Abstract
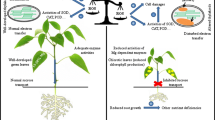
The effects of nitric oxide (NO) and/or iron (Fe) supplied to Fe deficient plants have been investigated in peanut (Arachis hypogaea L.) grown in Hoagland nutrient solution with or without Fe. Two weeks after Fe deprivation, recovery was induced by addition of 250 μM sodium nitroprusside (SNP, a NO donor) and/or 50 μM Fe (Fe-EDTA) to the Fe deprived (-Fe) nutrient solution. Activities of antioxidant enzymes, leaf chlorophyll (Chl), and active Fe content decreased, whereas activities of H+-ATPase, ferric-chelate reductase (FCR), nitrate reductase, and nitric oxide synthase and NO production increased in Fe deficient plants, consequently an Fe chlorosis symptom appeared obviously. In contrast, these symptoms disappeared gradually after two weeks with NO and/or Fe supply, which caused an increases in leaf Chl and active Fe content, especially following by co-treatment with NO and Fe to values found in Fe sufficient plants. Increased activities of antioxidant enzymes (superoxide dismutase, peroxidase, and catalase) and decreased accumulation of reactive oxygen species (H2O2, O •−2 ) and malondialdehyde enhanced the ability of resistance to oxidative stress. Supplied NO alone had the obvious effect on increased NO production and on activity of H+-ATPase and FCR, whereas root length and root/shoot ratio were most effectively increased by Fe supplied alone. Co-treatment with NO and Fe did the best effects on recovery peanut chlorosis symptoms by significantly increased Chl and available Fe content and adjusted distribution of Fe and other mineral elements (Ca, Mg, and Zn) in both leaves and roots.
Similar content being viewed by others
Abbreviations
- Car:
-
carotenoid
- CAT:
-
catalase
- Chl:
-
chlorophyll
- FCR:
-
ferric-chelate reductase
- LSD:
-
least significant diffrence
- MDA:
-
malondialdehyde
- NR:
-
nitrate reductase
- NOS:
-
nitric oxide synthase
- O •−2 :
-
superoxide anion
- PM:
-
plasma membrane
- POD:
-
peroxidase
- ROS:
-
reactive oxygen species
- SNP:
-
sodium nitroprusside
- SOD:
-
superoxide dismutase
References
Airaki, M., Leterrier, M., Mateos, R.M., Valderrama, R., Chaki, M., Barroso, J.B., Del Río, L.A., Palma, J.M., Corpas, F.J.: Metabolism of reactive oxygen species and reactive nitrogen species in pepper (1 L.) plants under low temperature stress. - Plant Cell Environ. 35: 281–295, 2012.
Arnaud, N., Murgia, I., Boucherez, J., Briat, J.F., Cellier, F., Gaymard, F.: An iron induced nitric oxide burst precedes ubiquitin-dependent protein degradation for Arabidopsis AtFer1 ferritin gene expression. - J. biol. Chem. 281: 23579–88, 2006.
Asai, S., Ohta, K., Yoshioka, H.: MAPK signaling regulates nitric oxide and NADPH oxidase-dependent oxidative bursts in Nicotiana benthamiana. - Plant Cell. 20: 1390–1406, 2008.
Attree, R., Dua, B., Xu, B.J.: Distribution of phenolic compounds in seed coat and cotyledon, and their contribution to antioxidant capacities of red and black seed coat peanuts (1 L.). - Ind. Crops Prod. 67: 448–456, 2015.
Bacaicoa, E., Mora, V., Zamarreño, Á. M., Fuentes, M., Casanova, E., García-Mina, J.M.: Auxin: a major player in the shoot-to-root regulation of root Fe-stress physiological responses to Fe deficiency in cucumber plants. - Plant Physiol. Biochem. 49: 545–556, 2011.
Besson-Bard, A., Pugin, A., Wendehenne, D.: New insights into nitric oxide signaling in plants. - Annu. Rev. Plant Biol. 59: 21–39, 2008.
Bradford, M.M.: A rapid and sensitive method for the quantitation of microgram quantitites of protein utilizing the principle of protein-dye binding. - Anal. Biochem. 72: 248–254, 1976.
Briat, J.F., Dubos, C., Gaymard, F.: Iron nutrition, biomass production, and plant product quality. - Trends Plant Sci. 20: 33–40, 2014.
Briat, J.F., Duc, C., Ravet, K., Gaymard, F.: Ferritin and iron storage in plants. - Biochim. biophys. Acta. 1800: 806–814, 2010.
Briskin, D.P., Leonard, R.T., Hodges, T.K.: Isolation of the plasma membrane: markers and general principles. - Method. Enzymol. 148: 542–558 1987.
Chen, W.W., Yang, J.L., Qin, C., Jin, C.W., Mo, J.H., Ye, T., Zheng, S.J.: Nitric oxide acts downstream of auxin to trigger root ferric-chelate reductase activity in response to iron deficiency in Arabidopsis. - Plant Physiol. 154: 810–819, 2010.
Colangelo, E.P., Guerinot, M.L.: The essential basic helix-loophelix protein FIT1 is required for the iron deficiency response. - Plant Cell. 16: 3400–3412, 2004.
Connolly, E.L., Campbell, N.H., Grotz, N., Prichard, C.L., Guerinot, M.L.: Overexpression of the FRO2 ferric chelate reductase confers tolerance to growth on low iron and uncovers posttranscriptional control. - Plant Physiol. 133: 1102–1110, 2003.
Conte, S.S., Walker, E.L.: Transporters contribute to iron trafficking in plants. - Mol. Plant. 4: 464–76, 2011.
Ding, F., Wang, X.F., Shi, Q.H., Wang, L., Yang, F.J., Gao, Q.H.: Exogenous nitric oxide alleviated the inhibition of photosynthesis and antioxidant enzyme activities in irondeficient Chinese cabbage (Brassica chinensis L.) - Agr. Sci. China 7: 168–179, 2008.
Du, S.T., Liu, Y., Zhang, P., Liu, H.J., Zhang, X.Q., Zhang, R.R.: Atmospheric application of trace amounts of nitric oxide enhances tolerance to salt stress and improves nutritional quality in spinach (Spinacia oleracea L.). - Food Chem. 173: 905–911, 2015.
Fernández-Marcos, M., Sanz, L., Lewis, D.R., Lorenzo, O.: Nitric oxide causes root apical meristem defects and growth inhibition while reducing PIN-FORMED 1(PIN1)- dependent acropetal auxin transport. - Proc. nat. Acad. Sci. USA 108: 18506–18511, 2011.
Gao, L., Shi, Y.X.: Genetic differences in resistance to iron deficiency chlorosis in peanut. - J. Plant Nutr. 30: 37–52, 2007.
García, M.J., Suárez, V., Romera, F.J., Alcántara, E., Pérez-Vicente, R.: A new model involving ethylene, nitric oxide and Fe to explain the regulation of Fe-acquisition genes in Strategy I plants. - Plant Physiol. Biochem. 49: 537–544, 2011.
García-Mina, J.M., Bacaicoa, E., Fuentes, M., Casanova, E.: Fine regulation of leaf iron use efficiency and iron root uptake under limited iron bioavailability. - Plant Sci. 198: 39–45, 2013.
Graziano, M., Beligni, M.V., Lamattina, L.: Nitric oxide improves internal iron availability in plants. - Plant Physiol. 130: 1852–1859, 2002.
Graziano, M., Lamattina, L.: Nitric oxide and iron in plants: an emerging and converging story. - Trends Plant Sci. 10: 4–8, 2005.
Graziano, M., Lamattina, L.: Nitric oxide accumulation is required for molecular and physiological responses to iron deficiency in tomato roots. - Plant J. 52: 949–960, 2007.
Guo, F.Q., Okamoto, M., Crawford, N.M.: Identification of a plant nitric oxide synthase gene involved in hormonal signaling. - Science 302: 100–103, 2003.
Halliwell, B.: Reactive species and antioxidants redox biology is a fundamental theme of aerobic life. - Plant Physiol. 141: 312–322, 2006.
Hakan, C. A., Vahap, K.: Some parameters in relation to iron nutrition status of peach orchads. - J. Biol. environ. Sci. 1: 111–115, 2007.
Heath, R.L., Packer, L.: Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. - Arch. Biochem. Biophys. 125: 189–198, 1968.
Hell, R., Stephan, U.W.: Iron uptake, trafficking and homeostasis in plants. - Planta 216: 541–551, 2003.
Hindt, M.N., Guerinot, M.L.: Getting a sense for signals: regulation of the plant iron deficiency response. - Biochim. biophys. Acta 1823: 1521–1530, 2012.
Hoagland, D.R., Arnon, D.I.: The water-culture method for growing plants without soil. - Calif. Agr. Exp. Sta. Circular 347: 1–32, 1950.
Ishimaru, Y., Kim, S.A., Tsukamoto, T., Oki, H., Kobayashi, T., Watanabe, S., Matsuhashi, S., Takahashi, M., Nakanishi, H., Mori, S., Nishizawa, N.K.: Mutational reconstructed ferric chelate reductase confers enhanced tolerance in rice to iron deficiency in calcareous soil. - Proc. nat. Acad. Sci. USA. 104: 7373–7378, 2007.
Jasid, S., Simontacchi, M., Puntarulo, S.: Exposure to nitric oxide protects against oxidative damage but increases the labile iron pool in sorghum embryonic axes. - J. exp. Bot. 59: 3953–3962, 2008.
Jaworski, E.G.: Nitrate reductase assay in intact plant tissues. - Biochem. biophys. Res. Commun. 43: 1274–1279, 1971.
Jelali, N., Donnini, S., Dell’Orto, M., Abdelly, C., Gharsalli, M., Zocchi, G.: Root antioxidant responses of two Pisum sativum cultivars to direct and induced Fe deficiency. - Plant Biol. 16: 607–614, 2014.
Kabir, A.H., Rahman, M.M., Haider, S.A., Paul, N.K.: Mechanisms associated with differential tolerance to Fe deficiency in okra (Abelmoschus esculentus Moench). - Environ. exp. Bot. 112: 16–26, 2015.
Knudson, L.L., Tibbitts, T.W., Edwards, G.E.: Measurement of ozone injury by determination of leaf chlorophyll concentration. - Plant Physiol. 60: 606–608, 1977.
Kobayashi, T., Nishizawa, N.K.: Iron uptake, translocation and regulation in higher plants. - Annu Rev. Plant Biol. 63: 131–152, 2012.
Kong, J., Dong, Y.J., Song, Y.L., Bai, X.Y., Tian, X.Y., Xu, L.L., Liu, S., He, Z.L.: Role of exogenous nitric oxide in alleviating iron deficiency stress of peanut seedlings (Arachis hypogaea L.). - J. Plant Growth Regul. 35: 31–43, 2016.
Kong, J., Dong, Y.J., Xu, L.L., Liu, S., Bai, X.Y.: Effects of foliar application of salicylic acid and nitric oxide in alleviating iron deficiency induced chlorosis of Arachis hypogaea L. - Bot. Stud. 55: 1–9, 2014.
Kumar, P., Tewari, R.K., Sharma, P.N.: Sodium nitroprussidemediated alleviation of iron deficiency and modulation of antioxidant responses in maize plants. - AoB Plants 2010, plq002. doi: 10.1093/aobpla/plq002
Larbi, A., Abadía, A., Abadía, J., Morales, F.: Down coregulation of light absorption, photochemistry, and carboxylation in Fe-deficient plants growing in different environments. - Photosynth. Res. 89: 113–126, 2006.
Larbi, A., Abadía, A., Morales, F., Abadía, J.: Fe resupply to Fe-deficient sugar beet plants leads to rapid changes in the violaxanthin cycle and other photosynthetic characteristics without de novo chlorophyll synthesis. - Photosynth. Res. 79: 59–69, 2004.
Larbi, A., Morales, F., Abadía, A., Abadía, J.: Changes in iron and organic acid concentrations in xylem sap and apoplastic fluid of iron-deficient Beta vulgaris plants in response to iron resupply. - J. Plant Physiol. 167: 255–260, 2010.
Lee, S., Chiecko, J.C., Kim, S.A., Walker, E.L., Lee, Y., Guerinot, M.L., An, G.: Disruption of OsYSL15 leads to iron inefficiency in rice plants. - Plant Physiol. 150: 786–800, 2009.
Li, H.: Principles and Techniques of Plant Physiological Experiment. - Higher Education Press, Beijing 2000.
Liu, B., Rennenberg, H., Kreuzwieser, J.: Hypoxia induces stem and leaf nitric oxide (NO) emission from poplar seedlings. - Planta 241: 579–589, 2015.
Liu, S.L., Yang, R.T., Pan, Y.Z., Ma, M.D., Pan, J., Zhao, Y., Cheng, Q.S., Wu, M.X., Wang, M.H., Zhang, L.: Nitric oxide contributes to minerals absorption, proton pumps and hormone equilibrium under cadmium excess in Trifolium repens L. plants. - Ecotox. Environ. Safety 119: 35–46, 2015.
López-Millán, A.F., Morales, F., Abadía, A., Abadía, J.: Changes induced by Fe deficiency and Fe resupply in the organic acid metabolism of sugar beet (Beta vulgaris L.) leaves. - Physiol Plant. 112: 3–38, 2001.
Manai, J., Gouia, H., Corpas, F.J.: Redox and nitric oxide homeostasis are affected in tomato (Solanum lycopersicum) roots under salinity-induced oxidative stress. - J Plant Physiol. 171: 1028–1035, 2014.
Mahmoudi, H., Labidi, N., Ksouri, R., Gharsalli, M., Abdelly, C.: Differential tolerance to iron deficiency of chickpea varieties and Fe resupply effects. - Compt. Rend. Biol. 330: 237–246, 2007.
Martin, M., Colman, M.J.R., Gomez-Casati, D.F., Lamattina, L., Zabaleta, E.J.: Nitric oxide accumulation is required to protect against iron-mediated oxidative stress in frataxindeficient Arabidopsis plants. - FEBS Lett. 583: 542–548, 2009.
Mengel, K., Kirkby, E.A.: Principles of Plant Nutrition. - Kluwer Academic Publishers, Dordrecht 2001.
Morrissey, J., Guerinot, M.L.: Iron uptake and transport in plants: the good, the bad, and the ionome. - Chem. Rev. 109: 4553–67, 2009.
Nickel, K.S., Cunningham, B.A.: Improved peroxidase assay method using leuco 2,3,6-trichloro indophenol and application to comparative measurements of peroxidase catalysis. - Anal. Biochem. 27: 292–299, 1969.
Ohinishi, T., Gall, R.S., Mayer, M.L.: An improved assay of inorganic phosphate in the presence of extralabile phosphate compounds: application to the ATPase assay in the presence of phosphocreatine. - Anal. Biochem. 69: 261–267, 1975.
Osorio, J., Osorio, M.L., Correia, P.J., De Varennes, A., Pestana, M.: Chlorophyll fluorescence imaging as a tool to understand the impact of iron deficiency and resupply on photosynthetic performance of strawberry plants. - Sci. Hort. 165: 148–155, 2014.
Palmgren, M.G.: Plant plasma membrane H+-ATPase: powerhouses for nutrient uptake. - Annu. Rev. Plant Biol. 52: 817–845, 2001.
Patra, H.L., Kar, M., Mishre, D.: Catalase activity in leaves and cotyledons during plant development and senescence. - Biochem. Pharmacol. 172: 385–390, 1978.
Pätsìkkä, E., Kairavuo, M., Šeršen, F., Aro, E.M., Tyystjärvi, E.: Excess copper predisposes photosystem II to photoinhibition in vivo by outcompeting iron and causing decrease in leaf chlorophyll. - Plant Physiol. 129: 1359–1367, 2002.
Pereira, M.P., Santos, C., Gomes, A., Vasconcelos, M.W.: Cultivar variability of iron uptake mechanisms in rice (Oryza sativa L.) - Plant Physiol. Biochem. 85: 21–30, 2014.
Pestana, M., Correia, P.J., Saavedra, T., Gama, F., Abadía, A., De Varennes, A.: Development and recovery of iron deficiency by iron resupply to roots or leaves of strawberry plants. - Plant Physiol. Biochem. 53: 1–5, 2012.
Pii, Y., Penn, A., Terzano, R., Crecchio, C., Mimmo, T., Cesco, S.: Plant-microorganism-soil interactions influence the Fe availability in the rhizosphere of cucumber plants. - Plant Physiol. Biochem. 87: 45–52, 2015.
Ramirez, L., Simontacchi, M., Murgia, I., Zabaleta, E., Lamattina, L.: Nitric oxide, nitrosyl iron complexes, ferritin and frataxin: a well equipped team to preserve plant iron homeostasis. - Plant Sci. 181: 582–592, 2011.
Ramirez, L., Zabaleta, E.J., Lamattina, L.: Nitric oxide and frataxin: Two players contributing to maintain cellular iron homeostasis. - Ann. Bot. 105: 801–810, 2010.
Ranieri, A., Castagna, A., Baldan, B., Soldatini, G.F.: Iron deficiency differently affects peroxidase isoforms in sunflower. - J. exp. Bot. 52: 25–35, 2001.
Rockel, P., Strube, F., Rockel, A., Wildt, J., Kaiser, W.M.: Regulation of nitric oxide (NO) production by plant nitrate reductase in vivo and in vitro. - J. exp. Bot. 53: 103–110, 2002.
Schmidt, W.: Iron solutions: acquisition strategies and signaling pathways in plants. - Trends Plant Sci. 8: 188–193, 2003.
Shi, G.R., Su, G.Q., Lu, Z.W., Liu, C.F., Wang, X.M.: Relationship between biomass, seed components and seed Cd concentration in various peanut (Arachis hypogaea L.) cultivars grown on Cd-contaminated soils. - Ecotox. Environ. Safety 110: 174–181, 2014.
Shi, Q. H., Ding, F., Wang, X. F., Wei, M.: Exogenous nitric oxide protect cucumber roots against oxidative stress induced by salt stress. - Plant Physiol. Biochem. 45: 542–550, 2007.
Shi, Q.H., Zhu, Z.J.: Effects of exogenous salicylic acid on manganese toxicity, element contents and antioxidative system in cucumber. - Environ. exp. Bot. 63: 317–326, 2008.
Simontacchi, M., Buet, A., Lamattina, L., Puntarulo, S.: Exposure to nitric oxide increases the nitrosyl-iron complexes content in Sorghum embryonic axes. - Plant Sci. 183: 159–166, 2012.
Sokolovski, S.G., Blatt, M.R.: Nitric oxide and plant ion channel control. - In: Lamattina, L., Polacco, J.C. (ed.): Nitric Oxide in Plant Growth, Development and Stress Physiology. Pp. 153–171. Springer, Berlin - Heidelberg 2007.
Stewart, R.C., Bewley, J.D.: Lipid peroxidation associated with accelerated aging of soybean axes. - Plant Physiol. 65: 245–248, 1980.
Sun, B., Jing, Y., Chen, K., Song, L., Chen, F., Zhang, L.: Protective effect of nitric oxide on iron deficiency- induced oxidative stress in maize (Zea mays). - J. Plant Physiol. 164: 536–543, 2007.
Su, Y., Liu, J.L., Lu, Z.W., Wang, X.M., Zhang, Z., Shi, G.G.: Effects of iron deficiency on subcellular distribution and chemical forms of cadmium in peanut roots in relation to its translocation. - Environ. exp. Bot. 97: 40–48, 2014.
Takker, P.N., Kaur. N.P.: HCl method for Fe2+ estimation to resolve iron chlorosis in plants. - J. Plant Nutr. 7: 81–90, 1984.
Tomasi, N., Rizzardo, C., Monte, R., Gottardi, S., Jelali, N., Terzano, R., Vekemans, B., De Nobili, M., Varanini, Z., Pinton, R., Cesco, S.: Micro-analytical, physiological and molecular aspects of Fe acquisition in leaves of Fe-deficient tomato plants resupplied with natural Fe-complexes in nutrient solution. - Plant Soil 325: 25–38, 2009.
Ueno, T., Yoshimura, T.: The physiological activity and in vivo distribution of dinitrosyl dithiolate iron complex. - Jap. J. Pharmacol 82: 95–101, 2000.
Velikova, V., Yordanov, I., Edreva, A.: Oxidative stress and some antioxidant systems in acid rain-treated bean plants. - Plant Sci. 151: 59–66, 2000.
Vert, G.A., Grotz, N., Dédaldéchamp, F., Gaymard, F., Guerinot, M.L., Briat, J.F., Curie, C.: IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. - Plant Cell 14: 1223–1233, 2002.
Wendehenne, D., Pugin, A., Klessig, D.F., Durner, J.: Nitric oxide: comparative synthesis and signaling in animal and plant cells. - Trends Plant Sci. 6: 177–83, 2001.
Xiong, H.C., Guo, X.T., Kobayashi, T., Kakei, Y., Nakanishi, H., Nozoye, T., Zhang, L.X., Shen, H.Y., Qiu, W., Nishizawa, N.K., Zuo, Y.M.: Expression of peanut iron regulated transporter 1 in tobacco and rice plants confers improved iron nutrition. - Plant Physiol. Biochem. 80: 83–89, 2014.
Xiong, Y., Yuan, L.W., Deng, H.W., Li, Y.J., Chen, B.M.: Elevated serum endogenous inhibitor of nitric oxide synthase and endothelial dysfunction in aged rats. - Clin. exp. Pharmacol. 28: 842–847, 2001.
Xu, J., Yin, H.X., Liu, X.J., Yuan, T., Mi, Q., Yang, L.L., Xie, Z.X., Wang, W.Y.: Nitric oxide alleviates Fe deficiencyinduced stress in Solanum nigrum. - Biol. Plant. 53: 784–788, 2009.
Xu, Y.F., Sun, X.L., Jin, J.W., Zhou. H.: Protective effect of nitric oxide on light-induced oxidative damage in leaves of tall fescue. - J. Plant Physiol. 167: 512–518, 2010.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgments: The authors thank Dr. Pingping Yang (the College of Animal Science and Technology, the Shandong Agricultural University, China) for supplying instruments and patient guidance. This research was financially supported by the Taishan Scholars at Seed Industry Talent Project, the Shandong Province Seed Industry Project, and the Shandong Provincial Natural Science Foundation of China (ZR2013CM003).
Rights and permissions
About this article
Cite this article
Song, Y.L., Dong, Y.J., Tian, X.Y. et al. Effects of nitric oxide and Fe supply on recovery of Fe deficiency induced chlorosis in peanut plants. Biol Plant 61, 155–168 (2017). https://doi.org/10.1007/s10535-016-0642-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-016-0642-2