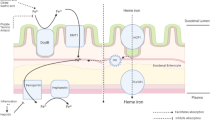
Abstract
Objective Evaluate the safety and efficacy of bovine lactoferrin (bLf) versus the ferrous sulphate standard intervention in curing iron deficiency (ID) and ID anaemia (IDA) in pregnant women affected by hereditary thrombophilia (HT). Design Interventional study. Setting Secondary-level hospital for complicated pregnancies in Rome, Italy. Population 295 HT pregnant women (≥18 years) suffering from ID/IDA. Methods Women were enrolled in Arm A or B in accordance with their personal choice. In Arm A, 156 women received oral administration of 100 mg of bLf twice a day; in Arm B, 139 women received 520 mg of ferrous sulphate once a day. Therapies lasted until delivery. Main outcome measures Red blood cells, haemoglobin, total serum iron, serum ferritin (haematological parameters) were assayed before and every 30 days during therapy until delivery. Serum IL-6, key factor in inflammatory and iron homeostasis disorders, was detected at enrolment and after therapy at delivery. Possible maternal, foetal, and neonatal adverse effects were assessed. Results Haematological parameters were significantly higher in Arm A than in Arm B pregnant women (P ≤ 0.0001). Serum IL-6 significantly decreased in bLf-treated women and increased in ferrous sulphate-treated women. BLf did not exert any adverse effect. Adverse effects in 16.5 % of ferrous sulphate-treated women were recorded. Arm A women experienced no miscarriage compared to five miscarriages in Arm B women. Conclusions Differently from ferrous sulphate, bLf is safe and effective in curing ID/IDA associated with a consistent decrease of serum IL-6. The absence of miscarriage among bLf-treated women provided an unexpected benefit. Trial registration: ClinicalTrials.gov Identifier NCT01221844.



Similar content being viewed by others
References
Abboud S, Haile DJ (2000) A novel mammalian iron-regulated protein involved in intracellular iron metabolism. J Biol Chem 275:19906–19912. doi:10.1074/jbc.M000713200
Apgar V (1953) A proposal for a new method of evaluation of the newborn infant. Curr Res Anesth Analg 32:260–267
Baker EN, Baker HM (2005) Molecular structure, binding properties and dynamics of lactoferrin. Cell Mol Life Sci 62:2531–2539. doi:10.1007/s00018-005-5368-9
Baker HM, Baker EN (2012) A structural perspective on lactoferrin function. Biochem Cell Biol 90:320–328. doi:10.1139/o11-071
Bastin J, Drakesmith H, Rees M, Sargent I, Townsend A (2006) Localisation of proteins of iron metabolism in the human placenta and liver. Br J Haematol 134:532–543. doi:10.1111/j.1365-2141.2006.06216.x
Bothwell TH (2000) Iron requirements in pregnancy and strategies to meet them. Am J Clin Nutr 72:257–264
Bradley J, Leibold EA, Harris ZL, Wobken JD, Clarke S, Zumbrennen KB, Eisenstein RS, Georgieff MK (2004) Influence of gestational age and fetal iron status on IRP activity and iron transporter protein expression in third-trimester human placenta. Am J Physiol Regul Integr Comp Physiol 287:R01–R894. doi:10.1152/ajpregu.00525.2003
Collard KJ (2009) Iron homeostasis in the neonate. Pediatrics 123:1208–1221. doi:10.1542/peds.2008-1047
Donovan A, Lima CA, Pinkus JL, Pinkus GS, Zon LI, Robine S, Andrews NC (2005) The iron exporter ferroportin Slc40a1 is essential for iron homeostasis. Cell Metab 1:191–200. doi:10.1016/j.cmet.2005.01.003
Fox EA, Kahn SR (2005) The relationship between inflammation and venous thrombosis. A systematic review of clinical studies. Thromb Haemost 94:362–365. doi:10.1160/TH05-04-0266
Ganz T (2005) Cellular iron: ferroportin is the only way out. Cell Metab 1:155–157. doi:10.1016/j.cmet.2005.02.006
Ganz T (2013) Systemic iron homeostasis. Physiol Rev 93:1721–1741. doi:10.1152/physrev.00008.2013
Hagberg H, Mallard C, Jacobsson B (2005) Role of cytokines in preterm labour and brain injury. BJOG 112:16–18. doi:10.1111/j.1471-0528.2005.00578.x
Jackson R, Holmes K, Phansalkar A, Rodgers GM (2008) Testing for hereditary thrombophilia: a retrospective analysis of testing referred to a national laboratory. BMC Clin Pathol 8:1–7. doi:10.1186/1472-6890-8-3
Kaandorp S, Di Nisio M, Goddijn M, Middeldorp S (2009) Aspirin or anticoagulants for treating recurrent miscarriage in women without antiphospholipid syndrome. Cochrane Database Syst Rev 21:4734. doi:10.1002/14651858.CD004734.pub3
Kadiiska MB, Burkitt MJ, Xiang QH, Mason RP (1995) Iron supplementation generates hydroxyl radical in vivo. An ESR spin-trapping investigation. J Clin Invest 96:1653–1657. doi:10.1172/JCI118205
Khan S, Dickerman JD (2006) Hereditary thrombophilias. Thromb J 4:15. doi:10.1186/1477-9560-4-15
Lönnerdal B, Bryant A (2006) Absorption of iron from recombinant human lactoferrin in young US women. Am J Clin Nutr 83:305–309
Meier PR, Nickerson HJ, Olson KA, Berg RL, Meyer JA (2003) Prevention of iron deficiency anemia in adolescent and adult pregnancies. Clin Med Res 1:29–36. doi:10.3121/cmr.1.1.29
Nemeth E, Ganz T (2006) Regulation of iron metabolism by hepcidin. Annu Rev Nutr 26:323–342. doi:10.1146/annurev.nutr.26.061505.111303
Nemeth E, Tuttle MS, Powelson J, Vaughn MB, Donovan A, Ward DM, Ganz T, Kaplan J (2004) Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306:2090–2093. doi:10.1126/science.1104742
Nicolas G, Bennoun M, Devaux I, Beaumont C, Grandchamp B, Kahn A, Vaulont S (2001) Lack of hepcidin gene expression and severe tissue iron overload in upstream stimulatory factor 2 (USF2) knockout mice. Proc Natl Acad Sci USA 98:8780–8785. doi:10.1073/pnas.151179498
Nicolas G, Chauvet C, Viatte L, Danan JL, Bigard X, Devaux I, Beaumont C, Kahn A, Vaulont S (2002) The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. J Clin Invest 110:1037–1044. doi:10.1172/JCI15686
Oldenburg B, Van Berge Henegouwen GP, Rennick D, Van Asbeck BS, Koningsberger JC (2000) Iron supplementation affects the production of proinflammatory cytokines in IL-10 deficient mice. Eur J Clin Invest 30:505–510. doi:10.1046/j.1365-2362.2000.00650.x
Paesano R, Torcia F, Berlutti F, Pacifici E, Ebano V, Moscarini M, Valenti P (2006) Oral administration of lactoferrin increases hemoglobin and total serum iron in pregnant women. Biochem Cell Biol 8:377–380
Paesano R, Pietropaoli M, Gessani S, Valenti P (2009) The influence of lactoferrin, orally administered, on sistemi iron homeostasis in pregnant women suffering of iron deficiency and iron deficiency anemia. Biochimie 91:44–51. doi:10.1016/j.biochi.2008.06.004
Paesano R, Berlutti F, Pietropaoli M, Pantanella F, Pacifici E, Goolsbee W, Valenti P (2010a) Lactoferrin efficacy versus ferrous sulfate in curing iron deficiency and iron deficiency anemia in pregnant women. Biometals 23:411–417. doi:10.1007/s10534-010-9335-z
Paesano R, Berlutti F, Pietropaoli M, Goolsbee W, Pacifici E, Valenti P (2010b) Lactoferrin efficacy versus ferrous sulfate in curing iron disorders in pregnant and non pregnant women. Int J Immunopathol Pharmacol 23:577–587
Paesano R, Pietropaoli M, Berlutti F, Valenti P (2012a) Bovine lactoferrin in preventing preterm delivery associated with sterile inflammation. Biochem Cell Biol 90:468–475. doi:10.1139/o11-060
Paesano R, Natalizi T, Berlutti F, Valenti P (2012b) Body iron delocalization: the serious drawback in iron disorders in both developing and developed countries. Pathog Glob Health 106:200–216. doi:10.1179/2047773212Y.0000000043
Park CH, Valore EV, Waring AJ, Ganz T (2001) Hepcidin, a urinary antimicrobial peptide synthesized in the liver. J Biol Chem 276:7806–7810. doi:10.1074/jbc.M008922200
Patnaik MM, Haddad T, Morton CT (2007) Pregnancy and thrombophilia. Expert Rev Cardiovasc Ther 5:753–765. doi:10.1586/14779072.5.4.753
Petrozella LN, Dashe JS, McIntire DD, Leveno KJ (2011) Clinical significance of borderline amniotic fluid index and oligohydramnios in preterm pregnancy. Obstet Gynecol 117:338–342. doi:10.1097/AOG.0b013e3182056766
Qiao B, Sugianto P, Fung E, Del-Castillo-Rueda A, Moran-Jimenez MJ, Ganz T, Nemeth E (2012) Hepcidin-induced endocytosis of ferroportin is dependent on ferroportin ubiquitination. Cell Metab 15:918–924. doi:10.1016/j.cmet.2012.03.018
Reifen R, Matas Z, Zeidel L, Berkovitch Z, Bujanover Y (2000) Iron supplementation may aggravate inflammatory status of colitis in a rat model. Dig Dis Sci 45:394–397
Rosendaal FR (1999) Venous thrombosis: a multicausal disease. Lancet. doi:10.1016/S0140-6736(98)10266-0
Scholl TO (2005) Iron status during pregnancy: setting the stage for mother and infant. Am J Clin Nutr 81:1218S–1222S
Shafir T, Angulo-Barroso R, Jing Y, Angelilli ML, Jacobson SW, Lozoff B (2008) Iron deficiency and infant motor development. Early Hum Dev 84:479–485. doi:10.1016/j.earlhumdev.2007.12.009
Stella CL, Sibai BM (2006) Thrombophilia and adverse maternal perinatal outcome. Clin Obstet Gynecol 49:850–860
Umbreit J (2005) Iron deficiency: a concise review. Am J Hematol 78:225–231. doi:10.1002/ajh.20249
Valenti P, Antonini G (2005) Lactoferrin: an important host defense against microbial and viral attack. Cell Mol Life Sci 62:2576–2587. doi:10.1007/s00018-005-5372-0
Wessling-Resnick M (2010) Iron homeostasis and the inflammatory response. Annu Rev Nutr 30:105–122. doi:10.1146/annurev.nutr.012809.104804
Zhang DL, Senecal T, Ghosh MC, Ollivierre-Wilson H, Tu T, Rouault TA (2011) Hepcidin regulates ferroportin expression and intracellular iron homeostasis of erythroblasts. Blood 118:2868–2877. doi:10.1182/blood-2011-01-330241
Zhao S, Gua Y, Dong Q, Fan R, Wang Y (2008) Altered interleukin-6 receptor, IL-6R and gp130, production and expression and decreased SOCS-3 expression in placentas from women with pre-eclampsia. Placenta 29:1024–1028. doi:10.1016/j.placenta.2008.09.011
Acknowledgments
This project is supported by Microbo srl, Biotechnology Company, Rome-Italy Grant. IL-6 analyses are supported by Sapienza University of Rome, Italy Grant to PV.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paesano, R., Pacifici, E., Benedetti, S. et al. Safety and efficacy of lactoferrin versus ferrous sulphate in curing iron deficiency and iron deficiency anaemia in hereditary thrombophilia pregnant women: an interventional study. Biometals 27, 999–1006 (2014). https://doi.org/10.1007/s10534-014-9723-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-014-9723-x