Abstract
Objective
To compare methods for erythroid differentiation of K562 cells that will be promising in the treatment of beta-thalassemia by inducing γ-globin synthesis.
Results
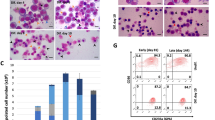
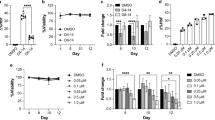
Cells were treated separately with: RPMI 1640 medium without glutamine, RPMI 1640 medium without glutamine supplemented with 1 mM sodium butyrate, RPMI 1640 medium supplemented with 1 mM sodium butyrate, 25 µg cisplatin/ml, 0.1 µg cytosine arabinoside/ml. The highest differentiation (84 %) with minimum toxicity was obtained with cisplatin at 15 µg /ml. Real-time RT-PCR showed that expression of the γ-globin gene was significantly higher in the cells differentiated with cisplatin compared to undifferentiated cells (P < 0.001).
Conclusions
Cisplatin is useful in the experimental therapy of ß-globin gene defects and can be considered for examining the basic mechanism of γ-reactivation.






Similar content being viewed by others
References
Ahmadvand M, Noruzinia M, Fard AD, Zohour MM et al (2014) The role of epigenetics in the induction of fetal hemoglobin: a combination therapy approach. Int J Hematol Oncol Stem Cell Res 8:9–14
Biancha G, Fiorentini P, Gambari R, Nastruzzi C, Barbieri R, Sessarego M, Ravazzolo R, Garre C (1989) Isolation and characterization of a K562 cell line resistant to 1-beta-D-arabinofuranosylcytosine-mediated erythroid induction. Exp Hematol 17:859–864
Bianchi GL, Romani M, Coviello DA, Garre C, Ravazzolo R, Vidali G, Ajmar F (1986) Terminal erythroid differentiation in the K-562 cell line by 1-beta-D-arabinofuranosylcytosine: accompaniment by c-myc messenger RNA decrease. Cancer Res 46:6327–6332
Bianchi N, Ongaro F, Chiarabelli C, Gualandi L, Mischiati C, Bergamini P, Gambari R (2000) Induction of erythroid differentiation of human K562 cells by cisplatin analogs. Biochem Pharmacol 60:31–40
Bianchi N, Chiarabelli C, Borgatti M, Mischiati C, Fibach E, Gambari R (2001) Accumulation of gamma-globin mRNA and induction of erythroid differentiation after treatment of human leukaemic K562 cells with tallimustine. Br J Haematol 113:951–961
Bruchova-Votavova H, Yoon D, Prchal JT (2010) miR-451 enhances erythroid differentiation in K562 cells. Leuk Lymphoma 51:686–693
Chenais B, Andriollo M, Guiraud P, Belhoussine R, Jeannesson P (2000) Oxidative stress involvement in chemically induced differentiation of K562 cells. Free Radic Biol Med 28:18–27
Cioe L, McNab A, Hubbell HR, Meo P, Curtis P, Rovera G (1981) Differential expression of the globin genes in human leukemia K562(S) cells induced to differentiate by hemin or butyric acid. Cancer Res 41:237–243
Erard F, Dean A, Schechter AN (1981) Inhibitors of cell division reversibly modify hemoglobin concentration in human erythroleukemia K562 cells. Blood 58:1236–1239
Gambari R, Raschella G, Biagini R, Tripodi M, Farace MG, Romeo A, Fantoni A (1983) Predominant expression of zeta and epsilon globin genes in human leukemia K-562(S6) variant cell line. Experientia 39:415–416
Gambari R, del Senno L, Barbieri R, Viola L, Tripodi M, Raschella G, Fantoni A (1984) Human leukemia K-562 cells: induction of erythroid differentiation by 5-azacytidine. Cell Differ 14:87–97
Gambari R, Barbieri R, Buzzoni D, Bernardi F, Marchetti G, Amelotti F, Piva R, Viola L, del Senno L (1986) Human leukemic K562 cells: suppression of hemoglobin accumulation by a monoclonal antibody to human transferrin receptor. Biochim Biophys Acta 886:203–213
Gonzalez VM, Fuertes MA, Alonso C, Perez JM (2001) Is cisplatin-induced cell death always produced by apoptosis? Mol Pharmacol 59:657–663
He Q, Yuan LB (2007) Dopamine inhibits proliferation, induces differentiation and apoptosis of K562 leukaemia cells. Chin Med J (Engl) 120:970–974
Hiep N, Kinohira S, Furuyama K, Taketani S (2012) Depletion of glutamine enhances sodium butyrate-induced erythroid differentiation of K562 cells. J Biochem 152:509–519
Huang P, Plunkett W (1995) Fludarabine- and gemcitabine-induced apoptosis: incorporation of analogs into DNA is a critical event. Cancer Chemother Pharmacol 36:181–188
Huo XF, Yu J, Peng H, Du ZW et al (2006) Differential expression changes in K562 cells during the hemin-induced erythroid differentiation and the phorbol myristate acetate (PMA)-induced megakaryocytic differentiation. Mol Cell Biochem 292:155–167
Li L, Zhang G, Zhang Y, Tan J, Huang H, Huang B, Lu J (2008) Sodium butyrate-induced upregulation of p18(INK4C) gene affects K562 cell G(0)/G(1) arrest and differentiation. Mol Cell Biochem 319:9–15
Mymryk JS, Zaniewski E, Archer TK (1995) Cisplatin inhibits chromatin remodeling, transcription factor binding, and transcription from the mouse mammary tumor virus promoter in vivo. Proc Natl Acad Sci USA 92:2076–2080
Newsholme P, Costa Rosa LF, Newsholme EA, Curi R (1996) The importance of fuel metabolism to macrophage function. Cell Biochem Funct 14:1–10
Osti F, Corradini FG, Hanau S, Matteuzzi M, Gambari R (1997) Human leukemia K562 cells: induction to erythroid differentiation by guanine, guanosine and guanine nucleotides. Haematologica 82:395–401
Pajak B, Orzechowski A, Gajkowska B (2007) Molecular basis of sodium butyrate-dependent proapoptotic activity in cancer cells. Adv Med Sci 52:83–88
Park JI, Choi HS, Jeong JS, Han JY, Kim IH (2001) Involvement of p38 kinase in hydroxyurea-induced differentiation of K562 cells. Cell Growth Differ 12:481–486
Parodi MT, Tonini GP, Bologna R, Franchini E, Cornaglia-Ferraris P (1988) Cisplatin-induced erythroid differentiation in K562 cells: modulation of transferrin receptor. Boll Ist Sieroter Milan 67:142–148
Rosenberg B, Vancamp L, Krigas T (1965) Inhibition of cell division in Escherichia coli by electrolysis products from a platinum electrode. Nature 205:698–699
Rutherford TR, Clegg JB, Weatherall DJ (1979) K562 human leukaemic cells synthesise embryonic haemoglobin in response to haemin. Nature 280:164–165
Shelton LM, Huysentruyt LC, Seyfried TN (2010) Glutamine targeting inhibits systemic metastasis in the VM-M3 murine tumor model. Int J Cancer 127:2478–2485
Takagaki K, Katsuma S, Kaminishi Y, Horio T, Tanaka T, Ohgi T, Yano J (2005) Role of Chk1 and Chk2 in Ara-C-induced differentiation of human leukemia K562 cells. Genes Cells 10:97–106
Witt O, Sand K, Pekrun A (2000) Butyrate-induced erythroid differentiation of human K562 leukemia cells involves inhibition of ERK and activation of p38 MAP kinase pathways. Blood 95:2391–2396
Woessmann W, Zwanzger D, Borkhardt A (2004) ERK signaling pathway is differentially involved in erythroid differentiation of K562 cells depending on time and the inducing agent. Cell Biol Int 28:403–410
Yang J, Kawai Y, Hanson RW, Arinze IJ (2001) Sodium butyrate induces transcription from the G alpha(i2) gene promoter through multiple Sp1 sites in the promoter and by activating the MEK-ERK signal transduction pathway. J Biol Chem 276:25742–25752
Yuan JY, Wang F, Yu J, Yang GH, Liu XL, Zhang JW (2009) MicroRNA-223 reversibly regulates erythroid and megakaryocytic differentiation of K562 cells. J Cell Mol Med 13:4551–4559
Acknowledgments
We are deeply grateful to Tehran University of Medical Sciences deputy of research (Grant No. 92-03-87-21886) and Iran national science foundation (Grant No. 92005949) due to their support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
Rights and permissions
About this article
Cite this article
Shariati, L., Modaress, M., Khanahmad, H. et al. Comparison of different methods for erythroid differentiation in the K562 cell line. Biotechnol Lett 38, 1243–1250 (2016). https://doi.org/10.1007/s10529-016-2101-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-016-2101-8