (2010) Behavioural and neurophysiological effects of repetitive transcranial magnetic stimulation (rTMS) on minimally conscious state: a case study. Neurorehabil Neural Repair 25:98–102
Posner MI, Rothbart MK, Sheese BE, Tang Y (2007) The anterior cingulate gyrus and the mechanism of self-regulation. Cogn Affect Behav Neurosci 7(4):391–395
Rappaport M, Hall KM, Hopkins K, Belleza T, Cope DN (1982) Disability rating scale for severe head trauma: coma to community. Arch Phys Med Rehabil 63:118–123
Rodriguez R, Kallenbach U, Singer W, Munk MHJ (2004) Short- and long-term effects of cholinergic modulation on gamma oscillations and response synchronization in the visual cortex. J Neurosci 24:10369–10378
Sadaghiani S, Hesselmann G, Kleinschmidt A (2009) Distributed and antagonistic contributions of ongoing activity fluctuations to auditory stimulus detection. J Neurosci 29:13410–13417
Schiff ND, Plum F (2000) The role of arousal and "gating" systems in the neurology of impaired consciousness. J Clin Neurophysiol 17:438–452
Schiff ND, Ribary U, Moreno DR et al (2002) Residual cerebral activity and behavioural fragments can remain in the persistently vegetative brain. Brain 125:1210–1234
Sherman SM (2005) Thalamic relays and cortical functioning. Prog Brain Res 149:107–126
Shinotoh H, Fukushi K, Nagatsuka S, Tanaka N, Aotsuka A, Ota T, Namba H, Tanada S, Irie T (2003) The amygdala and Alzheimer’s disease: positron emission tomography study of the cholinergic system. Ann N Y Acad Sci 985:411–419
Srinivasan R, Winter WR, Ding J, Nunez PL (2007) EEG and MEG coherence: measures of functional connectivity at distinct spatial scales of neocortical dynamics. J Neurosci Methods 15(166):41–52
The Multi-Society Task Force on PVS (1994) Medical aspects of the persistent vegetative state–second of two parts. N Engl J Med 333:1572–1579
Van der Hiele K, Vein AA, Reijntjes RH et al (2007) EEG correlates in the spectrum of cognitive decline. Clin Neurophysiol 118:1931–1939
Wallace BE, Wagner AK, Wagner EP, McDeavitt JT (2001) A history and review of quantitative electroencephalography in traumatic brain injury. J Head Trauma Rehabil 16(2):165–190
Waterhouse BD, Border B, Wahl L, Mihailoff GA (1993) Topographic organization of rat locus coeruleus and dorsal raphe nuclei: distribution of cells projecting to visual system structures. J Comp Neurol 336:345–361
Young GB (2000) The EEG in coma. J Clin Neurophysiol 17:473–485
Zandbergen EG, de Haan RJ, Stoutenbeek CP, Koelman JH, Hijdra A (1998) Systematic review of early prediction of poor outcome in anoxic-ischaemic coma. Lancet 352:1808–1812
Zeman A (2001) Conscious Brain 124:1263–1289
Zeman A (2009) The problem of unreportable awareness. Prog Brain Res 177:1–9
Acknowledgment
This work was partially supported by Myosens (FP7-PEOPLE-2011-IAPP-286208) project and by the Region of Veneto, current research funding (3984/08). We would like to thank Felice Silverio, and Paolo Fusaro for the skilled assistance in conducting the experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cavinato, M., Genna, C., Manganotti, P. et al. Coherence and Consciousness: Study of Fronto-Parietal Gamma Synchrony in Patients with Disorders of Consciousness. Brain Topogr 28, 570–579 (2015). https://doi.org/10.1007/s10548-014-0383-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-014-0383-5