Abstract
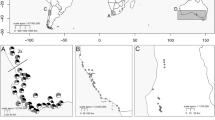
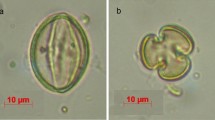
Oxalis pes-caprae, a tristylous flowering plant native to South Africa, is described in the western Mediterranean basin as an asexual—only 5x short-styled morph (5x S-morph) invasive weed losing all mating partners after introduction. The objective of this study was to reassess the patterns of floral morph and cytotype distribution and the sexual reproduction ability in this invaded range. For that, floral morph and cytotype composition were evaluated in 39 populations of O. pes-caprae in a methodical sampling. The reproductive success of natural populations was assessed as fruit and seed production and seed germination for all floral morphs and cytotypes detected. Self- and morph-incompatibility were also studied with controlled hand pollinations. A remarkable diversity in floral morph and cytotype composition was observed. Furthermore, we observed successful sexual reproduction in several localities across the surveyed area. The S-morph is still dominant in this invaded area, and although it was mostly 5x, an additional cytotype (4x) was also recorded. Records of both a mid-styled morph (M-morph) and an area with trimorphic populations of this species are reported here for the first time in the invasive range of the Mediterranean basin. The long-styled morph appears to occur randomly across the surveyed area, while the M-morph is concentrated mainly in Estremadura province (Portugal), where a breakdown in the incompatibility system was observed. These distribution patterns may result from events of sexual reproduction after incompatibility breakdown and/or from multiple introduction events from the native area. The ability to reproduce sexually, undetected so far, may have important impacts in the population dynamics and major consequences for the adaptation and selection potential of O. pes-caprae in this invaded area.



Similar content being viewed by others
References
Arroyo J, Barrett SCH, Hidalgo R, Cole WW (2002) Evolutionary maintenance of stigma-height dimorphism in Narcissus papyraceus (Amaryllidaceae). Am J Bot 89:1242–1249
Ater M (2005) Biologie de la reproduction d’Oxalis pes-caprae au Maroc. In: Congresso 2005 de la Sociedad Española de Malherbologia. Malherbologia Ibérica: Soluciones comunes a problemas comunes, Huelva
Baker HG (1965) Characteristics and modes of origin of weeds. In: Baker HG, Stebbins GL (eds) The genetics of colonizing species. Academic Press, New York, pp 147–168
Barrett SCH (1979) Evolutionary breakdown of tristyly in Eichhornia crassipes (Mart) Solms (Water Hyacinth). Evolution 33:499–510
Barrett SCH (1980) Dimorphic incompatibility and gender in Nymphoides indica (Menyanthaceae). Can J Bot 58:1938–1942
Barrett SCH (1992) Heterostylous genetic polymorphisms: model systems for evolutionary analysis. In: Barrett SCH (ed) Evolution and function of heterostyly. Springer, Berlin, pp 1–29
Barrett SCH (2011) Why reproductive systems matter for the invasion biology of plants. In: Richardson DM (ed) Fifty years of invasion ecology: the legacy of Charles Elton. Blackwell Publishing Ltd, Oxford, pp 195–210
Barrett SCH, Forno IW (1982) Style morph distribution in New World populations of Eichhornia crassipes (Mart) Solms Laubach (Water Hyacinth). Aquat Bot 13:299–306
Barrett SCH, Coberti RI, Eckert CG (2008) Plant reproductive systems and evolution during biological invasion. Mol Ecol 17:373–383
Castro S, Loureiro J, Santos C, Ater M, Ayensa G, Navarro L (2007) Distribution of flower morphs, ploidy level and sexual reproduction of the invasive weed Oxalis pes-caprae in the western area of the Mediterranean region. Ann Bot 99:507–517
Charlesworth D (1979) The evolution and breakdown of tristyly. Evolution 33:486–498
Colautti RI, White NA, Barrett SCH (2010) Variation of self-incompatibility within invasive populations of purple loosestrife (Lythrum salicaria L.) from eastern North America. Int J Plant Sci 171:158–166
Costa J, Ferrero V, Loureiro J, Castro M, Navarro L, Castro S (2013) Sexual reproduction in the invasive pentaploid short-styled Oxalis pes-caprae allows the production of viable offspring. Plant Biology. doi:10.1111/plb.12010
Doležel J, Göhde W (1995) Sex determination in dioecious plants Melandrium album and M. rubrum using high-resolution flow cytometry. Cytometry 19:103–106
Dulberger R (1992) Floral polymorphisms and their functional significance in the heterostylous syndrome. In: Barrett SCH (ed) Evolution and function of heterostyly. Springer, Berlin, pp 41–84
Eckert CG, Barrett SCH (1992) Stochastic loss of style morphs from populations of tristylous Lythrum salicaria and Decodon verticillatus (Lythraceae). Evolution 46:1014–1029
Eckert CG, Barrett SCH (1993) Clonal reproduction and patterns of genotypic diversity in Decodon verticillatus (Lythraceae). Am J Bot 80:1175–1182
Eckert CG, Barrett SCH (1995) Style morph ratios in tristylous Decodon verticillatus (Lythraceae): selection vs historical contingency. Ecology 76:1051–1066
Emshwiller E (2002) Ploidy levels among species in the ‘Oxalis tuberosa Alliance’ as inferred by flow cytometry. Ann Bot 89:741–753
Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP, Firoozabady E (1983) Rapid flow cytometric analysis of the cell-cycle in intact plant tissues. Science 220:1049–1051
Ganders FR (1979) The biology of heterostyly. N Z J Bot 17:607–635
Heuch I (1979) Equilibrium populations of heterostylous plants. Theor Popul Biol 15:43–57
Hodgins KA, Barrett SCH (2008) Asymmetrical mating patterns and the evolution of biased style-morph ratios in a tristylous daffodil. Genet Res 90:3–15
Kohn JR, Barrett SCH (1992) Experimental studies on the functional significance of heterostyly. Evolution 46:43–55
Kolař F, Stech M, Travníček P, Rauchová J, Urfuš T, Vít P, Kubešová M, Suda J (2009) Towards resolving the Knautia arvensis agg. (Dipsacaceae) puzzle: primary and secondary contact zones and ploidy segregation at landscape and microgeographic scales. Ann Bot 103:963–974
Krejčíková J, Sudová R, Oberlander KC, Dreyer LL, Suda J (2013) Cytogeography of Oxalis pes-caprae in its native range: where are the pentaploids? Biol Invasions. doi:10.1007/s10530-012-0370-2
Legendre P, Fortin M-J (1989) Spatial patterns and ecological analysis. Vegetatio 80:107–138
Legendre P, Legendre L (1998) Numerical ecology. 2nd English edition edn. Elsevier Science BV, Amsterdam
Lewis D, Jones DA (1992) The genetics of heterostyly. In: Barrett SCH (ed) Evolution and function of heterostyly. Springer-Verlag, Berlin, pp 129–150
Loureiro J, Rodriguez E, Doležel J, Santos C (2006) Comparison of four nuclear isolation buffers for plant DNA flow cytometry. Ann Bot 86:679–689
Lourteig A (2000) Oxalis L. subgénero Monoxalis (Small) Lourt., Oxalis y Trifidus Lourt. Bradea 7:201–629
Luo SX, Zhang DX, Renner SS (2006) Oxalis debilis in China: distribution of flower morphs, sterile pollen and polyploidy. Ann Bot 98:459–464
Michael P (1964) The indentity and origin of varieties of Oxalis pes-caprae L. naturalized in Australia. Trans R Soc S Aust 88:167–173
Morgan MT, Barrett SCH (1988) Historical factors and anisoplethic population structure in tristylous Pontederia cordata: a reassessment. Evolution 42:496–504
Mulcahy DL (1964) Reproductive biology of Oxalis priceae. Am J Bot 51:1045–1050
Oberlander KC, Emshwiller E, Bellstedt DU, Dreyer LL (2009) A model of bulb evolution in the eudicot genus Oxalis L. Mol Phylogenet Evol 51:54–63
O’Brien SP, Calder DM (1989) The breeding biology of Epacris impressa. Is this species heterostylous? Aust J Bot 37:43–54
O’Neil P (1992) Variation in male and female reproductive success among floral morphs in the tristylous plant Lythrum salicaria (Lythraceae). Am J Bot 79:1024–1030
Ornduff R (1972) Breakdown of trimorphic incompatibility in Oxalis Section Corniculatae. Evolution 26:52–65
Ornduff R (1986) The origin of weediness in Oxalis pes-caprae. Am J Bot 73:779–780
Ornduff R (1987) Reproductive systems and chromosome races of Oxalis pes-caprae L. and their bearing on the genesis of a noxious weed. Ann Mo Bot Gard 74:79–84
Otto F (1992) Preparation and staining of cells for high-resolution DNA analysis. In: Radbruch A (ed) Flow cytometry and cell sorting. Springer, Berlin, pp 101–104
Pütz N (1994) Vegetative spreading of Oxalis pes-caprae (Oxalidaceae). Plant Syst Evol 191:57–67
Pyšek P, Richardson DM (2010) Invasive species, environmental change and management, and health. Annu Rev Environ Resour 35:25–55
Ramsey J, Schemske DW (1998) Pathways, mechanisms, and rates of polyploid formation in flowering plants. Annu Rev Ecol Syst 29:467–501
Rappa F (1911) Osservazioni sull Oxalis cernua Thunb. Bollettino del Orto Botanico Palermo 10:142–183
Ray PM, Chisaki HF (1957) Studies on Amsinckia. Am J Bot 44:529–544
Richardson DM, Pyšek P, Rejmánek M, Barbour MG, Panetta FD, West CJ (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107
Risso-Pascotto C, Pagliarini MS, Do Valle CB, Mendes-Bonato AB (2003) Chromosome number and microsporogenesis in a pentaploid accession of Brachiaria brizantha (Gramineae). Plant Breed 122:136–140
Rosenberg MS, Anderson CD (2011) PASSaGE: pattern analysis, spatial statistics, and geographic exegesis. Version 2. Methods Ecol Evol 2:229–232
Sakai AK, Allendorf FW, Holt JS, Lodge DM, Molofsky J, With KA, Baughman S, Cabin RJ, Cohen JE, Ellstrand NC, McCauley DE, O’Neil P, Parker IM, Thompson JN, Weller SG (2001) The population biology of invasive species. Annu Rev Ecol Syst 32:305–332
Salter TM (1944) The genus Oxalis in South Africa: a taxonomic revision. J S Afr Bot, Supplement 1:1–355
Schönswetter P, Suda J, Popp M, Weiss-Schneeweiss H, Brochmann C (2007) Circumpolar phylogeography of Juncus biglumis (Juncaceae) inferred from AFLP fingerprints, cpDNA sequences, nuclear DNA content and chromosome numbers. Mol Phylogenet Evol 42:92–103
Stockwell CA, Hendry AP, Kinnison MT (2003) Contemporary evolution meets conservation biology. Trends Ecol Evol 18:94–101
Symon D (1961) The species of Oxalis established in South Australia. Trans R Soc S Aust 84:71–77
te Beest M, Le Roux JJ, Richardson DM, Brysting AK, Suda J, Kubesova M, Pyšek P (2012) The more the better? The role of polyploidy in facilitating plant invasions. Ann Bot 109:19–45
Turketti SS (2010) A study of tristyly in South African Oxalis. PhD Thesis, University of Stellenbosch
Vignoli L (1937) Fenomeni reproduttivi di Oxalis cernua Thunb. Lavori Inst Bot Palermo 8:5–30
Vilà M, Bartomeus I, Gimeno I, Traveset A, Moragues E (2006) Demography of the invasive geophyte Oxalis pes-caprae across a Mediterranean Island. Ann Bot 97:1055–1062
Wang Y, Wang QF, Guo YH, Barrett SCH (2005) Reproductive consequences of interactions between clonal growth and sexual reproduction in Nymphoides peltata: a distylous aquatic plant. New Phytol 165:329–335
Young D (1968) Oxalis L. In: Tutin T, Heywood V, Burges N et al (eds) Flora Europaea, vol 2. Cambridge University Press, Cambridge, p 193
Zar JH (1984) Biostatistical analysis, 2nd edn. Prentice-Hall, Inc., New Jersey
Acknowledgments
Authors are thankful to Conceição Santos and Eleazar Rodriguez for their support in the preliminary flow cytometric analysis. This research was supported by FEDER funds through the COMPETE Program and by Portuguese Foundation for Science and Technology (FCT) funds in the ambit of the project PTDC/BIA-BIC/110824/2009, by CRUP Acções Integradas Luso-Espanholas 2010 with the project E10/10, by MCI-Programa de Internacionalización de la I + D (PT2009-0068) and by by the Spanish DGICYT (CGL2009-10466), FEDER funds from the European Union, and the Xunta de Galicia (INCITE 09-3103009PR). FCT also supported the work of Sílvia Castro (FCT/BPD/41200/2007) and Joana Costa (CB/C05/2009/209; PTDC/BIA-BIC/110824/2009). Fundación Ramón Areces supported the work of Victoria Ferrero.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Castro, S., Ferrero, V., Costa, J. et al. Reproductive strategy of the invasive Oxalis pes-caprae: distribution patterns of floral morphs, ploidy levels and sexual reproduction. Biol Invasions 15, 1863–1875 (2013). https://doi.org/10.1007/s10530-013-0414-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-013-0414-2